Abstract
Key message
This manuscript describes the identification, isolation and sequencing of a single chromosome containing high value resistance genes from a complex polyploid where sequencing the whole genome is too costly.
Abstract
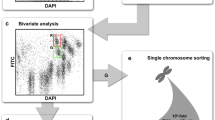
The large complex genomes of many crops constrain the use of new technologies for genome-assisted selection and genetic improvement. One method to simplify a genome is to break it into individual chromosomes by flow cytometry; however, in many crop species most chromosomes cannot be isolated individually. Flow sorting of a single copy of a chromosome has been developed in wheat, and here we demonstrate its use to identify markers of interest in an Erianthus/Sacchurum hybrid. Erianthus/Saccharum hybrids are of interest because Erianthus is known to be highly resistant to soil borne diseases which cause extensive sugarcane yield losses in Australia. Sugarcane (Saccharum) cultivars are autopolyploids with a highly complex genome and over 100 chromosomes. Flow cytometry for sugarcane, as in most crops, does not resolve individual chromosomes to a karyotype peak for sorting. To isolate a single chromosome, we used genomic in situ hybridization (GISH) to identify the flow karyotype region containing the Erianthus chromosomes, flow sorted single chromosomes from this region, PCR screened for the Erianthus chromosomes and sequenced them. One Erianthus chromosome amplified and sequenced well, and from this data we could identify 57 resistant type genes and SNPs in nearly half of these genes. We developed KASP SNP assays and demonstrated that the identified SNP markers segregated as expected in a small introgression population. The pipeline we developed here to flow sort and sequence single chromosomes could be used in any crop with a large complex genome to rapidly discover and develop markers to important loci.





Similar content being viewed by others
Availability of data and material
All data are available on request to corresponding author.
Code Availability
Not applicable.
Abbreviations
- WGA:
-
Whole Genome Amplification
- FISHIS:
-
Fluorescence in situ Hybridization in Suspension
- GISH:
-
Genomic in situ Hybridization
- MDA:
-
Multiple Displacement Amplification
- SNP:
-
Single Nucleotide Polymorphism
- STP:
-
Single Tiling Path
References
Aitken K, Li J, Piperidis G, Qing C, Yuanhong F, Jackson P (2018) Worldwide genetic diversity of the wild species and level of diversity captured within sugarcane breeding programs. Crop Sci 58:218. https://doi.org/10.2135/cropsci2017.06.0339
Aitken KS, NcNeil M (2010) Diversity analysis. In: Henry R, Kole C (eds) Genetics genomics and breeding of sugarcane. Science Publishers: Enfield, USA, pp. 19–42 ISBN 978-57808-684-9.
Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ (1990) Basic local alignment search tool. J Mol Biol 215:403–410. https://doi.org/10.1016/S0022-2836(05)80360-2
Andrews S (2015) FastQC: a quality control tool for high throughput sequence data [Online]. Available online: http://www.bioinformatics.babraham.ac.uk/projects/fastqc/.
Arceneaux G (1965) Cultivated sugarcane of the world and their botanical derivation. Proc Int Soc Sugarcane Technol 12:844–854
Arumuganathan K, Martin GB, Telenius H, Tanksley SD, Earle ED (1994) Chromosome 2-specific DNA clones from flow-sorted chromosomes of tomato. Mol Gen Genet 242:551–558. https://doi.org/10.1007/BF00285278
Begum MC, Islam M, Sarkar MR, Azad MAS, Huda AKMN, Kabir AH (2016) Auxin signaling is closely associated with Zn-efficiency in rice (Oryza sativa L.). J Plant Interact 11:124–129. https://doi.org/10.1080/17429145.2016.1220026.
Besse P, McIntyre CL (1998) Isolation and characterisation of repeated DNA sequences from Erianthus spp. (Saccharinae: Andropogoneae). Genome 41:408–416. https://doi.org/10.1139/g98-034.
Bhuiyan SA, Croft BJ, Stirling GR, Wong E, Jackson P, Cox M (2016) Assessment of resistanc to root-lesion and root-knot nematodes in Australian hybrid clones of sugarcane and its wild relatives. Aust Plant Pathol 45:165–173. https://doi.org/10.1007/s13313-016-0400-0
Blair BL, Stirling GR (2007) The role of plant-parasitic nematodes in reducing yield of sugarcane in fine-textured soils in Queensland. Aust Aust J Exp Agric 47:620–634. https://doi.org/10.1071/EA05287
Bolger AM, Lohse M, Usadel B (2014) Trimmomatic: a flexible trimmer for illumina sequence data. Bioinformatics 30:2114–2120. https://doi.org/10.1093/bioinformatics/btu170
Cai Q, Aitken K, Deng HH, Chen XW, Fu C, Jackson PA, McIntyre CL (2005) Verification of the introgression of Erianthus arundinaceus germplasm into sugarcane using molecular markers. Plant Breed 124:322–328. https://doi.org/10.1111/j.1439-0523.2005.01099.x
Cápal P, Blavet N, Vrána J, Kubaláková M, Doležel J (2015) Multiple displacement amplification of the DNA from single flow-sorted plant chromosome. Plant J 84:838–844. https://doi.org/10.1111/tpj.13035
Cápal P, Endo TR, Vrána J, Kubaláková M, Karafiátová M, Komínková E, Mora-Ramírez I, Weschke W, Doležel J (2016) The utility of flow sorting to identify chromosomes carrying a single copy transgene in wheat. Plant Methods 12:24. https://doi.org/10.1186/s13007-016-0124-8
Cheung VG, Nelson SF (1996) Whole genome amplification using a degenerate oligonucleotide primer allows hundreds of genotypes to be performed on less than one nanogram of genomic DNA. Proc Natl Acad Sci U S A 93:14676–14679. https://doi.org/10.1073/pnas.93.25.14676
CIMMYT (2005) Laboratory protocols: CIMMYT applied molecular genetics laboratory, 3rd edn. CIMMYT: Mexico, D.F., 2005; ISBN: 9686923306.
Dean FB, Hosono S, Fang L, Wu X, Faruqi AF, Bray-Ward P, Sun Z, Zong Q, Du Y, Du J et al (2002) Comprehensive human genome amplification using multiple displacement amplification. Proc Natl Acad Sci U S A 99:5261–5266. https://doi.org/10.1073/pnas.082089499
Deng HH, Liao ZZ, Li QW, Lao FY, Fu C, Chen XW, Zhang CM, Liu SM, Yang YH (2002) Breeding and isozyme marker assisted selection of F2 hybrids from Saccharum spp. x Erianthus arundinaceus. Sugarcane Canesugar 1:1–5.
D’Hont A (2005) Unraveling the genome structure of polyploids using FISH and GISH; examples of sugarcane and banana. Cytogenet Genome Res 109:27–33. https://doi.org/10.1159/000082378
D’Hont A, Glaszmann JC (2001) Sugarcane genome analysis with molecular markers: a first decade of research. In International Society of Sugar Cane Technologists. Proceedings of the XXIV Congress, pp. 556–559.
D’Hont A, Grivet L, Feldmann P, Rao S, Berding N, Glaszmann JC (1996) Characterisation of the double genome structure of modern sugarcane cultivars (Saccharum spp.) by molecular cytogenetics. Mol Gen Genet 250:405–413.
D’Hont A, Ison D, Alix K, Roux C, Glaszmann JC (1998) Determination of basic chromosome numbers in the genus Saccharum by physical mapping of ribosomal RNA genes. Genome 41:221–225. https://doi.org/10.1139/gen-41-2-221
Domingues DS, Cruz GMQ, Metcalfe CJ, Nogueira FTS, Vicentini R, de Alves SC, Van Sluys M-A (2012) Analysis of plant LTR-retrotransposons at the fine-scale family level reveals individual molecular patterns. BMC Genomics 13:137. https://doi.org/10.1186/1471-2164-13-137.
Fan HC, Wang J, Potanina A, Quake SR (2011) Whole-genome molecular haplotyping of single cells. Nat Biotechnol 29:51–57. https://doi.org/10.1038/nbt.1739
Fu J, Wang S (2011) Insights into auxin signaling in plant-pathogen interactions. Front Plant Sci 2:1–7. https://doi.org/10.3389/fpls.2011.00074
Garsmeur O, Droc G, Antonise R, Grimwood J, Potier B, Aitken K, Jenkins J, Martin G, Charron C, Hervouet C et al (2018) A mosaic monoploid reference sequence for the highly complex genome of sugarcane. Nat Commun 9:2638. https://doi.org/10.1038/s41467-018-05051-5
Giorgi D, Farina A, Grosso V, Gennaro A, Ceoloni C, Lucretti S (2013) FISHIS: fluorescence in situ hybridization in suspension and chromosome flow sorting made easy. PLoS ONE 8:e57994. https://doi.org/10.1371/journal.pone.0057994
Grotz N, Fox T, Connolly E, Park W, Guerinot M, Lou Eide D (1998) Identification of a family of zinc transporter genes from Arabidopsis that respond to zinc deficiency. Proc Natl Acad Sci U S A 95:7220–7224. https://doi.org/10.1073/pnas.95.12.7220.
Hotta CT, Lembke CG, Domingues DS, Ochoa EA, Cruz GMQ, Melotto-Passarin DM, Marconi TG, Santos MO, Mollinari M, Margarido GRA et al (2010) The biotechnology roadmap for sugarcane improvement. Trop Plant Biol 3:75–87. https://doi.org/10.1007/s12042-010-9050-5
Kellogg EA (1998) Relationships of cereal crops and other grasses. Proc Natl Acad Sci U S A 95:2005–2010. https://doi.org/10.1073/pnas.95.5.2005
Kirkness EF, Grindberg RV, Yee-Greenbaum J, Marshall CR, Scherer SW, Lasken RS, Venter JC (2013) Sequencing of isolated sperm cells for direct haplotyping of a human genome. Genome Res 23:826–832. https://doi.org/10.1101/gr.144600.112
Kreplak J, Madoui MA, Cápal P, Novák P, Labadie K, Aubert G, Bayer P, Gali KK, Syme RA, Main D (2019) Areference genome for pea provides insight into legume genome evolution. Nat Genet 51:1411–1422
Lasswell J, Rogg LE, Nelson DC, Rongey C, Bartel B (2000) Cloning and characterization of IAR1, a gene required for auxin conjugate sensitivity in arabidopsis. Plant Cell 12:2395–2408. https://doi.org/10.1105/tpc.12.12.2395
Li H (2013) Aligning sequence reads, clone sequences and assembly contigs with BWA-MEM. Available online: arxiv:1303.3997v1 [q-bio.GN].
Li Li H, Handsaker B, Wysoker A, Fennell T, Ruan J, Homer N, Marth G, Abecasis G, Durbin R (2009) The sequence alignment/map format and SAMtools. Bioinformatics 25:2078–2079. https://doi.org/10.1093/bioinformatics/btp352
Lizardi PM, Huang X, Zhu Z, Bray-Ward P, Thomas DC, Ward DC (1998) Mutation detection and single-molecule counting using isothermal rolling-circle amplification. Nat Genet 19:225–232. https://doi.org/10.1038/898
Ma L, Xiao Y, Huang H, Wang Q, Rao W, Feng Y, Zhang K, Song Q (2010) Direct determination of molecular haplotypes by chromosome microdissection. Nat Methods 7:299–301. https://doi.org/10.1038/nmeth.1443
Magarey RC, Bull JI (2003) Relating cultivar Pachymetra root rot resistance to sugarcane yield using breeding selection trial analyses. Aust J Exp Agric 43:617–622. https://doi.org/10.1071/EA02144
Magarey R, Croft B (1996) Pachymetra root rot: Incidence and potential solutions to its influence on yield decline in Queensland. In: Wilson J, Hogarth D, Campbell J, Garside A (eds) Sugarcane: research towards efficient and sustainable production. CSIRO Division of Tropical Crops and Pastures: Brisbane, Australia, pp. 151–152.
Marcy Y, Ishoey T, Lasken RS, Stockwell TB, Walenz BP, Halpern AL, Beeson KY, Goldberg SMD, Quake SR (2007) Nanoliter reactors improve multiple displacement amplification of genomes from single cells. PLOS Genet 3:1702–1708. https://doi.org/10.1371/journal.pgen.0030155
Matsubara K, Iwasaki Y, Nishiki I, Nomura K, Fujiwara A (2018) Identification of genetic linkage group 1-linked sequences in Japanese eel (Anguilla japonica) by single chromosome sorting and sequencing. PLoS ONE 13:e0197040. https://doi.org/10.1371/journal.pone.0197040
Meng Z, Han J, Lin Y, Zhao Y, Lin Q, Wang J, Zhang M, Zhang L, Yang Q, Wang K (2019) Characterisation of a Saccharum spontaneum with a basic chromosomes number of x=10 provides new insights on genome evolution in genus Saccharum. Theor Appl Genet. https://doi.org/10.1007/s00122-019-03450-w
Metcalfe CJ, Li J, Giorgi D, Doležel J, Piperidis N, Aitken KS (2019) Flow cytometric characterisation of the complex polyploid genome of Saccharum officinarum and modern sugarcane cultivars. Sci Rep 9:19362. https://doi.org/10.1038/s41598-019-55652-3
Paunio T, Reima I, Syvänen AC (1996) Preimplantation diagnosis by whole-genome amplification, PCR amplification, and solid-phase minisequencing of blastomere DNA. Clin Chem 42:1382–1390
Piperidis N (2014) GISH: Resolving Interspecifc and Intergeneric Hybrids. In: Besse P (ed) Molecular plant taxonomy: methods and protocols, methods in molecular biology. Springer Science+Business Media: New York, Vol. 1115, pp. 325–336. ISBN 978-1-62703-766-2.
Piperidis N, Chen J, Deng H, Wang L-P, Jackson P, Piperidis G (2010a) GISH characterization of Erianthus arundinaceus chromosomes in three generations of sugarcane intergeneric hybrids. Genome 53:331–336. https://doi.org/10.1139/G10-010
Piperidis G, Piperidis N, D’Hont A (2010b) Molecular cytogenetic investigation of chromosome composition and transmission in sugarcane. Mol Genet Genomics 284:65–73. https://doi.org/10.1007/s00438-010-0546-3
Piperidis N, Tom C, Aitken KS, Atkin FC, Piperidis G (2019) Exploiting Erianthus diversity to enhance sugarcane cultivars. In: 41st Annu. Conf. -Aust. Soc. Sugar Cane Technol. 2019, vol 41, pp 108–116.
Quinlan AR, Hall IM (2010) BEDTools: a flexible suite of utilities for comparing genomic features. Bioinformatics 26:841–842. https://doi.org/10.1093/bioinformatics/btq033
Rhee M, Light YK, Meagher RJ, Singh AK (2016) Digital droplet multiple displacement amplification (ddMDA) for whole genome sequencing of limited DNA samples. PLoS ONE 11:e0153699. https://doi.org/10.1371/journal.pone.0153699
Roach B (1972) Nobilisation of sugarcane. Proc Int Soc Sugar Cane Technol 14:206–216
Sánchez-Martín J, Steuernagel B, Ghosh S, Herren G, Hurni S, Adamski N, Vrána J, Kubaláková M, Krattinger SG, Wicker T et al (2016) Rapid gene isolation in barley and wheat by mutant chromosome sequencing. Genome Biol 17:1–7. https://doi.org/10.1186/s13059-016-1082-1
Santchurn D, Ramdoyal K, Badaloo MGH, Labuschagne MT (2014) From sugar industry to cane industry: evaluation and simultaneous selection of different types of high biomass canes. Biomass Bioenerg 61:82–92. https://doi.org/10.1016/j.biombioe.2013.11.023
Sayers EW, Barrett T, Benson DA, Bolton E, Bryant SH, Canese K, Chetvernin V, Church DM, DiCuccio M, Federhen S et al (2011) Database resources of the National Center for Biotechnology Information. Nucleic Acids Res 39:D38–D51. https://doi.org/10.1093/nar/gkq1172
Spits C, Le Caignec C, De Rycke M, Van Haute L, Van Steirteghem A, Liebaers I, Sermon K (2006) Whole-genome multiple displacement amplification from single cells. Nat Protoc 1:1965–1970. https://doi.org/10.1038/nprot.2006.326
Telenius H, Carter NP, Bebb CE, Nordenskjöld M, Ponder BAJ, Tunnacliffe A (1992) Degenerate oligonucleotide-primed PCR: general amplification of target DNA by a single degenerate primer. Genomics 13:718–725. https://doi.org/10.1016/0888-7543(92)90147-K
Untergasser A, Nijveen H, Rao X, Bisseling T, Geurts R, Leunissen JAM (2007) Primer3Plus, an enhanced web interface to Primer3. Nucleic Acids Res 35:W71–W74. https://doi.org/10.1093/nar/gkm306
Vrána J, Cápal P, Šimková H, Karafiátová M, Čížková J, Doležel J (2016) Flow analysis and sorting of plant chromosomes. Curr Protoc Cytom 78:5.3.1–5.3.43, https://doi.org/10.1002/cpcy.9.
Waclawovsky AJ, Sato PM, Lembke CG, Moore PH, Souza GM (2010) Sugarcane for bioenergy production: an assessment of yield and regulation of sucrose content. Plant Biotechnol J 8:263–276. https://doi.org/10.1111/j.1467-7652.2009.00491.x
Wang X, Heazlewood SP, Krause DO, Florin THJ (2003) Molecular characterization of the microbial species that colonize human ileal and colonic mucosa by using 16S rDNA sequence analysis. J Appl Microbiol 95:508–520. https://doi.org/10.1046/j.1365-2672.2003.02005.x
Wu J, Huang Y, Lin Y, Fu C, Liu S, Deng Z, Li Q, Huang Z, Chen R, Zhang M (2014) Unexpected inheritance pattern of Erianthus arundinaceus chromosomes in the intergeneric progeny between Saccharum spp. and Erianthus arundinaceus. PLoS One 9. https://doi.org/10.1371/journal.pone.0110390.
Yang H, Chen X, Wong WH (2011) Completely phased genome sequencing through chromosome sorting. Proc Natl Acad Sci U S A 108:12–17. https://doi.org/10.1073/pnas.1016725108
Zhang L, Cui X, Schmitt K, Hubert R, Navidi W, Arnheim N (1992) Whole genome amplification from a single cell: Implications for genetic analysis. Proc Natl Acad Sci U S A 89:5847–5851. https://doi.org/10.1073/pnas.89.13.5847
Zhang J, Zhou M, Walsh J, Zhu L, Chen Y, Ming R (2014) Sugarcane genetics and genomics. Sugarcane Physiol Biochem Funct Biol, pp 623–643, https://doi.org/10.1002/9781118771280.ch23.
Acknowledgements
We would like to acknowledge the support of Sugar Research Australia who provided the genotype for flow sorting. We would also like to thank Dalia Khalil from the TRI Flow Cytometry Facility at the Translational Research Institute who was instrumental in getting the flow sorting parameters correct for sugarcane. This work was conducted in collaboration by the Joint Genome Institute and is supported by the Office of Science in the U.S. Department of Energy under Contract No. DE-AC02-05CH11231.
Funding
Work was funded by CSIRO, Sugar Research Australia.
Author information
Authors and Affiliations
Contributions
Conceptualization, KSA and NP; Funding acquisition, KSA and NP; Investigation, CJM and JL; Formal analysis, CJM, AH, BZ and JS; Original draft preparation, CJM; Review and editing, KSA, CJM and NP; Supervision, KSA.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethics approval
Not applicable.
Consent for publication
All authors have read and agreed to the published version of the manuscript.
Additional information
Communicated by Maria Laura Federico.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Metcalfe, C.J., Li, J., Zheng, B. et al. Isolation and sequencing of a single copy of an introgressed chromosome from a complex genome for gene and SNP identification. Theor Appl Genet 135, 1279–1292 (2022). https://doi.org/10.1007/s00122-022-04030-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00122-022-04030-1