Abstract
Key message
Catalytically active indica SSIIa allele in high amylose rice with down-regulated japonica SBEIIb can increase starch content and modify the starch structure and properties without changing its amylose content.
Abstract
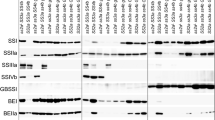
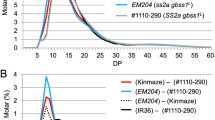
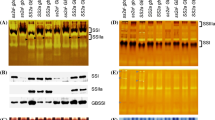
Rice (Oryza sativa) genotypes with inactive starch synthase IIa (SSIIa) with recessive variants of starch branching enzyme IIb (SBEIIb) exhibit a range of alterations in grain phenotype, starch granule morphology, starch granule bound proteins, starch structure, and functional properties. However, the interactions between the two enzymes have not been thoroughly investigated yet. We analysed recombinant rice lines having down-regulated SBEIIb expression (SBEIIbDR) with either indica or japonica type SSIIa (SSIIaind or SSIIajap). In SBEIIbDR rice starch granules, the increased abundance of two protein bands (SSI and SSIIa) was found with eight additional protein bands not generally associated with starch granules. The amount of SSIIa was higher in SSIIaindSBEIIbDR than SSIIajapSBEIIbDR, which indicated that indica type SSIIa, possibly in the monomer form, was extensively involved in starch biosynthesis in the SBEIIbDR endosperm. Furthermore, SSIIaindSBEIIbDR grains had higher total starch content and higher starch swelling power than SSIIajapSBEIIbDR lines, but the amylopectin gelatinization temperatures and enthalpy and the apparent amylose content remained similar. In summary, this work suggests that SSIIaind can partly compensate for the alteration of starch synthesis resulting from the SBEIIb down-regulation in japonica background without reducing its amylose content. The study provides insight into the starch structural and textural improvements of high amylose starch.








Similar content being viewed by others
Data availability
All data generated or analysed during this study are included in this published article and its supplementary information file.
References
Abe N, Asai H, Yago H, Oitome NF, Itoh R, Crofts N, Nakamura Y, Fujita N (2014) Relationships between starch synthase I and branching enzyme isozymes determined using double mutant rice lines. BMC Plant Biol 14:80–91
Ahmed Z, Tetlow IJ, Ahmed R, Morell MK, Emes MJ (2015) Protein-protein interactions among enzymes of starch biosynthesis in high-amylose barley genotypes reveal the differential roles of heteromeric enzyme complexes in the synthesis of A and B granules. Plant Sci 233:95–106
Asai H, Abe N, Matsushima R, Crofts N, Oitome NF, Nakamura Y, Fujita N (2014) Deficiencies in both starch synthase IIIa and branching enzyme IIb lead to a significant increase in amylose in SSIIa-inactive japonica rice seeds. J Exp Bot 65:5497–5507
Ball SG, Morell MK (2003) From bacterial glycogen to starch: understanding the biogenesis of the plant starch granule. Annu Rev Plant Biol 54:207–233
Bird AR, Conlon MA, Christophersen CT, Topping DL (2010) Resistant starch, large bowel fermentation and a broader perspective of prebiotics and probiotics. Benef Microbes 1:423–431
Butardo VM, Fitzgerald MA, Bird AR et al (2011) Impact of down-regulation of starch branching enzyme IIb in rice by artificial microRNA- and hairpin RNA-mediated RNA silencing. J Exp Bot 62:4927–4941
Butardo VM, Arrowhead DV, Colgrave ML et al (2012) Analysis of starch and starch granule proteins in the high amylose rice mutant Goami 2. J Agri Food Chem 60:11576–11585
Calingacion M, Laborte A, Nelson A et al (2014) Diversity of global rice markets and the science required for consumer-targeted rice breeding. PLoS ONE 9:e85106
Champ M, Langkilde AM, Brouns F, Kettlitz B, Bail-Collet YL (2003) Advances in dietary fibre characterisation. 2. Consumption, chemistry, physiology and measurement of resistant starch; implications for health and food labelling. Nutr Res Rev 16:143–161
Crofts N, Abe N, Oitome NF, Matsushima R, Hayashi M, Tetlow IJ, Emes MJ, Nakamura Y, Fujita N (2015) Amylopectin biosynthetic enzymes from developing rice seed form enzymatically active protein complexes. J Exp Bot 66:4469–4482
Crofts N, Sugimoto K, Oitome NF, Nakamura Y, Fujita N (2017) Differences in specificity and compensatory functions among three major starch synthases determine the structure of amylopectin in rice endosperm. Plant Mol Biol. https://doi.org/10.1007/s11103-017-0614-8
Crofts N, Iizuka Y, Abe N, Miura S, Kikuchi K, Matsushima R, Fujita N (2018) Rice mutants lacking starch synthase I or branching enzyme IIb activity altered starch biosynthetic protein complexes. Front Plant Sci. https://doi.org/10.3389/fpls.2018.01817
Dhital S, Butardo VM Jr, Jobling SA, Gidley MJ (2015) Rice starch granule amylolysis—differentiating effects of particle size, morphology, thermal properties and crystalline polymorph. Carbohydr Polym 115:305–316
Faisant N, Champ M, Colonna P, Buleon A, Molis C, Langkilde AM, Schweizer T, Flourie B, Galmiche JP (1993) Structural features of resistant starch at the end of the human small-intestine. Eur J Clin Nutr 47:285–296
Fedoroff N, Wessler S, Shure M (1983) Isolation of the transposable maize controlling elements Ac and Ds. Cell 35:235–242
Fitzgerald MA, McCouch SR, Hall RD (2009) Not just a grain of rice: the quest for quality. Trends Plant Sci 14:133–139
Fuentes-Zaragoza E, Sanchez-Zapata E, Sendra E, Sayas E, Navarro C, Fernandez-Lopez J, Perez-Alvarez JA (2011) Resistant starch as prebiotic: a review. Starch-Stärke 63:406–415
Fujita N, Yoshida M, Asakura N, Ohdan T, Miyao A, Hirochika H, Nakamura Y (2006) Function and characterization of starch synthase I using mutants in rice. Plant Physiol 140:1070–1084
Fujita N, Yoshida M, Kondo T et al (2007) Characterization of SSIIIa-Deficient mutants of rice: the function of SSIIIa and pleiotropic effects by SSIIIa deficiency in the rice endosperm. Plant Physiol 144:2009–2023
Grimaud F, Rogniaux H, James MG, Myers AM, Planchot V (2008) Proteome and phosphoproteome analysis of starch granule-associated proteins from normal maize and mutants affected in starch biosynthesis. J Exp Bot 59:3395–3406
Hanashiro I, Higuchi T, Aihara S, Nakamura Y, Fujita N (2011) Structures of starches from rice mutants deficient in the starch synthase isozyme SSI or SSIIIa. Biomacromol 12:1621–1628
Hennen-Bierwagen TA, Liu F, Marsh RS, Kim S, Gan Q, Tetlow IJ, Emes MJ, James MG, Myers AM (2008) Starch biosynthetic enzymes from developing maize endosperm associate in multisubunit complexes. Plant Physiol 146:1892–1908
Hennen-Bierwagen TA, Lin Q, Grimaud F, Planchot V, Keeling PL, James MG, Myers AM (2009) Proteins from multiple metabolic pathways associate with starch biosynthetic enzymes in high molecular weight complexes: a model for regulation of carbon allocation in maize amyloplasts. Plant Physiol 149:1541–1559
Higgins JA, Brown IL (2013) Resistant starch: a promising dietary agent for the prevention/treatment of inflammatory bowel disease and bowel cancer. Curr Opin Gastroenterol 29:190–194
Ischebeck T, Seiler S, Heilmann I (2010) At the poles across kingdoms: phosphoinositides and polar tip growth. Protoplasma 240:13–31
Itoh Y, Crofts N, Abe M, Hosaka Y, Fujita N (2017) Characterization of the endosperm starch and the pleiotropic effectsof biosynthetic enzymes on their properties in novel mutant rice lineswith high resistant starch and amylose content. Plant Sci 258:52–60
Jane J, Chen YY, Lee LF, McPherson AE, Wong KS, Radosavljevic M, Kasemsuwan T (1999) Effects of amylopectin branch chain length and amylose content on the gelatinization and pasting properties of starch. Cereal Chem 76:629–637
Juliano BO (2001) Asian perspective on rice sensory quality. Cereal Food World 46:531–535
Konik-Rose C, Moss R, Rahman S, Appels R, Stoddard F, McMaster G (2001) Evaluation of the 40 mg swelling test for measuring starch functionality. Starch Stärke 53:14–20
Konik-Rose C, Thistleton J, Chanvrier H et al (2007) Effects of starch synthase IIa gene dosage on grain, protein and starch in endosperm of wheat. Theor Appl Genet 115:1053–1065
Kosar-Hashemi B, Li Z, Larroque O, Regina A, Yamamori M, Morell MK, Rahman S (2007) Multiple effects of the starch synthase II mutation in developing wheat endosperm. Func Plant Biol 34:431–438
Kubo A, Akdogan G, Nakaya M, Shojo A, Suzuki S, Satoh H, Kitamura S (2010) Structure, physical, and digestive properties of starch from wx ae double-mutant rice. J Agr Food Chem 58:4463–4469
Li H, Prakash S, Nicholson TM, Fitzgerald MA, Gilbert RG (2016) The importance of amylose and amylopectin fine structure for textural properties of cooked rice grains. Food Chem 196:702–711
Liu F, Makhmoudova A, Lee EA, Wait R, Emes MJ, Tetlow IJ (2009) The amylose extender mutant of maize conditions novel protein-protein interactions between starch biosynthetic enzymes in amyloplasts. J Exp Bot 60:4423–4440
Liu F, Ahmed Z, Lee EA, Donner E, Liu Q, Ahmed R, Morell MK, Emes MJ, Tetlow IJ (2011) Allelic variants of the amylose extender mutation of maize demonstrate phenotypic variation in starch structure resulting from modified protein-protein interactions. J Exp Bot 63:1167–1183
Liu F, Romanova N, Lee EA, Ahmed R, Evans M, Gilbert EP, Morell MK, Emes MJ, Tetlow IJ (2012) Glucan affinity of starch synthase IIa determines binding of starch synthase I and starch branching enzyme IIb to starch granules. The Biochem J 448:373–387
Luo J, Jobling SA, Millar A, Morell MK, Li Z (2015a) Allelic effects on starch structure and properties of six starch biosynthetic genes in a rice recombinant inbred line population. Rice 8:15
Luo J, Regina A, Kosar-Hashemi B et al (2015b) The different effects of starch synthase IIa mutations or variation on endosperm amylose content of barley, wheat and rice are determined by the distribution of starch synthase I and starch branching enzyme IIb between the starch granule and amyloplast stroma. Theor Appl Genet 128:1407–1419
Martin C, Smith AM (1995) Starch Biosynthesis. Plant Cell 7:971–985
Matsumoto K, Maekawa M, Nakaya M, Takemitsu H, Satoh H, Kitamura S (2012) Wx/ae double-mutant brown rice prevents the rise in plasma lipid and glucose levels in mice. Biosci Biotech Biochem 76:2112–2117
Morell MK, Kosar-Hashemi B, Cmiel M, Samuel MS, Chandler P, Rahman S, Buleon A, Batey IL, Li Z (2003) Barley sex6 mutants lack starch synthase IIa activity and contain a starch with novel properties. Plant J 34:172–184
Nakamura Y (2002) Towards a better understanding of the metabolic system for amylopectin biosynthesis in plants: rice endosperm as a model tissue. Plant Cell Physiol 43:718–725
Nakamura Y, Utsumi Y, Sawada T, Aihara S, Utsumi C, Yoshida M, Kitamura S (2010) Characterization of the reactions of starch branching enzymes from rice endosperm. Plant Cell Physiol 51:776–794
Nakamura Y, Aihara S, Crofts N, Sawada T, Fujita N (2014) In vitro studies of enzymatic properties of starch synthases and interactions between starch synthase I and starch branching enzymes from rice. Plant Sci 224:1–8
Nelson OE, Rines HW (1962) The enzymatic deficiency in the waxy mutant of maize. Biochem Biophys Res Commun 9:297–300
Nishi A, Nakamura Y, Tanaka N, Satoh H (2001) Biochemical and genetic analysis of the effects of amylose-extender mutation in rice endosperm. Plant Physiol 127:459–472
O’Shea MG, Samuel MS, Konik CM, Morell MK (1998) Fluorophoreassisted carbohydrate electrophoresis (FACE) of oligosaccharides: efficiency of labeling and high-resolution separation. Carbohydr Res 307:1–12
Patterson JA, Tetlow IJ, Emes MJ (2018) Bioinformatic and in vitro analyses of Arabidopsis starch synthase 2 reveal post-translational regulatory mechanisms. Front Plant Sci. https://doi.org/10.3389/fpls.2018.01338
Ring SG, Gee JM, Whittam M, Orford P, Johnson IT (1988) Resistant starch: its chemical form in foodstuffs and effect on digestibility in vitro. Food Chem 28:97–109
Sawada T, Itoh M, Nakamura Y (2018) Contributions of three starch branching enzyme isozymes to the fine structure of amylopectin in rice endosperm. Front Plant Sci. https://doi.org/10.3389/fpls.2018.01536
Shure M, Wessler S, Fedoroff N (1983) Molecular-identification and isolation of the waxy locus in maize. Cell 35:225–233
Su P-H, Li H-M (2010) Stromal Hsp70 is important for protein translocation into pea and Arabidopsis chloroplasts. Plant Cell 22:1516–1531
Sun Y, Jiao G, Liu Z et al (2017) Generation of high-amylose rice through CRISPR/Cas9-mediated targeted mutagenesis of starch branching enzymes. Front Plant Sci 8:298
Tanaka N, Fujita N, Nishi A, Satoh H, Hosaka Y, Ugaki M, Kawasaki S, Nakamura Y (2004) The structure of starch can be manipulated by changing the expression levels of starch branching enzyme IIb in rice endosperm. Plant Biotech J 2:507–516
Tetlow IJ, Wait R, Lu Z, Akkasaeng R, Bowsher CG, Esposito S, Kosar-Hashemi B, Morell MK, Emes MJ (2004) Protein phosphorylation in amyloplasts regulates starch branching enzyme activity and protein-protein interactions. Plant Cell 16:694–708
Tetlow IJ, Beisel KG, Cameron S, Makhmoudova A, Liu F, Bresolin NS, Wait R, Morell MK, Emes MJ (2008) Analysis of protein complexes in wheat amyloplasts reveals functional interactions among starch biosynthetic enzymes. Plant Physiol 146:1878–1891
Topping DL, Clifton PM (2001) Short-chain fatty acids and human colonic function: roles of resistant starch and nonstarch polysaccharides. Physiol Rev 81:1031–1064
Tsai CY (1974) Function of waxy locus in starch synthesis in maize endosperm. Biochem Genet 11:83–96
Umemoto T, Aoki N (2005) Single-nucleotide polymorphisms in rice starch synthase IIa that alter starch gelatinisation and starch association of the enzyme. Func Plant Biol 32:763–768
Umemoto T, Yano M, Satoh H, Shomura A, Nakamura Y (2002) Mapping of a gene responsible for the difference in amylopectin structure between japonica-type and indica-type rice varieties. Theor Appl Genet 104:1–8
Umemoto T, Aoki N, Lin HX, Nakamura Y, Inouchi N, Sato Y, Yano M, Hirabayashi H, Maruyama S (2004) Natural variation in rice starch synthase IIa affects enzyme and starch properties. Func Plant Biol 31:671–684
Umemoto T, Horibata T, Aoki N, Hiratsuka M, Yano M, Inouchi N (2008) Effects of variations in starch synthase on starch properties and eating quality of rice. Plant Prod Sci 11:472–480
Wei CX, Qin FL, Zhu LJ, Zhou WD, Chen YF, Wang YP, Gu MH, Liu QQ (2010) Microstructure and ultrastructure of high-amylose rice resistant starch granules modified by antisense RNA inhibition of starch branching enzyme. J Agri Food Chem 58:1224–1232
Yamamori M, Fujita S, Hayakawa K, Matsuki J, Yasui T (2000) Genetic elimination of a starch granule protein, SGP-1, of wheat generates an altered starch with apparent high amylose. Theor Appl Genet 101:21–29
Yu Y, Mu H-H, Mu-Forster C, Wasserman BP (1998) Polypeptides of the maize amyloplast stroma: stromal localization of starch-biosynthetic enzymes and identification of an 81-kilodalton amyloplast stromal heat-shock cognate. Plant Physiol 116:1451–1460
Acknowledgements
Authors would like to thank Oscar Larroque for SEC and CE analysis and data collection; Hong Wang for amylose content and total starch content assay.
Funding
Jixun Luo was supported by the Chinese Scholarship Council and Australian National University scholarships. This work was funded by CSIRO Food Future National Research Flagship.
Author information
Authors and Affiliations
Contributions
JL carried out the experiments and wrote the draft; QY finished the SDS-PAGE detection; CK did particle size and DSC analyses; MLC did mass spectrometry analysis; VMB contributed to experimental design; AM, SJ and ZL designed the experiments and revised the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no competing interests.
Additional information
Communicated by Takuji Sasaki.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Luo, J., Butardo, V.M., Yang, Q. et al. The impact of the indica rice SSIIa allele on the apparent high amylose starch from rice grain with downregulated japonica SBEIIb. Theor Appl Genet 133, 2961–2974 (2020). https://doi.org/10.1007/s00122-020-03649-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00122-020-03649-2