Abstract
Numerous risk factors for atrial fibrillation (AF) progression have been identified. However, the biomarkers mentioned in the guidelines do not have any clinically relevant predictive value. Some research groups investigated the potential utility of galectin-3 (gal-3) as a diagnostic, prognostic, and predictive biomarker in AF. In this review, we have thoroughly summarized the current data on the role of gal-3 in AF based on the original research in this field. Patients suffering from AF present with increased levels of gal-3. The concentration of gal-3 differs between patients with AF depending on the type of AF — it is higher in patients with persistent AF than in patients with paroxysmal AF. Multiple studies investigating the reappearance of AF in patients who underwent ablation have shown that gal-3 is a promising biomarker to predict the outcome of this therapy. Patients with increased levels of gal-3 are at higher risk of AF recurrence. Although the research considered in this work addressed many aspects of the role of gal-3 in AF, most of it has been conducted on a small group of patients. Therefore, further research and extensive clinical trials confirming described findings are highly warranted.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
According to the European Society of Cardiology (ESC) definition, atrial fibrillation (AF) is a supraventricular tachyarrhythmia with uncoordinated atrial electrical activation and consequently ineffective atrial contraction [1]. More than 60% of patients suffering from AF have a decreased quality of life. AF is a significant risk factor for ischaemic stroke and heart failure, increasing mortality [2]. Currently, the estimated prevalence of AF in adults is between 2 and 4%, being the most frequent cardiac arrhythmia nowadays [1, 3]. AF incidence and prevalence have increased over the last 20 years and will likely continue rising over the next 30 years [3].
AF progression from paroxysmal to non-paroxysmal is associated with multiple risk factors, including age, and many diseases, i.e., heart failure, hypertension, chronic kidney disease, chronic pulmonary diseases, diabetes mellitus, and previous stroke [1, 4]. However, the biomarkers mentioned in the guidelines do not have clinically relevant predictive value [1]. Because of that, further research on the role of biomarkers in AF is highly warranted [5, 6]. Knowledge about the pathogenesis of the disease should always be the rationale for the search for potential biomarkers. The ongoing fibrosis in the left atrium is one of the leading causes of AF since it delays electromechanical conduction and creates a substrate for AF [7]. Multiple proteins involved in fibrosis have been investigated so far. Recently, much scientific attention has been drawn to galectin-3 (gal-3).
Gal-3 is a β-galactoside-binding protein belonging to the lectin family. It plays a vital role in many physiological cellular functions, including cellular growth, differentiation, proliferation, apoptosis, cellular adhesion, and tissue repair [8]. The concentration of gal-3 levels can be measured with the use of immunoassays. Enzyme-linked immunosorbent assay (ELISA) is the most commonly used technique. Nevertheless, other variants of immunoassays can also be used, including enzyme-linked fluorescent assay (ELFA) or chemiluminescent microparticle immunoassay (CMIA). Although there are some differences in the specificity and sensitivity of these tests, price, and availability seem to be decisive factors for test choice, favoring ELISA.
How gal-3 is implicated in the pathogenesis of various cardiovascular diseases has been extensively explored, mainly referring to its involvement in inflammation and tissue fibrosis processes [8, 9]. Much research has focused on its role in AF.
Some research groups investigated the potential utility of gal-3 as a diagnostic and disease severity/prognostic biomarker in AF. In contrast, others studied the correlation between gal-3 concentrations and the possibility of AF recurrence after different interventions, such as ablation [10, 11]. The understanding of the role of gal-3 in the induction and the progression of AF may lead to better management of patients as well as may provide new targets for treatment.
In this review, we have thoroughly summarized the current data on the role of gal-3 in AF based on the original research in this field. We aim to contribute to setting objectives for future research on gal-3 and its use as a biomarker in daily clinical practice.
Galectin-3 as a potential biomarker in atrial fibrillation
Numerous clinical studies investigating the role of gal-3 in AF have already been conducted. In our review, we included only original clinical research. Reviews, letters to the editors, and commentaries were not included. We searched PubMed Database by the query: “(galectin-3) AND (atrial fibrillation)” which yielded a total of 117 records. Excluding unsuitable titles, article types, or abstracts, we retrieved in complete form and assessed 51 studies. We evaluated the complete data reports for eligibility and excluded 17 studies irrelevant to the field. We eventually included 34 original clinical research relevant to the discussed area (Fig. 1).
We divided these studies into the following parts: (i) differences in the levels of gal-3 between patients with AF and patients in sinus rhythm, (ii) differences in the levels of gal-3 between patients with different types of AF, (iii) gal-3 in patients with AF undergoing cardioversion, (iv) gal-3 as a predictor of AF recurrence after ablation and (v) gal-3 in patients with AF undergoing surgeries and other invasive procedures (Fig. 2).
Differences in the levels of galectin-3 between patients with atrial fibrillation and patients in sinus rhythm
Ho et al. studied the relationship between gal-3 levels and the probability of AF occurrence in participants of the Framingham Offspring cohort. They discovered that higher concentrations of circulating gal-3 were associated with an increased risk of developing AF [12]. Another research group compared the levels of gal-3 between patients with non-valvular AF and age-matched patients in sinus rhythm. It was shown that patients with AF had significantly higher concentrations of this biomarker than the control group. Moreover, the authors found a positive correlation between the concentration of gal-3 in blood and the left atrial volume index [13]. Consistently, Pauklin et al. analyzed the levels of gal-3 in patients suffering from paroxysmal or persistent AF admitted for electrical cardioversion or pulmonary vein isolation (PVI) and compared them with healthy controls. The study revealed that the levels of gal-3 were higher in patients with AF [14].
Similarly, Selcoki et al. analyzed gal-3 levels in patients with paroxysmal AF and preserved left ventricular systolic function and compared them with healthy controls matched by age and gender. It was shown that patients with AF had higher levels of gal-3 than controls. Moreover, the authors found a positive correlation between left atrial diameter and serum gal-3 levels [15].
Contrary results to these studies were found by Begg et al. They considered gal-3 level a marker of fibrosis in patients who underwent ablation. Therefore, they analyzed patients suffering from AF (paroxysmal or non-paroxysmal) who underwent ablation and compared them to non-AF control patients. They proved no significant difference in gal-3 levels between these groups [16]. However, the authors found that peripheral gal-3 levels were significantly higher than left atrial gal-3 levels.
Fashanu et al. studied the relationship between gal-3 levels and the probability of an AF incident. It was shown that patients with gal-3 levels ≥ 90th percentile had a higher risk of an AF incident than those with gal-3 levels < 90th percentile [17].
In another study, Shen et al. compared two groups of patients who underwent single mitral valve repair/replacement. The first group consisted of patients suffering from persistent AF, while the second group had sinus rhythm patients. It was proven that the levels of gal-3 measured in serum collected from coronary sinus blood and gal-3 measured in atrial tissues were higher in the first group than in the second one [18].
Hernández-Romero et al. analyzed gal-3 levels in patients with permanent AF who underwent cardiac surgery and compared them to those with aortic valve or ischaemic heart diseases who also underwent cardiac surgery. They observed differences in serum concentrations of gal-3 between the groups– gal-3 levels were higher in patients with permanent AF. Moreover, high levels of gal-3 were found to be an independent predictor of fibrosis [19]. All studies discussed in this section with additional information have been presented in Table 1.
Differences in the levels of galectin-3 between patients with different types of atrial fibrillation
Gurses et al. analyzed gal-3 levels in persistent or paroxysmal AF patients with preserved left ventricular systolic function and compared them to the age-and-gender-matched controls. It was proved that gal-3 levels and left atrial volume index were higher in AF patients than in the controls. Moreover, gal-3 levels were significantly higher in patients with persistent AF than in those with paroxysmal AF [20]. Accordingly, Chen et al. compared gal-3 levels between new-onset AF patients and patients with chronic AF. The study revealed that patients with new-onset AF had higher gal-3 levels than those with pre-existing, chronic AF [21].
Tang et al. compared patients with all three types of AF: paroxysmal, persistent, and permanent. It was shown that patients with persistent or permanent AF had higher levels of gal-3 compared to patients with paroxysmal AF. Moreover, gal-3 was an independent determinant of LAA thrombus in AF patients [22].
Another interesting research was held by Arbault-Biton et al. They divided the group of AF patients based on the AF duration: there were 47 patients with AF ≤ 48 h and 51 patients with AF > 48 h. It was proved that gal-3 concentrations did not differ between the groups [23].
Interestingly, Zaslavskaya et al. compared gal-3 levels in patients suffering from metabolic syndrome (MS) with coexisting paroxysmal/persistent AF or without arrhythmia to healthy controls. They showed that gal-3 levels were higher in patients with coexisting MS and AF than in patients with only MS and healthy controls. Moreover, gal-3 levels were higher in patients with persistent AF than in those with paroxysmal AF [24].
Wang et al. studied patients suffering from paroxysmal AF, dividing them based on the progression to persistent AF. It was demonstrated that patients with AF progression to a persistent state had higher gal-3 levels than those without AF progression. Moreover, gal-3 concentration was associated with AF progression [25]. All studies discussed in this section with additional information have been presented in Table 2.
Galectin-3 in patients with atrial fibrillation undergoing cardioversion
Begg et al. examined gal-3 levels as a potential prognostic factor of AF recurrence after direct current cardioversion (DCCV). They included patients who underwent DCCV for AF and compared them to healthy controls. They demonstrated that there was no difference in gal-3 levels between these groups. Moreover, gal-3 levels did not predict AF recurrence after DCCV [26]. Consistently, Kisheva et al. examined gal-3 levels as a predictive factor of AF recurrence in patients suffering from AF after sinus rhythm restoration. It was shown that gal-3 levels did not affect the number of AF recurrences after sinus rhythm restoration [27].
A similar study was conducted by Gürses et al. They evaluated the group of 90 patients suffering from persistent AF who underwent DCCV. As presented, 28 patients experienced early AF recurrence within 3 months. Contrary to the studies above, this research group showed that the patients with AF recurrence had higher gal-3 levels compared to those who did not experience AF recurrence after the procedure [28].
Wałek et al. examined correlations between the levels of gal-3 and the echocardiographic parameters of the left atrium and left ventricle in patients suffering from persistent AF with left atrial enlargement qualified for DCCV. The authors observed negative correlations between the concentrations of gal-3 and left atrial: dimensions, volume, contractility, and compliance. Moreover, negative correlations were found between the levels of gal-3 and left ventricular: volume and contractility [29]. All studies discussed in this section with additional information have been presented in Table 3.
Galectin-3 as a predictor of atrial fibrillation recurrence after ablation
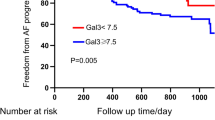
Wu et al. analyzed plasma concentrations of gal-3 in patients with persistent AF without coexisting structural heart disease who underwent first-time catheter ablation and compared them to healthy controls. The levels of gal-3 were higher in patients suffering from persistent AF. Furthermore, the concentrations of gal-3 were increased in patients with recurrence of AF after ablation, compared to patients without AF recurrence. Moreover, gal-3 was an independent predictor of AF reappearance after catheter ablation [30]. Similarly, Ruan et al. analyzed the relationship between the preoperative level of gal-3 and AF recurrence in patients undergoing radiofrequency catheter ablation (RFCA). It was shown that patients who experienced the recurrence of AF after the RFCA presented higher levels of gal-3 (measured at baseline) than patients who did not develop AF again after the procedure. It was proved that gal-3 could be considered an independent predictor of AF reappearance after the procedure [31]. Consistently, Lee et al. investigated the correlation between levels of gal-3 and the risk of atrial tachyarrhythmias recurrence in patients who underwent ablation for AF. Increased levels of gal-3 were associated with an increased risk of atrial tachyarrhythmias recurrence [32].
Takemoto et al. compared the levels of gal-3 measured in intracardiac serum obtained from the coronary sinus and left atrium between patients with persistent AF and patients with paroxysmal AF. Higher levels of this biomarker were found in patients with persistent AF. They also showed that gal-3 was an independent predictor of atrial tachyarrhythmia recurrence after a single ablation procedure [33].
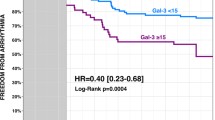
Clementy et al. investigated gal-3 levels and left atrial diameter (LAD) as potential predictive factors of AF recurrence in patients suffering from AF who underwent ablation. Patients who experienced AF recurrence after the procedure appeared to have higher gal-3 levels and larger left atrium. Gal-3 levels and LAD were independent predictors of AF recurrence [34].
Contrary results were obtained by Kornej et al. They conducted a study including AF patients who underwent catheter ablation and compared them to AF-free controls. It was shown that gal-3 levels were higher in AF patients compared to AF-free controls. However, the authors concluded that gal-3 did not help predict the rhythm outcome of the catheter ablation [35]. Another research team had similar findings: Celik et al. analyzed gal-3 levels as a predictive factor of AF recurrence in patients who underwent PVI. It was proven that gal-3 levels were not associated with AF recurrence. Moreover, PVI did not significantly affect serum gal-3 levels [36].
Berger et al. studied the relationship between gal-3 levels and the probability of AF recurrence in patients undergoing thoracoscopic surgery for AF. A higher recurrence rate was observed in patients with increased gal-3 levels after ablation compared to baseline than in patients with unchanged or decreased gal-3 level [37].
Yalcin et al. evaluated patients suffering from paroxysmal AF undergoing cryoballoon-based AF ablation. They studied the relationship between the levels of gal-3 and left atrium fibrosis assessed by delayed-enhancement magnetic resonance imaging (DE-MRI) and atrial electromechanical delay (AEMD). They proved that the concentrations of gal-3 were independently correlated with the extent of left atrium fibrosis detected with DE-MRI. Moreover, the levels of gal-3 correlated with intra- left and inter-AEMD [38].
Aksan et al. investigated the association between gal-3 level and low voltage areas (LVA) severity in patients suffering from paroxysmal AF who underwent PVI. It was proved that gal-3 levels were higher in paroxysmal AF patients with moderate and severe LVA compared to those with mild LVA. Moreover, gal-3 levels were higher in paroxysmal AF patients with LVA than in those without LVA [39]. All studies discussed in this section with additional information have been presented in Table 4.
Galectin-3 in patients with atrial fibrillation undergoing surgeries and other invasive procedures
Szadkowska et al. evaluated patients with first acute myocardial infarction who underwent primary percutaneous coronary intervention with stent implantation. The authors showed that new-onset AF was independently associated with increased levels of gal-3 (particularly > 16 ng/mL) [40]. Similarly, Wang et al. evaluated patients admitted with acute myocardial infarction (AMI). The authors compared patients who developed post-AMI new-onset AF to those who did not. It was evidenced that the first group had increased levels of gal-3. Furthermore, the plasma concentration of gal-3 was found as an independent predictor of post-AMI new-onset AF [41].
Richter et al. hypothesized that gal-3 level could independently predict postoperative atrial fibrillation (POAF). Based on these assumptions, they enrolled patients undergoing elective cardiac surgeries. They proved that the levels of gal-3 were higher in patients who developed POAF compared to those who did not develop it. Moreover, gal-3 levels were an independent predictor of POAF and mortality after cardiac surgery [42]. Erdem et al. obtained similar results, investigating patients undergoing coronary artery bypass graft (CABG) surgery. It was shown that gal-3 could predict POAF possessing high specificity and sensitivity [43].
Aksan et al. investigated gal-3 levels as a marker of AF recurrence expressed as atrial high-rate episodes (AHRE) in patients who underwent cardiac resynchronization therapy. The study revealed that gal-3 levels were higher in patients with AHRE than those without AHRE. Also, there was a positive correlation between gal-3 levels and the percent of time spent in total AHRE [44].
Tan et al. investigated patients suffering from heart failure (HF), dividing them based on the coexistence of AF. It was proved that gal-3 predicted HF-related hospitalization, but only in patients with coexisting AF [45]. All studies discussed in this section with additional information have been presented in Table 5.
Conclusions and future perspectives
Many studies regarding gal-3 as a biomarker of AF have been conducted so far. The main findings from these studies are that (i) there is a correlation between higher levels of gal-3 and activation of the profibrotic pathway, (ii) the concentration of this biomarker differs between patients depending on the type of AF (paroxysmal, persistent, and permanent), (iii) gal-3 is a promising biomarker to predict the recurrence of AF in patients after ablation and POAF after cardiac surgery.
Almost all research groups confirmed higher levels of gal-3 in patients with AF compared to non-AF controls [13,14,15, 18, 19]. Begg et al. obtained contrary results in 2 studies: (i) comparing AF patients undergoing ablation to healthy controls and (ii) comparing AF patients undergoing DCCV to age-and-disease-matched controls [16, 26]. The potential rationale for this observed difference may be found in the characteristics of the included population. Patients undergoing ablation are most commonly relatively young, and their AF cannot be permanent. Similarly, patients undergoing DCCV are often diagnosed quite recently, and thus the fibrosis process is less advanced than it would be after, e.g., 20 years of AF occurrence.
The next conflicting findings regard the predictive value of gal-3 for AF recurrence after DCCV. Most research found no predictive value of this biomarker in this use [26, 27]. Nevertheless, Gürses et al. showed that patients with AF recurrence after DCCV had higher levels of gal-3 [28]. Moreover, in the multivariate analysis, they found that serum gal-3 level was independently associated with early recurrence of AF after a successful DCCV. This discrepancy with other studies requires further investigation and a search for an explanation.
Although the research considered in this manuscript addressed many aspects of the role of gal-3 in AF, most of it was conducted on small groups of patients. Therefore, more extensive research, optimally a randomized clinical trial, would be of great value to standardize and confirm the previous findings, particularly the thesis regarding the role of gal-3 as a predictive biomarker of AF recurrence after ablation (Fig. 3).
Further improvement in our knowledge about the role of gal-3 in AF would lead to a better understanding of the pathogenesis of this disease. Significantly, it could contribute to the earlier diagnosis of AF, even before the physical signs of an irregular heart rhythm. As paroxysmal AF is very difficult to spot during the electrocardiographic examination, establishing a biomarker enabling the diagnosis of AF based on its concentration would help diagnose AF at its earliest stages.
Gal-3 acts on fibroblasts’/myofibroblasts’ migration and proliferation. Macrophages are critical mediators of this process since gal-3 is most expressed in tissue-resident macrophages [46]. This plays a crucial role in activating a pro-fibrotic phenotype of macrophages and the following fibroblasts’/myofibroblasts’ activation [47]. Although much is known, there are still gaps in evidence regarding the exact role of gal-3 in the fibrosis process. Therefore, further research on the effects of gal-3 inhibition in preclinical and clinical settings may contribute to a better understanding of the gal-3 role in fibrosis and AF pathogenesis and a possible introduction of a new targeted therapy in AF. In conclusion, future studies are recommended to fully confirm the role of gal-3 as an AF biomarker and open doors for the new targeted therapy in this disease.
References
Hindricks G, Potpara T, Dagres N, Arbelo E, Bax JJ, Blomström-Lundqvist C, Boriani G, Castella M, Dan GA, Dilaveris PE et al (2021) 2020 ESC Guidelines for the diagnosis and management of atrial fibrillation developed in collaboration with the European Association for Cardio-Thoracic Surgery (EACTS): The Task Force for the diagnosis and management of atrial fibrillation of the European Society of Cardiology (ESC) Developed with the special contribution of the European Heart Rhythm Association (EHRA) of the ESC. Eur Heart J 42:373–498. https://doi.org/10.1093/eurheartj/ehaa612
Migdady I, Russman A, Buletko AB (2021) Atrial fibrillation and ischemic stroke: a clinical review. Semin Neurol 41:348–364. https://doi.org/10.1055/s-0041-1726332
Lippi G, Sanchis-Gomar F, Cervellin G (2021) Global epidemiology of atrial fibrillation: an increasing epidemic and public health challenge. Int J Stroke 16:217–221. https://doi.org/10.1177/1747493019897870
Benjamin EJ, Levy D, Vaziri SM, D’Agostino RB, Belanger AJ, Wolf PA (1994) Independent risk factors for atrial fibrillation in a population-based cohort. The Framingham Heart Study Jama 271:840–844
Procyk G, Bilicki D, Balsam P, Lodziński P, Grabowski M, Gąsecka A (2022) Extracellular vesicles in atrial fibrillation-state of the art. Int J Mol Sci 23. https://doi.org/10.3390/ijms23147591
Zygadło J, Procyk G, Balsam P, Lodziński P, Grabowski M, Gąsecka A (2023) Autoantibodies in atrial fibrillation-state of the art. Int J Mol Sci 24. https://doi.org/10.3390/ijms24031852
Burstein B, Nattel S (2008) Atrial fibrosis: mechanisms and clinical relevance in atrial fibrillation. J Am Coll Cardiol 51:802–809. https://doi.org/10.1016/j.jacc.2007.09.064
Blanda V, Bracale UM, Di Taranto MD, Fortunato G (2020) Galectin-3 in cardiovascular diseases. Int J Mol Sci 21. https://doi.org/10.3390/ijms21239232
Tymińska A, Kapłon-Cieślicka A, Ozierański K, Budnik M, Wancerz A, Sypień P, Peller M, Maksym J, Balsam P, Opolski G et al (2019) Association of galectin-3 and soluble ST2 with in-hospital and 1-year outcomes in patients with ST-segment elevation myocardial infarction treated with primary percutaneous coronary intervention. Pol Arch Intern Med 129:770–780. https://doi.org/10.20452/pamw.15030
Sygitowicz G, Maciejak-Jastrzębska A, Sitkiewicz D (2021) The diagnostic and therapeutic potential of galectin-3 in cardiovascular diseases. Biomolecules 12. https://doi.org/10.3390/biom12010046
Clementy N, Piver E, Bisson A, Andre C, Bernard A, Pierre B, Fauchier L, Babuty D (2018) Galectin-3 in atrial fibrillation: mechanisms and therapeutic implications. Int J Mol Sci 19. https://doi.org/10.3390/ijms19040976
Ho JE, Yin X, Levy D, Vasan RS, Magnani JW, Ellinor PT, McManus DD, Lubitz SA, Larson MG, Benjamin EJ (2014) Galectin 3 and incident atrial fibrillation in the community. Am Heart J 167:729-734.e721. https://doi.org/10.1016/j.ahj.2014.02.009
Sonmez O, Ertem FU, Vatankulu MA, Erdogan E, Tasal A, Kucukbuzcu S, Goktekin O (2014) Novel fibro-inflammation markers in assessing left atrial remodeling in non-valvular atrial fibrillation. Med Sci Monit 20:463–470. https://doi.org/10.12659/msm.890635
Pauklin P, Zilmer M, Eha J, Tootsi K, Kals M, Kampus P (2022) Markers of inflammation, oxidative stress, and fibrosis in patients with atrial fibrillation. Oxid Med Cell Longev 2022:4556671. https://doi.org/10.1155/2022/4556671
Selcoki Y, Aydin H, Celik TH, Isleyen A, Erayman A, Demircelik MB, Demirin H, Kosus A, Eryonucu B (2016) Galectin-3: a biochemical marker to detect paroxysmal atrial fibrillation? Clin Invest Med 39:27528
Begg GA, Karim R, Oesterlein T, Graham LN, Hogarth AJ, Page SP, Pepper CB, Rhode K, Lip GYH, Holden AV et al (2017) Intra-cardiac and peripheral levels of biochemical markers of fibrosis in patients undergoing catheter ablation for atrial fibrillation. Europace 19:1944–1950. https://doi.org/10.1093/europace/euw315
Fashanu OE, Norby FL, Aguilar D, Ballantyne CM, Hoogeveen RC, Chen LY, Soliman EZ, Alonso A, Folsom AR (2017) Galectin-3 and incidence of atrial fibrillation: the atherosclerosis risk in communities (ARIC) study. Am Heart J 192:19–25. https://doi.org/10.1016/j.ahj.2017.07.001
Shen H, Wang J, Min J, Xi W, Gao Y, Yin L, Yu Y, Liu K, Xiao J, Zhang YF et al (2018) Activation of TGF-β1/α-SMA/Col I profibrotic pathway in fibroblasts by galectin-3 contributes to atrial fibrosis in experimental models and patients. Cell Physiol Biochem 47:851–863. https://doi.org/10.1159/000490077
Hernández-Romero D, Vílchez JA, Lahoz Á, Romero-Aniorte AI, Jover E, García-Alberola A, Jara-Rubio R, Martínez CM, Valdés M, Marín F (2017) Galectin-3 as a marker of interstitial atrial remodelling involved in atrial fibrillation. Sci Rep 7:40378. https://doi.org/10.1038/srep40378
Gurses KM, Yalcin MU, Kocyigit D, Canpinar H, Evranos B, Yorgun H, Sahiner ML, Kaya EB, Ozer N, Tokgozoglu L et al (2015) Effects of persistent atrial fibrillation on serum galectin-3 levels. Am J Cardiol 115:647–651. https://doi.org/10.1016/j.amjcard.2014.12.021
Chen D, Procter N, Goh V, Liu S, Chua SJ, Assadi-Khansari B, Stewart S, Horowitz JD, Sverdlov AL, Ngo DT (2016) New onset atrial fibrillation is associated with elevated galectin-3 levels. Int J Cardiol 223:48–49. https://doi.org/10.1016/j.ijcard.2016.08.172
Tang Z, Zeng L, Lin Y, Han Z, Gu J, Wang C, Zhang H (2019) Circulating galectin-3 is associated with left atrial appendage remodelling and thrombus formation in patients with atrial fibrillation. Heart Lung Circ 28:923–931. https://doi.org/10.1016/j.hlc.2018.05.094
Arbault-Biton C, Chenevier-Gobeaux C, Legallois D, Msadek S, Boubaya M, Roule V, Boukertouta T, Goudot FX, Beygui F, Meune C (2021) Multiple biomarkers measurement to estimate the duration of atrial fibrillation. Ann Clin Biochem 58:102–107. https://doi.org/10.1177/0004563220975171
Va A, Zaslavskaya EL, Soboleva AV, Baranova EI, Listopad OV, Nifontov SE, Nrady AO, Shlyakhto EV (2016) Galectin-3 in patients with paroxysmal and persistent atrial fibrillation and metabolic syndrome. Kardiologiia 56(6):41–45. Russian. https://doi.org/10.18565/cardio.2016.6.17-22. PMID: 28290846
Wang Q, Xu L, Dong Y, Fu Y, Pan Y, Luan Q, Liu Y, Liu Z, Yang X, Chen M et al (2021) Plasma Galectin-3 is associated with progression from paroxysmal to persistent atrial fibrillation. BMC Cardiovasc Disord 21:226. https://doi.org/10.1186/s12872-021-02043-0
Begg GA, Lip GY, Plein S, Tayebjee MH (2017) Circulating biomarkers of fibrosis and cardioversion of atrial fibrillation: a prospective, controlled cohort study. Clin Biochem 50:11–15. https://doi.org/10.1016/j.clinbiochem.2016.09.008
Kisheva A, Yotov Y, Chervenkov T, Angelov A, Bocheva Y (2021) Galectin-3 in patients with atrial fibrillation and restored sinus rhythm. Folia Med (Plovdiv) 63:329–336. https://doi.org/10.3897/folmed.63.e55313
Gürses KM, Yalçın MU, Koçyiğit D, Canpınar H, Ateş AH, Canpolat U, Yorgun H, Güç D, Aytemir K (2019) Serum galectin-3 level predicts early recurrence following successful direct-current cardioversion in persistent atrial fibrillation patients. Turk Kardiyol Dern Ars 47:564–571. https://doi.org/10.5543/tkda.2019.58399
Wałek P, Grabowska U, Cieśla E, Sielski J, Roskal-Wałek J, Wożakowska-Kapłon B (2021) Analysis of the correlation of galectin-3 concentration with the measurements of echocardiographic parameters assessing left atrial remodeling and function in patients with persistent atrial fibrillation. Biomolecules 11. https://doi.org/10.3390/biom11081108
Wu XY, Li SN, Wen SN, Nie JG, Deng WN, Bai R, Liu N, Tang RB, Zhang T, Du X et al (2015) Plasma galectin-3 predicts clinical outcomes after catheter ablation in persistent atrial fibrillation patients without structural heart disease. Europace 17:1541–1547. https://doi.org/10.1093/europace/euv045
Ruan ZB, Gao RF, Wang F, Chen GC, Zhu JG, Ren Y, Zhu L (2022) Circulating galectin-3 and aldosterone for predicting atrial fibrillation recurrence after radiofrequency catheter ablation. Cardiovasc Ther 2022:6993904. https://doi.org/10.1155/2022/6993904
Lee KN, Kim DY, Boo KY, Kim YG, Roh SY, Baek YS, Kim DH, Lee DI, Shim J, Choi JI et al (2022) Therapeutic implications of galectin-3 in patients with atrial fibrillation. Sci Rep 12:784. https://doi.org/10.1038/s41598-022-04894-9
Takemoto Y, Ramirez RJ, Yokokawa M, Kaur K, Ponce-Balbuena D, Sinno MC, Willis BC, Ghanbari H, Ennis SR, Guerrero-Serna G et al (2016) Galectin-3 regulates atrial fibrillation remodeling and predicts catheter ablation outcomes. JACC Basic Transl Sci 1:143–154. https://doi.org/10.1016/j.jacbts.2016.03.003
Clementy N, Benhenda N, Piver E, Pierre B, Bernard A, Fauchier L, Pages JC, Babuty D (2016) Serum galectin-3 levels predict recurrences after ablation of atrial fibrillation. Sci Rep 6:34357. https://doi.org/10.1038/srep34357
Kornej J, Schmidl J, Ueberham L, John S, Daneschnejad S, Dinov B, Hindricks G, Adams V, Husser D, Bollmann A (2015) Galectin-3 in patients with atrial fibrillation undergoing radiofrequency catheter ablation. PLoS One 10:e0123574. https://doi.org/10.1371/journal.pone.0123574
Celik AI, Kanadasi M, Demir M, Deniz A, Akilli RE, Deveci OS, Cagliyan CE, Ozmen C, Ikikardes F, Karaaslan MB (2019) Predictors of the paroxysmal atrial fibrillation recurrence following cryoballoon-based pulmonary vein isolation: assessment of left atrial volume, left atrial volume index, galectin-3 level and neutrophil-to-lymphocyte ratio. Indian Pacing Electrophysiol J 19:9–14. https://doi.org/10.1016/j.ipej.2018.07.003
Berger WR, Jagu B, van den Berg NWE, Chan Pin Yin D, van Straalen JP, de Boer OJ, Driessen AHG, Neefs J, Krul SPJ, van Boven WP et al (2018) The change in circulating galectin-3 predicts absence of atrial fibrillation after thoracoscopic surgical ablation. Europace 20:764–771. https://doi.org/10.1093/europace/eux090
Yalcin MU, Gurses KM, Kocyigit D, Canpinar H, Canpolat U, Evranos B, Yorgun H, Sahiner ML, Kaya EB, Hazirolan T et al (2015) The association of serum galectin-3 levels with atrial electrical and structural remodeling. J Cardiovasc Electrophysiol 26:635–640. https://doi.org/10.1111/jce.12637
Aksan G, Yanık A, Yontar OC, Boyacı F, Uçar M, Şahin MK, Soylu K (2022) The predictive value of galectin-3 levels on left atrial low voltage areas assessed by high-density mapping in patients with paroxysmal atrial fibrillation. J Arrhythm 38:353–362. https://doi.org/10.1002/joa3.12703
Szadkowska I, Wlazeł RN, Migała M, Szadkowski K, Zielińska M, Paradowski M, Pawlicki L (2013) The association between galectin-3 and clinical parameters in patients with first acute myocardial infarction treated with primary percutaneous coronary angioplasty. Cardiol J 20:577–582. https://doi.org/10.5603/cj.2013.0157
Wang Q, Huai W, Ye X, Pan Y, Yang X, Chen M, Ma QB, Gao Y, Zhang Y (2022) Circulating plasma galectin-3 predicts new-onset atrial fibrillation in patients after acute myocardial infarction during hospitalization. BMC Cardiovasc Disord 22:392. https://doi.org/10.1186/s12872-022-02827-y
Richter B, Koller L, Hofer F, Kazem N, Hammer A, Silbert BI, Laufer G, Steinlechner B, Wojta J, Hengstenberg C et al (2022) Galectin-3 is an independent predictor of postoperative atrial fibrillation and survival after elective cardiac surgery. Heart Rhythm. https://doi.org/10.1016/j.hrthm.2022.06.019
Erdem K, Kurtoglu E, Oc M, Oc B, Ilgenli TF, Unlu A, Eryavuz Onmaz D (2022) The plasma galectin-3 level has high specificity and sensitivity for predicting postoperative atrial fibrillation after coronary artery bypass surgery. Eur Rev Med Pharmacol Sci 26:9072–9078. https://doi.org/10.26355/eurrev_202212_30656
Aksan G, Yanık A, Yontar OC, Gedikli Ö, Arslan U, Soylu K (2021) Galectin-3 levels and the prediction of atrial high-rate episodes in patients with cardiac resynchronization therapy. J Investig Med 69:20–27. https://doi.org/10.1136/jim-2020-001459
Tan ESJ, Chan SP, Liew OW, Chong JPC, Leong GKT, Yeo DPS, Ong HY, Jaufeerally F, Yap J, Sim D et al (2021) Atrial fibrillation and the prognostic performance of biomarkers in heart failure. Clin Chem 67:216–226. https://doi.org/10.1093/clinchem/hvaa287
Liu FT, Hsu DK, Zuberi RI, Kuwabara I, Chi EY, Henderson WR Jr (1995) Expression and function of galectin-3, a beta-galactoside-binding lectin, in human monocytes and macrophages. Am J Pathol 147:1016–1028
Duffield JS, Forbes SJ, Constandinou CM, Clay S, Partolina M, Vuthoori S, Wu S, Lang R, Iredale JP (2005) Selective depletion of macrophages reveals distinct, opposing roles during liver injury and repair. J Clin Invest 115:56–65. https://doi.org/10.1172/jci22675
Acknowledgements
The figures were created with BioRender.com, a licensed version by Aleksandra Gąsecka.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study’s conception and design. The idea for the article, the literature search, and the draft preparation were performed by Grzegorz Procyk, Aleksandra Czapla, and Kamila Jałocha. The work was critically revised by Agata Tymińska, Marcin Grabowski, and Aleksandra Gąsecka.
Corresponding author
Ethics declarations
Ethics approval
The manuscript does not contain clinical studies or patient data.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Procyk, G., Czapla, A., Jałocha, K. et al. The role of galectin-3 in atrial fibrillation. J Mol Med 101, 1481–1492 (2023). https://doi.org/10.1007/s00109-023-02378-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00109-023-02378-5