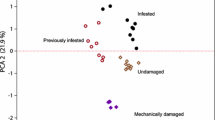
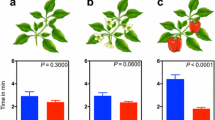
Abstract
The use of nectar-producing companion plants in crops is a well-known strategy of conserving natural enemies in biological control. However, the role of floral volatiles in attracting parasitoids and effects on host location via herbivore-induced plant volatiles is poorly known. Here, we examined the role of floral volatiles from marigold (Tagetes erecta), alone or in combination with volatiles from sweet pepper plant (Capsicum annuum), in recruiting Aphidius platensis, an important parasitoid of the green peach aphid Myzus persicae. We also investigated whether marigold floral volatiles are more attractive to the parasitoid than those emitted by sweet pepper plants infested by M. persicae. Olfactometry assays indicated that floral volatiles attracted A. platensis to the marigold plant and are more attractive than sweet pepper plant volatiles. However, volatiles emitted by aphid-infested sweet pepper were as attractive to the parasitoid as those of uninfested or aphid-infested blooming marigold. The composition of volatile blends released by uninfested and aphid-infested plants differed between both blooming marigold and sweet pepper, but the parasitoid did not discriminate aphid-infested from uninfested blooming marigold. Volatile released from blooming marigold and sweet pepper shared several compounds, but that of blooming marigold contained larger amounts of fatty-acid derivatives and a different composition of terpenes. We discuss the potential implications of the aphid parasitoid attraction in a diversified crop management strategy.





Similar content being viewed by others
Data availability
Not applicable.
Code availability
Not applicable.
References
Albittar L, Ismail M, Bragard C, Hance T (2016) Host plants and aphid hosts influence the selection behaviour of three aphid parasitoids (Hymenoptera: Braconidae: Aphidiinae). Eur J Entomol 113:516–522. https://doi.org/10.14411/eje.2016.068
Alhmedi A, Haubruge E, Francis F (2010) Identification of limonene as a potential kairomone of the harlequin ladybird Harmonia axyridis (Coleoptera: Coccinellidae). Eur J Entomol 107:541–548
Aparicio Y, Gabarra R, Arnó J (2018) Attraction of Aphidius ervi (Hymenoptera: Braconidae) and Aphidoletes aphidimyza (Diptera: Cecidomyiidae) to sweet alyssum and assessment of plant resources effects on their fitness. J Econ Entomol 111(2):533–541. https://doi.org/10.1093/jee/tox365
Araj SE, Wratten SD (2015) Comparing existing weeds and commonly used insectary plants as floral resources for a parasitoid. Biol Control 81:15–20. https://doi.org/10.1016/j.biocontrol.2014.11.003
Baggen LR, Gurr GM, Meats A (1999) Flowers in tri-trophic systems: mechanisms allowing selective exploitation by insect natural enemies for conservation biological control. Entomol Exp Appl 91(1):155–161. https://doi.org/10.1046/j.1570-7458.1999.00478.x
Benelli G, Giunti G, Messing RH et al (2013) Visual and olfactory female-borne cues evoke male courtship in the aphid parasitoid Aphidius colemani Viereck (Hymenoptera: Braconidae). J Insect Behav 26(5):695–707. https://doi.org/10.1007/s10905-013-9386-4
Ben-Issa R, Gomez L, Gautier H (2017) Companion plants for aphid pest management. Insects 8(4):112. https://doi.org/10.3390/insects8040112
Bruijn JAC, Vosteen I, Vet LEM et al (2021) Multi-camera field monitoring reveals costs of learning for parasitoid foraging behavior. J Anim Ecol 90(7):1635–1646. https://doi.org/10.1111/1365-2656.13479
Chen Y, Mao J, Reynolds O et al (2020) Alyssum (Lobularia maritima) selectively attracts and enhances the performance of Cotesia vestalis, a parasitoid of Plutella xylostella. Sci Rep 10(1):1–9. https://doi.org/10.1038/s41598-020-62021-y
Colley MR, Luna JM (2000) Relative attractiveness of potential beneficial insectary plants to aphidophagous hoverflies (Diptera: Syrphidae). Environ Entomol 29(5):1054–1059
Desurmont GA, Laplanche D, Schiestl FP et al (2015) Floral volatiles interfere with plant attraction of parasitoids: ontogeny-dependent infochemical dynamics in Brassica rapa. BMC Ecol 15:1–11. https://doi.org/10.1186/s12898-015-0047-7
Desurmont GA, von Arx M, Turlings TCJ et al (2020) Floral odors can interfere with the foraging behavior of parasitoids searching for hosts. Front Ecol Evol 8(148):1–13. https://doi.org/10.3389/fevo.2020.00148
Du Y, Poppy GM, Powell W et al (1998) Identification of semiochemicals released during aphid feeding that attract the parasitoid Aphidius ervi. J Chem Ecol 24(8):1355–1368. https://doi.org/10.1023/A:1021278816970
Dudareva N, Klempien A, Muhlemann JK et al (2013) Biosynthesis, function and metabolic engineering of plant volatile organic compounds. New Phytol 198(1):16–32. https://doi.org/10.1111/nph.12145
Eben A, Benrey B, Sivinski J et al (2000) Host species and host plant effects on preference and performance of Diachasmimorpha longicaudata (Hymenoptera: Braconidae). Environ Entomol 29(1):87–94. https://doi.org/10.1603/0046-225X-29.1.87
Foti MC, Rostás M, Peri E et al (2017) Chemical ecology meets conservation biological control: identifying plant volatiles as predictors of floral resource suitability for an egg parasitoid of stink bugs. J Pest Sci 90(1):299–310. https://doi.org/10.1007/s10340-016-0758-3
Friberg M, Schwind C, Guimarães PR et al (2019) Extreme diversification of floral volatiles within and among species of Lithophragma (Saxifragaceae). Proc Natl Acad Sci 116(10):4406–4415. https://doi.org/10.1073/pnas.1809007116
Géneau CE, Wäckers FL, Luka H, Balmer O (2013) Effects of extrafloral and floral nectar of Centaurea cyanus on the parasitoid wasp Microplitis mediator: olfactory attractiveness and parasitization rates. Biol Control 66(1):16–20. https://doi.org/10.1016/j.biocontrol.2013.02.007
Gillespie MAK, Gurr GM, Wratten SD (2016) Beyond nectar provision: the other resource requirements of parasitoid biological control agents. Entomol Exp Appl 159(2):207–221. https://doi.org/10.1111/eea.12424
Gouinguené S, Pickett JA, Wadhams LJ et al (2005) Antennal electrophysiological responses of three parasitic wasps to caterpillar-induced volatiles from maize (Zea mays mays), cotton (Gossypium herbaceum), and cowpea (Vigna unguiculata). J Chem Ecol 31(5):1023–1038. https://doi.org/10.1007/s10886-005-4245-1
Hanson PE, Gauld ID (2006) Hymenoptera de la región Neotropical. American Entomological Institute. In: Shaw SR (ed) Familia Braconidae. The American Entomological Institute, Gainesville, pp 487–525
Haro MM, Silveira LCP, Wilby A (2018) Stability lies in flowers: plant diversification mediating shifts in arthropod food webs. PLoS ONE 13(2):e0193045. https://doi.org/10.1371/journal.pone.0193045
Herz A, Cahenzli F, Penvern S et al (2019) Managing floral resources in apple orchards for pest control: ideas, experiences and future directions. Insects 10(8):1–24. https://doi.org/10.3390/insects10080247
Hulcr J, Ubik K, Vrkoc J (2006) The role of semiochemicals in tritrophic interactions between the spruce bark beetle Ips typographus, its predators and infested spruce. J Appl Entomol 130(5):275–283. https://doi.org/10.1111/j.1439-0418.2006.01069.x
Jervis M, Hawkins B, Kidd NAC (1996) The usefulness of destructive host feeding parasitoids in classical biological control: theory and observation conflict. Ecol Entomol 21(1):41–46. https://doi.org/10.1111/j.1365-2311.1996.tb00264.x
Johanowicz DL, Mitchell ER (2000) Effects of sweet alyssum flowers on the longevity of the parasitoid wasps Cotesia marginiventris (Hymenoptera: Braconidae) and Diadegma insulare (Hymenoptera: Ichneumonidae). Fla Entomol 83:41–47
Jaworski CC, Xiao D, Xu Q et al (2019) Varying the spatial arrangement of synthetic herbivore-induced plant volatiles and companion plants to improve conservation biological control. J Appl Ecol 56(5):1176–1188. https://doi.org/10.1111/1365-2664.13353
Knudsen JT, Eriksson R, Gershenzon J et al (2006) Diversity and distribution of floral scent. Bot Rev 72:1–120. https://doi.org/10.1663/0006-8101(2006)72[1:DADOFS]2.0.CO;2
Kugimiya S, Shimoda T, Tabata J et al (2010) Present or past herbivory: a screening of volatiles released from Brassica rapa under caterpillar attacks as attractants for the solitary parasitoid, Cotesia vestalis. J Chem Ecol 36:620–628. https://doi.org/10.1007/s10886-010-9802-6
Landis DA, Wratten SD, Gurr GM (2000) Habitat management to conserve natural enemies of arthropod pests in agriculture. Annu Rev Entomol 45:175–201. https://doi.org/10.1146/annurev.ento.45.1.175
La Rossa FR, Vasicek A, López MC (2013) Effects of pepper (Capsicum annuum) cultivars on the biology and life table parameters of Myzus persicae (Sulz.) (Hemiptera: Aphididae). Neotrop Entomol 42:634–641. https://doi.org/10.1007/s13744-013-0166-9
Lee JC, Andow DA, Heimpel GE (2006) Influence of floral resources on sugar feeding and nutrient dynamics of a parasitoid in the field. Ecol Entomol 31(5):470–480. https://doi.org/10.1111/j.1365-2311.2006.00800.x
Marques MM, Morais SM, Vieira ÍG et al (2011) Larvicidal activity of Tagetes erecta against Aedes aegypti. J Am Mosq Contr Assoc 27(2):156–158. https://doi.org/10.2987/10-6056.1
Mauck KE, De Moraes CM, Mescher MC (2010) Deceptive chemical signals induced by a plant virus attract insect vectors to inferior hosts. Proc Natl Acad Sci U S A 107:3600–3605. https://doi.org/10.1073/pnas.0907191107
Mohammed K, Agarwal M, Du XB et al (2019) Behavioural responses of the parasitoid Aphytis melinus to volatiles organic compounds (VOCs) from Aonidiella aurantii on its host fruit Tahitian lime fruit Citrus latifolia. Biol Control 133:103–109. https://doi.org/10.1016/j.biocontrol.2019.03.015
Ngi-Song AJ, Overholt WA, Njagi PG et al (1996) Volatile infochemicals used in host and host habitat location by Cotesia flavipes Cameron and Cotesia sesamiae (Cameron) (Hymenoptera: Braconidae), larval parasitoids of stemborers on graminae. J Chem Ecol 22(2):307–323. https://doi.org/10.1007/BF02055101
Nilsson U, Eriksson A, Rämert B et al (2012) Male and female Trybliographa rapae (Hymenoptera: Figitidae) behavioural responses to food plant, infested host plant and combined volatiles. Arthropod-Plant Inte 6(2):251–258. https://doi.org/10.1007/s11829-011-9176-x
Peñaflor MFGV, Mauck KE, Alves KJ et al (2016) Effects of single and mixed infections of Bean pod mottle virus and Soybean mosaic virus on host-plant chemistry and host–vector interactions. Funct Ecol 30(10):1648–1659. https://doi.org/10.1111/1365-2435.12649
Pérez-Hedo M, Urbaneja-Bernat P, Jaques JA et al (2015) Defensive plant responses induced by Nesidiocoris tenuis (Hemiptera: Miridae) on tomato plants. J Pest Sci 88:543–554. https://doi.org/10.1007/s10340-014-0640-0
Perović DJ, Gurr GM, Raman A et al (2010) Effect of landscape composition and arrangement on biological control agents in a simplified agricultural system: a cost–distance approach. Biol Control 52(3):263–270. https://doi.org/10.1016/j.biocontrol.2009.09.014
Quinn NF, Brainard DC, Szendrei Z (2017) Floral strips attract beneficial insects but do not enhance yield in cucumber fields. J Econ Entomol 110(2):517–524. https://doi.org/10.1093/jee/tow306
Raguso RA (2008) Wake up and smell the roses: the ecology and evolution of floral scent. Annu Rev Ecol Evol Syst 39:549–569. https://doi.org/10.1146/annurev.ecolsys.38.091206.095601
Ribeiro AC, Guimarães PTG, Alvarez VVH (eds) (1999) Recomendação para o uso de corretivos e fertilizantes em Minas Gerais: 5a aproximação. Comissão de fertilidade de solo do estado de Minas Gerais/departamento de solos, Universidade Federal de Viçosa, Viçosa, Minas Gerais
Rodriguez-Saona C, Kaplan I, Braasch J et al (2011) Field responses of predaceous arthropods to methyl salicylate: a meta-analysis and case study in cranberries. Biol Control 59:294–303. https://doi.org/10.1016/j.biocontrol.2011.06.017
Rudgers JA, Gardener MC (2004) Extrafloral nectar as a resource mediating multispecies interactions. Ecology 85(6):1495–1502. https://doi.org/10.1890/03-0391
Russell M (2015) A meta-analysis of physiological and behavioral responses of parasitoid wasps to flowers of individual plant species. Biol Control 82:96–103. https://doi.org/10.1016/j.biocontrol.2014.11.014
Salamanca J, Souza B, Lundgren JG et al (2017) From laboratory to field: electro-antennographic and behavioral responsiveness of two insect predators to methyl salicylate. Chemoecology 27:51–63. https://doi.org/10.1007/s00049-017-0230-8
Sánchez DOS, Llanos LA, Estrada MEV et al (2012) Insecticidal activity of Tagetes erecta extracts on Spodoptera frugiperda (Lepidoptera: Noctuidae). Fla Entomol 95:428–432. https://doi.org/10.1653/024.095.0225
Sasso R, Iodice L, Digilio MC et al (2007) Host-locating response by the aphid parasitoid Aphidius ervi to tomato plant volatiles. J Plant Interact 2:175–183. https://doi.org/10.1080/17429140701591951
Shaw MR (2006) Habitat considerations for parasitic wasps (Hymenoptera). J Insect Conserv 10(2):117–127. https://doi.org/10.1007/s10841-006-6288-1
Shiojiri K, Ozawa R, Kugimiya S et al (2010) Herbivore-specific, density-dependent induction of plant volatiles: honest or “cry wolf” signals? PLoS ONE 5(8):e12161. https://doi.org/10.1371/journal.pone.0012161
Sidney LA, Bueno VH, Lins JC Jr et al (2010) Quality of different aphid species as hosts for the parasitoid Aphidius ervi Haliday (Hymenoptera: Braconidae: Aphidiinae). Neotrop Entomol 39(5):709–713. https://doi.org/10.1590/S1519-566X2010000500006
Silva DB, Bueno VHP, Van Loon JJA et al (2018) Attraction of three mirid predators to tomato infested by both the tomato leaf mining moth Tuta absoluta and the whitefly Bemisia tabaci. J Chem Ecol 44(1):29–39. https://doi.org/10.1007/s10886-017-0909-x
Silveira LCP, Berti Filho E, Pierre LSR et al (2009) Marigold (Tagetes erecta L.) as an attractive crop to natural enemies in onion fields. Sci Agric 66(6):780–787. https://doi.org/10.1590/S0103-90162009000600009
Souza IL, Marucci RC, Silveira LCP et al (2018a) Effects of marigold on the behavior, survival and nutrient reserves of Aphidius platensis. Biocontrol 63:543–553. https://doi.org/10.1007/s10526-018-9882-8
Souza IL, Tomazella VB, Santos AJN et al (2018b) Parasitoids diversity in organic sweet pepper (Capsicum annuum) associated with basil (Ocimum basilicum) and marigold (Tagetes erecta). Braz J Biol 79(4):603–611. https://doi.org/10.1590/1519-6984.185417
Tomanović Z, Petrović A, Mitrović M et al (2014) Molecular and morphological variability within the Aphidius colemani group with redescription of Aphidius platensis Brethes (Hymenoptera: Braconidae: Aphidiinae). Bull Entomol Res 104:552–565. https://doi.org/10.1017/S0007485314000327
Tooker JF, Hanks LM (2000) Flowering plant hosts of adult Hymenopteran parasitoids of central Illinois. Ann Entomol Soc Am 93:580–588. https://doi.org/10.1603/0013-8746(2000)093[0580:fphoah]2.0.co;2
Turlings TCJ, Tumlinson JH, Lewis WJ (1990) Exploitation of herbivore-induced plant odors by host-seeking parasitic wasps. Science 250(4985):1251–1253. https://doi.org/10.1126/science.250.4985.1251
van Lenteren JC (2000) A greenhouse without pesticides: fact or fantasy? Crop Prot 19(6):375–384. https://doi.org/10.1016/S0261-2194(00)00038-7
Wäckers FL (1994) The effect of food deprivation on the innate visual and olfactory preferences in Cotesia rubecula. J Insect Physiol 40(8):641–649. https://doi.org/10.1016/0022-1910(94)90091-4
Wäckers FL (2004) Assessing the suitability of flowering herbs as parasitoid food sources: flower attractiveness and nectar accessibility. Biol Control 29(3):307–314. https://doi.org/10.1016/j.biocontrol.2003.08.005
Wäckers FL, van Rijn PCJ, Heimpel GE (2008) Exploiting honeydew as a food source: making the best of a bad meal? Biol Control 45:176–185. https://doi.org/10.1016/j.biocontrol.2008.01.007
Wäckers FL, van Rijn PC (2012) Pick and mix: selecting flowering plants to meet the requirements of target biological control insects. In: Gurr GM, Wratten SD et al (eds) Biodiversity and insect pests: key issues for sustainable management, 1st edn. Wiley-Blackwell, New York, pp 139–165
Winkler K, Wäckers FL, Kaufman LV et al (2009) Nectar exploitation by herbivores and their parasitoids is a function of flower species and relative humidity. Biol Control 50(3):299–306. https://doi.org/10.1016/j.biocontrol.2009.04.009
Xiu C, Dai W, Pan H et al (2019) Herbivore-induced plant volatiles enhance field-level parasitism of the mirid bug Apolygus lucorum. Biol Control 135:41–47. https://doi.org/10.1016/j.biocontrol.2019.05.004
Yu H, Zhang Y, Wu K et al (2008) Field-testing of synthetic herbivore-induced plant volatiles as attractants for beneficial insects. Environ Entomol 37:1410–1415. https://doi.org/10.1603/0046-225X-37.6.1410
Acknowledgements
We thank the Coordination for the Improvement of Higher Education Personnel (Coordenação de Aperfeiçoamento de Pessoal de Nível Superior, CAPES) for granting a doctoral scholarship to the first author, the Department of Entomology of the Federal University of Lavras (Universidade Federal de Lavras, UFLA) for facilitating the completion of the study, and Arodí Prado Favaris and Rafaela Cristina dos Santos of the Laboratory of Chemical Ecology and Insect Behavior at ESALQ for collecting and identifying the volatiles. The research leading to these results was partially funded by the National Institute of Science and Technology (INCT) Semiochemicals in Agriculture (Fundação de Amparo à Pesquisa do Estado de São Paulo FAPESP and Conselho Nacional de Desenvolvimento Científico e Tecnológico CNPq, grant numbers 2014/50871-0 and 465511/2014-7, respectively), CAPES (process 1416746) and by the FAPESP (process 2016/12771-0).
Funding
The authors thank the INCT Semiochemicals in Agriculture (FAPESP and CNPq, grant numbers 2014/50871–0 and 465511/2014–7, respectively). The research leading to these results was partially funded by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior – CAPES (process 1416746) and FAPESP (process 2016/12771–0).
Author information
Authors and Affiliations
Contributions
The authors contributed to the study conception and design. Material preparation, data collection, analysis, and all writing were performed by Diego Bastos Silva and Ivana Lemos Souza. The first draft of the manuscript was written by Ivana Lemos Souza, Luís Cláudio Paterno, Maria Fernanda Gomes Villalba Peñaflor, and Rosangela Cristina Marucci. The work was carried out with resources from the laboratories of Maria Fernanda Gomes Villalba Peñaflor and José Maurício Simões Bento. All authors commented on previous versions of the manuscript. And all authors read, reviewed, and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Communicated by: John A. Byers
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Souza, I.L., Silva, D.B., Silveira, L.C.P. et al. A parasitoid’s dilemma between food and host resources: the role of volatiles from nectar-providing marigolds and host-infested plants attracting Aphidius platensis. Sci Nat 109, 9 (2022). https://doi.org/10.1007/s00114-021-01780-8
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00114-021-01780-8