Abstract
Summary
The study is about the regulatory effects of estrogen and parathyroid hormone (PTH) on sclerostin, a protein that inhibits the Wnt/β-catenin pathway. The results indicate that estrogen may down-regulate sclerostin expression and that estrogen displays synergistic action with PTH. These results provide a new perspective on the relationship between estrogen and bone.
Purpose
To investigate whether estrogen can down-regulate SOST and MEF2 (myocyte enhancer factor 2) expression and whether co-treatment with estrogen and PTH has a stronger effect on suppressing SOST than PTH applied alone in ovariectomized rats.
Methods
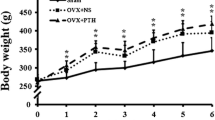
Forty-three-month-old virgin female Sprague–Dawley (SD) rats were ovariectomized and divided into four groups (n = 10). Another ten age-matched rats received sham operations as controls. After allowing 8 weeks for the development of vertebral osteopenia, the rats were administered the drug intervention. For this intervention, the estrogen group was subcutaneously injected with 17β-estradiol at 25 μg/kg body weight, the PTH group was injected with 80 μg/kg synthetic human PTH (1–34), and the co-treatment group was concurrently treated with PTH and estrogen at the above dosage. The OVX group and sham group were treated with vehicle. The drug treatment was conducted for 12 weeks. After the lumbar spine bone mineral density (BMD) was measured, the rats were sacrificed, and the lumbar spine and blood were collected for qPCR, Western blot, immunohistochemistry and other tests.
Results
Estrogen can down-regulate MEF2 and sclerostin expression, and co-treatment with estrogen and PTH has a stronger effect on suppressing MEF2 and SOST mRNA than PTH alone. The co-treatment group displayed slightly higher bone mass and biomechanical properties than the PTH group, but the differences were not significant.
Conclusions
Estrogen appears to be a regulator of sclerostin, and the effect may involve suppressing MEF2s. Combined treatment with PTH and estrogen is not more beneficial for vertebral bone mass and strength than treatment with PTH alone in ovariectomized rats.






Similar content being viewed by others
References
Balemans W, Ebeling M, Patel N et al (2001) Increased bone density in sclerosteosis is due to the deficiency of a novel secreted protein (SOST). Hum Mol Genet 10(5):537–543
Brunkow ME, Gardner JC, Van Ness J et al (2001) Bone dysplasia sclerosteosis results from loss of the SOST gene product, a novel cystine knot-containing protein. Am J Hum Genet 68(3):577–589
Staehling-Hampton K, Proll S, Paeper BW et al (2002) A 52-kb deletion in the SOST–MEOX1 intergenic region on 17q12–q21 is associated with van Buchem disease in the Dutch population. Am J Med Genet 110(2):144–152
Balemans W, Patel N, Ebeling M et al (2002) Identification of a 52 kb deletion downstream of the SOST gene in patients with van Buchem disease. J Med Genet 39(2):91–97
Winkler DG, Sutherland MK, Geoghegan JC et al (2003) Osteocyte control of bone formation via sclerostin, a novel BMP antagonist. EMBO J 22(23):6267–6276
Poole KE, van Bezooijen RL, Loveridge N et al (2005) Sclerostin is a delayed secreted product of osteocytes that inhibits bone formation. FASEB J 19(13):1842–1844
van Bezooijen RL, Roelen BA, Visser A et al (2004) Sclerostin is an osteocyte-expressed negative regulator of bone formation, but not a classical BMP antagonist. J Exp Med 199(6):805–814
Xu HG, Li ZR, Wang H et al (2013) Intermittent cyclic mechanical tension-induced down-regulation of ectonucleotide pyrophosphatase phosphodiesterase 1 gene expression is mainly dependent on TGF-beta1 in end-plate chondrocytes. Orthop Surg 5(1):40–45
Robling AG, Niziolek PJ, Baldridge LA et al (2008) Mechanical stimulation of bone in vivo reduces osteocyte expression of Sost/sclerostin. J Biol Chem 283(9):5866–5875
Semenov M, Tamai K, He X (2005) SOST is a ligand for LRP5/LRP6 and a Wnt signaling inhibitor. J Biol Chem 280(29):26770–26775
Ott SM (2005) Sclerostin and Wnt signaling—the pathway to bone strength. J Clin Endocrinol Metab 90(12):6741–6743
Li X, Zhang Y, Kang H et al (2005) Sclerostin binds to LRP5/6 and antagonizes canonical Wnt signaling. J Biol Chem 280(20):19883–19887
Hodsman AB, Bauer DC, Dempster DW et al (2005) Parathyroid hormone and teriparatide for the treatment of osteoporosis: a review of the evidence and suggested guidelines for its use. Endocr Rev 26(5):688–703
Rubin MR, Bilezikian JP (2005) Parathyroid hormone as an anabolic skeletal therapy. Drugs 65(17):2481–2498
Bellido T, Ali AA, Gubrij I et al (2005) Chronic elevation of parathyroid hormone in mice reduces expression of sclerostin by osteocytes: a novel mechanism for hormonal control of osteoblastogenesis. Endocrinology 146(11):4577–4583
Silvestrini G, Ballanti P, Leopizzi M et al (2007) Effects of intermittent parathyroid hormone (PTH) administration on SOST mRNA and protein in rat bone. J Mol Histol 38(4):261–269
Sims NA (2010) Building bone with a SOST − PTH partnership. J Bone Miner Res 25(2):175–177
Kramer I, Keller H, Leupin O et al (2010) Does osteocytic SOST suppression mediate PTH bone anabolism? Trends Endocrinol Metab 21(4):237–244
Kramer I, Loots GG, Studer A et al (2010) Parathyroid hormone (PTH)-induced bone gain is blunted in SOST overexpressing and deficient mice. J Bone Miner Res 25(2):178–189
Mirza FS, Padhi ID, Raisz LG et al (2010) Serum sclerostin levels negatively correlate with parathyroid hormone levels and free estrogen index in postmenopausal women. J Clin Endocrinol Metab 95(4):1991–1997
Drake MT, Srinivasan B, Modder UI et al (2010) Effects of parathyroid hormone treatment on circulating sclerostin levels in postmenopausal women. J Clin Endocrinol Metab 95(11):5056–5062
Leupin O, Kramer I, Collette NM et al (2007) Control of the SOST bone enhancer by PTH using MEF2 transcription factors. J Bone Miner Res 22(12):1957–1967
Ardawi MS, Al-Kadi HA, Rouzi AA et al (2011) Determinants of serum sclerostin in healthy pre- and postmenopausal women. J Bone Miner Res 26(12):2812–2822
Gatti D, Viapiana O, Adami S et al (2012) Bisphosphonate treatment of postmenopausal osteoporosis is associated with a dose dependent increase in serum sclerostin. Bone 50(3):739–742
Gooi JH, Pompolo S, Karsdal MA et al (2010) Calcitonin impairs the anabolic effect of PTH in young rats and stimulates expression of sclerostin by osteocytes. Bone 46(6):1486–1497
Shen V, Birchman R, Xu R et al (1995) Effects of reciprocal treatment with estrogen and estrogen plus parathyroid hormone on bone structure and strength in ovariectomized rats. J Clin Invest 96(5):2331–2338
Kobayashi S, Shimizu T, Mehdi R et al (1999) Advantages of concurrent use of anabolic and antiresorptive agents over single use of these agents in increasing trabecular bone volume, connectivity, and biomechanical competence of rat vertebrae. Bone 25(6):703–712
Shen V, Dempster DW, Mellish RW et al (1992) Effects of combined and separate intermittent administration of low-dose human parathyroid hormone fragment (1–34) and 17 beta-estradiol on bone histomorphometry in ovariectomized rats with established osteopenia. Calcif Tissue Int 50(3):214–220
Qi H, Li M, Wronski TJ (1995) A comparison of the anabolic effects of parathyroid hormone at skeletal sites with moderate and severe osteopenia in aged ovariectomized rats. J Bone Miner Res 10(6):948–955
Shen V, Dempster DW, Birchman R et al (1993) Loss of cancellous bone mass and connectivity in ovariectomized rats can be restored by combined treatment with parathyroid hormone and estradiol. J Clin Invest 91(6):2479–2487
Wronski TJ, Cintron M, Doherty AL et al (1988) Estrogen treatment prevents osteopenia and depresses bone turnover in ovariectomized rats. Endocrinology 123(2):681–686
Zhao M, Kim YT, Yoon BS et al (2006) Expression profiling of cyclin B1 and D1 in cervical carcinoma. Exp Oncol 28(1):44–48
Henslee AM, Spicer PP, Yoon DM et al (2011) Biodegradable composite scaffolds incorporating an intramedullary rod and delivering bone morphogenetic protein-2 for stabilization and bone regeneration in segmental long bone defects. Acta Biomater 7(10):3627–3637
Garnero P, Sornay-Rendu E, Chapuy MC et al (1996) Increased bone turnover in late postmenopausal women is a major determinant of osteoporosis. J Bone Miner Res 11(3):337–349
Canpolat S, Tug N, Seyran AD et al (2010) Effects of raloxifene and estradiol on bone turnover parameters in intact and ovariectomized rats. J Physiol Biochem 66(1):23–28
Riggs BL, Khosla S, Melton LR (2002) Sex steroids and the construction and conservation of the adult skeleton. Endocr Rev 23(3):279–302
Jastrzebski S, Kalinowski J, Stolina M et al (2013) Changes in bone sclerostin levels in mice after ovariectomy vary independently of changes in serum sclerostin levels. J Bone Miner Res 28(3):618–626
Brand NJ (1997) Myocyte enhancer factor 2 (MEF2). Int J Biochem Cell Biol 29(12):1467–1470
Genetos DC, Toupadakis CA, Raheja LF et al (2010) Hypoxia decreases sclerostin expression and increases Wnt signaling in osteoblasts. J Cell Biochem 110(2):457–467
Gatti D, Viapiana O, Fracassi E et al (2012) Sclerostin and DKK1 in postmenopausal osteoporosis treated with denosumab. J Bone Miner Res 27(11):2259–2263
Finkelstein JS, Wyland JJ, Lee H et al (2010) Effects of teriparatide, alendronate, or both in women with postmenopausal osteoporosis. J Clin Endocrinol Metab 95(4):1838–1845
Martin TJ, Sims NA (2005) Osteoclast-derived activity in the coupling of bone formation to resorption. Trends Mol Med 11(2):76–81
Acknowledgments
This study was supported by grants from the National Natural Science Foundation of China (No. 81102607), the Key Technologies & Program of Tianjin (No. 13ZCZDSY01700 and 11ZCGYSY01800) and the Scientific and Technological Project of Tianjin Public Health Bureau (No. 11KG137 and No. 12KG120).
Conflicts of interest
None
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jia, H., Ma, J., Ma, X. et al. Estrogen alone or in combination with parathyroid hormone can decrease vertebral MEF2 and sclerostin expression and increase vertebral bone mass in ovariectomized rats. Osteoporos Int 25, 2743–2754 (2014). https://doi.org/10.1007/s00198-014-2818-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-014-2818-y