Abstract
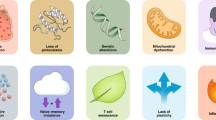
Immunosenescence occurs with progressive age. T cell aging is manifested by immunodeficiency and inflammation. The main mechanisms are thymic involution, mitochondrial dysfunction, genetic and epigenetic alterations, loss of protein stability, reduction of T cell receptor (TCR) repertoire, naïve-memory T cell ratio imbalance, T cell senescence, and lack of effector plasticity. Mesenchymal stem cells (MSCs) are thought to hold great potential as anti-aging therapy. However, the role of MCSs in T cell aging remains elusive. This review makes a tentative summary of the potential role of MSCs in the protection against T cell aging. It might provide a new idea to intervene in the aging of the immune system.







Similar content being viewed by others
Availability of data and materials
All data generated or analyzed during this study are included in this published article.
Abbreviations
- TCR:
-
T cell receptor
- MSCs:
-
1Mesenchymal stem cells
- EPO:
-
Erythropoietin
- TNF-α:
-
Tumor necrosis factor α
- IFN-γ:
-
Interferon γ
- TLR4:
-
Toll-like receptor 4
- LPS:
-
Lipopolysaccharides
- TLR3:
-
Toll-like receptor 3
- IL-6:
-
Interleukin-6
- IL-8:
-
Interleukin-8
- TGF-β:
-
Transforming growth factor β
- IL-4:
-
Interleukin-4
- IL-1RA:
-
Interleukin-1 receptor antagonist
- IDO:
-
Indoleamine 2,3-dioxygenase
- PGE2:
-
Prostaglandin e2
- HLA-G5:
-
Human leukocyte antigen g5
- Th2:
-
T helper 2 cell
- Th17:
-
T helper 17 cell
- Th1:
-
T helper 1 cell
- CXCR3:
-
Chemokine receptor chemokine (C-X-C motif) receptor 3
- CTEC:
-
Cortical thymic epithelial cells
- DN:
-
CD4 and CD8 double negative T cells
- DP:
-
CD4 and CD8 double positive T cells
- SP:
-
CD4 or CD8 single positive T cells
- MHC:
-
Major histocompatibility complexes
- EGF:
-
Epidermal growth factor
- IGF:
-
Insulin-like growth factor
- TEC:
-
Thymic epithelial cell
- S1P:
-
Sphingosine-1-phosphate
- nTMSC:
-
MSCs derived from human neonatal thymus
- MHC-II:
-
Major histocompatibility complex class II
- MHC-I:
-
Major histocompatibility complex class I
- T-MSCs:
-
Tonsillar mesenchymal stem cells
- BMT:
-
Bone marrow transplantation
- FLT3L:
-
Fms-related tyrosine kinase 3 ligand
- FGF7:
-
Fibroblast growth factor 7
- PBM:
-
Photobiomodulation
- ATP:
-
Adenosine triphosphate
- ROS:
-
Reactive oxygen species
- HGF:
-
Hepatocyte growth factor
- HGF-hATMSCs:
-
HGF-overexpressing adipose tissue-derived mesenchymal stem cells
- UC-MSCs:
-
Umbilical cord mesenchymal stem cells
- KGF:
-
Keratinocyte growth factor
- NAD:
-
Nicotinamide adenine dinucleotide
- NADH:
-
Nicotinamide adenine dinucleotide (reduced state)
- TNT:
-
Tunneling nanotubes
- PBMC:
-
Peripheral blood mononuclear cells
- FOXP3:
-
Forkhead box protein P3
- IL2RA:
-
Interleukin-2 receptor subunit alpha
- CTLA4:
-
Cytotoxic t lymphocyte-associated antigen-4
- iTreg:
-
Induced regulatory T cells
- MT:
-
Mitochondrial transfer
- CDKN1A:
-
Cyclin-dependent kinase inhibitor 1A
- MRE11A:
-
Meiotic recombination 11 homolog A
- CDKN2A:
-
Cyclin-dependent kinase inhibitor 2A
- PD-1:
-
Programmed cell death protein 1
- ROR-γ:
-
Related-orphan receptors γ
- SIRT1:
-
Sirtuin1
- VEGF:
-
Vascular endothelial growth factor
- PCNA:
-
Proliferating cell nuclear antigen
- AGRP:
-
Agouti gene-related protein
- STAT3:
-
Signal transducer and activator of transcription 3
- ICAM-1:
-
Intercellular cell adhesion molecule-1
- Tfh:
-
Follicular helper T cells
- OA:
-
Osteoarthritis
- Nrf2:
-
Nuclear erythroid 2-related factor 2
References
Yousefzadeh MJ, Flores RR, Zhu Y et al (2021) An aged immune system drives senescence and ageing of solid organs. Nature 594(7861):100–105
Mittelbrunn M, Kroemer G (2021) Hallmarks of T cell aging. Nat Immunol 22(6):687–698
Zhu Y, Ge J, Huang C, Liu H, Jiang H (2021) Application of mesenchymal stem cell therapy for aging frailty: from mechanisms to therapeutics. Theranostics 11(12):5675–5685
Salvadori M, Cesari N, Murgia A et al (2019) Dissecting the pharmacodynamics and pharmacokinetics of MSCs to overcome limitations in their clinical translation. Mol Ther Methods Clin Dev 14:1–15
Spees JL, Lee RH, Gregory CA (2016) Mechanisms of mesenchymal stem/stromal cell function. Stem Cell Res Ther 7(1):125
Thäte C, Woischwill C, Brandenburg G et al (2021) Non-clinical assessment of safety, biodistribution and tumorigenicity of human mesenchymal stromal cells. Toxicol Rep 8:1960–1969
Shoshani O, Massalha H, Shani N et al (2012) Polyploidization of murine mesenchymal cells is associated with suppression of the long noncoding RNA H19 and reduced tumorigenicity. Cancer Res 72(24):6403–6413
Zander AR, Lange C, Westenfelder C (2011) Mesenchymal stromal cells: main factor or helper in regenerative medicine? Kidney Int Suppl (2011) 1(3):74–76
Caplan AI (2010) Mesenchymal stem cells: the past, the present, the future. Cartilage 1(1):6–9
Trohatou O, Roubelakis MG (2017) Mesenchymal stem/stromal cells in regenerative medicine: past, present, and future. Cell Reprogram 19(4):217–224
Frenette PS, Pinho S, Lucas D, Scheiermann C (2013) Mesenchymal stem cell: keystone of the hematopoietic stem cell niche and a stepping-stone for regenerative medicine. Annu Rev Immunol 31:285–316
Waterman RS, Tomchuck SL, Henkle SL, Betancourt AM (2010) A new mesenchymal stem cell (MSC) paradigm: polarization into a pro-inflammatory MSC1 or an Immunosuppressive MSC2 phenotype. PLoS ONE 5(4):e10088
Galland S, Stamenkovic I (2020) Mesenchymal stromal cells in cancer: a review of their immunomodulatory functions and dual effects on tumor progression. J Pathol 250(5):555–572
Bernardo ME, Fibbe WE (2013) Mesenchymal stromal cells: sensors and switchers of inflammation. Cell Stem Cell 13(4):392–402
Abdolmohammadi K, Pakdel FD, Aghaei H et al (2019) Ankylosing spondylitis and mesenchymal stromal/stem cell therapy: a new therapeutic approach. Biomed Pharmacother 109:1196–1205
Alves VBF, de Sousa BC, Fonseca MTC et al (2019) A single administration of human adipose tissue-derived mesenchymal stromal cells (MSC) induces durable and sustained long-term regulation of inflammatory response in experimental colitis. Clin Exp Immunol 196(2):139–154
Li W, Chen W, Huang S et al (2020) Mesenchymal stem cells prevent overwhelming inflammation and reduce infection severity via recruiting CXCR3(+) regulatory T cells. Clin Transl Immunology 9(10):e1181
Yeo GEC, Ng MH, Nordin FB, Law JX (2021) Potential of mesenchymal stem cells in the rejuvenation of the aging immune system. Int J Mol Sci 22(11)
Cao W, Cao K, Cao J, Wang Y, Shi Y (2015) Mesenchymal stem cells and adaptive immune responses. Immunol Lett 168(2):147–153
Montel-Hagen A, Crooks GM (2019) From pluripotent stem cells to T cells. Exp Hematol 71:24–31
Nitta T, Murata S, Ueno T, Tanaka K, Takahama Y (2008) Thymic microenvironments for T-cell repertoire formation. Adv Immunol 99:59–94
Bleul CC, Corbeaux T, Reuter A et al (2006) Formation of a functional thymus initiated by a postnatal epithelial progenitor cell. Nature 441(7096):992–996
Alam SM, Travers PJ, Wung JL et al (1996) T-cell-receptor affinity and thymocyte positive selection. Nature 381(6583):616–620
Kim HJ, Alonzo ES, Dorothee G, Pollard JW, Sant’Angelo DB (2010) Selective depletion of eosinophils or neutrophils in mice impacts the efficiency of apoptotic cell clearance in the thymus. PLoS ONE 5(7):e11439
Manley NR, Richie ER, Blackburn CC, Condie BG, Sage J (2011) Structure and function of the thymic microenvironment. Front Biosci (Landmark Ed) 16:2461–2477
Nitta T, Ota A, Iguchi T, Muro R, Takayanagi H (2021) The fibroblast: an emerging key player in thymic T cell selection. Immunol Rev 302(1):68–85
Ferreirinha P, Pinheiro RGR, Landry JJM, Alves NL (2022) Identification of fibroblast progenitors in the developing mouse thymus. Development 149(10)
Nitta T, Tsutsumi M, Nitta S et al (2020) Fibroblasts as a source of self-antigens for central immune tolerance. Nat Immunol 21(10):1172–1180
Bockman DE, Kirby ML (1984) Dependence of thymus development on derivatives of the neural crest. Science 223(4635):498–500
Auerbach R (1960) Morphogenetic interactions in the development of the mouse thymus gland. Dev Biol 2:271–284
Itoi M, Kawamoto H, Katsura Y, Amagai T (2001) Two distinct steps of immigration of hematopoietic progenitors into the early thymus anlage. Int Immunol 13(9):1203–1211
Anderson G, Jenkinson EJ, Moore NC, Owen JJ (1993) MHC class II-positive epithelium and mesenchyme cells are both required for T-cell development in the thymus. Nature 362(6415):70–73
Suniara RK, Jenkinson EJ, Owen JJ (2000) An essential role for thymic mesenchyme in early T cell development. J Exp Med 191(6):1051–1056
Jenkinson WE, Jenkinson EJ, Anderson G (2003) Differential requirement for mesenchyme in the proliferation and maturation of thymic epithelial progenitors. J Exp Med 198(2):325–332
Jenkinson WE, Rossi SW, Parnell SM, Jenkinson EJ, Anderson G (2007) PDGFRalpha-expressing mesenchyme regulates thymus growth and the availability of intrathymic niches. Blood 109(3):954–960
Zachariah MA, Cyster JG (2010) Neural crest-derived pericytes promote egress of mature thymocytes at the corticomedullary junction. Science 328(5982):1129–1135
Sitnik KM, Kotarsky K, White AJ et al (2012) Mesenchymal cells regulate retinoic acid receptor-dependent cortical thymic epithelial cell homeostasis. J Immunol 188(10):4801–4809
Wendland K, Niss K, Kotarsky K et al (2018) Retinoic acid signaling in thymic epithelial cells regulates thymopoiesis. J Immunol 201(2):524–532
Mouiseddine M, Mathieu N, Stefani J, Demarquay C, Bertho JM (2008) Characterization and histological localization of multipotent mesenchymal stromal cells in the human postnatal thymus. Stem Cells Dev 17(6):1165–1174
Siepe M, Thomsen AR, Duerkopp N et al (2009) Human neonatal thymus-derived mesenchymal stromal cells: characterization, differentiation, and immunomodulatory properties. Tissue Eng Part A 15(7):1787–1796
Azghadi SM, Suciu M, Gruia AT et al (2016) Mesenchymal stromal cells support the viability and differentiation of thymocytes through direct contact in autologous co-cultures. Histochem Cell Biol 146(2):153–165
Hu KX, Wang MH, Fan C et al (2011) CM-DiI labeled mesenchymal stem cells homed to thymus inducing immune recovery of mice after haploidentical bone marrow transplantation. Int Immunopharmacol 11(9):1265–1270
Choi DW, Cho KA, Lee HJ et al (2020) Co-transplantation of tonsil-derived mesenchymal stromal cells in bone marrow transplantation promotes thymus regeneration and T cell diversity following cytotoxic conditioning. Int J Mol Med 46(3):1166–1174
Odinokov D, Hamblin MR (2018) Aging of lymphoid organs: can photobiomodulation reverse age-associated thymic involution via stimulation of extrapineal melatonin synthesis and bone marrow stem cells? J Biophotonics 11(8):e201700282
Jung WS, Han SM, Kim SM et al (2014) Stimulatory effect of HGF-overexpressing adipose tissue-derived mesenchymal stem cells on thymus regeneration in a rat thymus involution model. Cell Biol Int 38(10):1106–1117
Liu G, Wang L, Pang T et al (2014) Umbilical cord-derived mesenchymal stem cells regulate thymic epithelial cell development and function in Foxn1(-/-) mice. Cell Mol Immunol 11(3):275–284
Uhlin M, Sairafi D, Berglund S et al (2012) Mesenchymal stem cells inhibit thymic reconstitution after allogeneic cord blood transplantation. Stem Cells Dev 21(9):1409–1417
Maeda Y, Chida J (2013) Control of cell differentiation by mitochondria, typically evidenced in dictyostelium development. Biomolecules 3(4):943–966
Xie JH, Li YY, Jin J (2020) The essential functions of mitochondrial dynamics in immune cells. Cell Mol Immunol 17(7):712–721
Quinn KM, Palchaudhuri R, Palmer CS, La Gruta NL (2019) The clock is ticking: the impact of ageing on T cell metabolism. Clin Transl Immunology 8(11):e01091
Li W, Cheng H, Li G, Zhang L (2020) Mitochondrial damage and the road to exhaustion. Cell Metab 32(6):905–907
Desdín-Micó G, Soto-Heredero G, Aranda JF et al (2020) T cells with dysfunctional mitochondria induce multimorbidity and premature senescence. Science 368(6497):1371–1376
Rodriguez AM, Nakhle J, Griessinger E, Vignais ML (2018) Intercellular mitochondria trafficking highlighting the dual role of mesenchymal stem cells as both sensors and rescuers of tissue injury. Cell Cycle 17(6):712–721
Mahrouf-Yorgov M, Augeul L, Da Silva CC et al (2017) Mesenchymal stem cells sense mitochondria released from damaged cells as danger signals to activate their rescue properties. Cell Death Differ 24(7):1224–1238
Cho YM, Kim JH, Kim M et al (2012) Mesenchymal stem cells transfer mitochondria to the cells with virtually no mitochondrial function but not with pathogenic mtDNA mutations. PLoS ONE 7(3):e32778
Las G, Shirihai OS (2014) Miro1: new wheels for transferring mitochondria. Embo J 33(9):939–941
Lin HY, Liou CW, Chen SD et al (2015) Mitochondrial transfer from Wharton’s jelly-derived mesenchymal stem cells to mitochondria-defective cells recaptures impaired mitochondrial function. Mitochondrion 22:31–44
Court AC, Le-Gatt A, Luz-Crawford P et al (2020) Mitochondrial transfer from MSCs to T cells induces Treg differentiation and restricts inflammatory response. EMBO Rep 21(2):e48052
Do JS, Zwick D, Kenyon JD et al (2021) Mesenchymal stromal cell mitochondrial transfer to human induced T-regulatory cells mediates FOXP3 stability. Sci Rep 11(1):10676
Dutra Silva J, Su Y, Calfee CS et al (2021) Mesenchymal stromal cell extracellular vesicles rescue mitochondrial dysfunction and improve barrier integrity in clinically relevant models of ARDS. Eur Respir J 58(1)
Li Y, Shen Y, Hohensinner P et al (2016) Deficient activity of the nuclease MRE11A induces T cell aging and promotes arthritogenic effector functions in patients with rheumatoid arthritis. Immunity 45(4):903–916
Fali T, Papagno L, Bayard C et al (2019) New insights into lymphocyte differentiation and aging from telomere length and telomerase activity measurements. J Immunol 202(7):1962–1969
Ye Z, Li G, Kim C et al (2018) Regulation of miR-181a expression in T cell aging. Nat Commun 9(1):3060
Kim C, Jadhav RR, Gustafson CE et al (2019) Defects in antiviral T cell responses inflicted by aging-associated miR-181a deficiency. Cell Rep 29(8):2202-2216.e2205
Azevedo RI, Minskaia E, Fernandes-Platzgummer A et al (2020) Mesenchymal stromal cells induce regulatory T cells via epigenetic conversion of human conventional CD4 T cells in vitro. Stem Cells 38(8):1007–1019
Wang Q, Li X, Luo J et al (2012) The allogeneic umbilical cord mesenchymal stem cells regulate the function of T helper 17 cells from patients with rheumatoid arthritis in an in vitro co-culture system. BMC Musculoskelet Disord 13:249
Jeng MY, Hull PA, Fei M et al (2018) Metabolic reprogramming of human CD8(+) memory T cells through loss of SIRT1. J Exp Med 215(1):51–62
Ye F, Jiang J, Zong C et al (2020) Sirt1-overexpressing mesenchymal stem cells drive the anti-tumor effect through their pro-inflammatory capacity. Mol Ther 28(3):874–888
Santra M, Dill KA, de Graff AMR (2019) Proteostasis collapse is a driver of cell aging and death. Proc Natl Acad Sci USA 116(44):22173–22178
López-Otín C, Blasco MA, Partridge L, Serrano M, Kroemer G (2013) The hallmarks of aging. Cell 153(6):1194–1217
Pérez VI, Buffenstein R, Masamsetti V et al (2009) Protein stability and resistance to oxidative stress are determinants of longevity in the longest-living rodent, the naked mole-rat. Proc Natl Acad Sci U S A 106(9):3059–3064
Bikorimana JP, El-Hachem N, El-Kadiry AE et al (2020) Thymoproteasome-expressing mesenchymal stromal cells confer protective anti-tumor immunity via cross-priming of endogenous dendritic cells. Front Immunol 11:596303
Lee NK, Park SE, Kwon SJ et al (2017) Agouti related peptide secreted via human mesenchymal stem cells upregulates proteasome activity in an Alzheimer’s disease model. Sci Rep 7:39340
Yin N, Wu C, Qiu J et al (2020) Protective properties of heme oxygenase-1 expressed in umbilical cord mesenchymal stem cells help restore the ovarian function of premature ovarian failure mice through activating the JNK/Bcl-2 signal pathway-regulated autophagy and upregulating the circulating of CD8(+)CD28(-) T cells. Stem Cell Res Ther 11(1):49
Wang Z, Shi Y, Chen W, Wei H, Shang J (2020) Mesenchymal stem cells repair bone marrow damage of aging rats and regulate autophagy and aging genes. Cell Biochem Funct 38(6):792–800
Wang R, Shen Z, Yang L et al (2017) Protective effects of heme oxygenase-1-transduced bone marrow-derived mesenchymal stem cells on reduced-size liver transplantation: role of autophagy regulated by the ERK/mTOR signaling pathway. Int J Mol Med 40(5):1537–1548
Gu X, Li Y, Chen K et al (2020) Exosomes derived from umbilical cord mesenchymal stem cells alleviate viral myocarditis through activating AMPK/mTOR-mediated autophagy flux pathway. J Cell Mol Med 24(13):7515–7530
Xiang J, Jiang T, Zhang W et al (2019) Human umbilical cord-derived mesenchymal stem cells enhanced HK-2 cell autophagy through microRNA-145 by inhibiting the PI3K/AKT/mTOR signaling pathway. Exp Cell Res 378(2):198–205
Egorov ES, Kasatskaya SA, Zubov VN et al (2018) The changing landscape of naive T cell receptor repertoire with human aging. Front Immunol 9:1618
Barda-Saad M, Shav-Tal Y, Rozenszajn AL et al (2002) The mesenchyme expresses T cell receptor mRNAs: relevance to cell growth control. Oncogene 21(13):2029–2036
Zheng S, Huang K, Xia W et al (2021) Mesenchymal stromal cells rapidly suppress tcr signaling-mediated cytokine transcription in activated t cells through the ICAM-1/CD43 interaction. Front Immunol 12:609544
den Braber I, Mugwagwa T, Vrisekoop N et al (2012) Maintenance of peripheral naive T cells is sustained by thymus output in mice but not humans. Immunity 36(2):288–297
Mold JE, Réu P, Olin A et al (2019) Cell generation dynamics underlying naive T-cell homeostasis in adult humans. PLoS Biol 17(10):e3000383
Goronzy JJ, Weyand CM (2019) Mechanisms underlying T cell ageing. Nat Rev Immunol 19(9):573–583
Tu W, Rao S (2016) Mechanisms underlying T cell immunosenescence: aging and cytomegalovirus infection. Front Microbiol 7:2111
Jang E, Jeong M, Kim S et al (2016) Infusion of human bone marrow-derived mesenchymal stem cells alleviates autoimmune nephritis in a lupus model by suppressing follicular helper T-cell development. Cell Transplant 25(1):1–15
Fujii S, Miura Y, Fujishiro A et al (2018) Graft-versus-host disease amelioration by human bone marrow mesenchymal stromal/stem cell-derived extracellular vesicles is associated with peripheral preservation of naive T cell populations. Stem Cells 36(3):434–445
Svobodova E, Krulova M, Zajicova A et al (2012) The role of mouse mesenchymal stem cells in differentiation of naive T-cells into anti-inflammatory regulatory T-cell or proinflammatory helper T-cell 17 population. Stem Cells Dev 21(6):901–910
Cortinovis M, Casiraghi F, Remuzzi G, Perico N (2015) Mesenchymal stromal cells to control donor-specific memory T cells in solid organ transplantation. Curr Opin Organ Transplant 20(1):79–85
Luque-Campos N, Contreras-López RA, Jose Paredes-Martínez M et al (2019) Mesenchymal stem Cells improve rheumatoid arthritis progression by controlling memory t cell response. Front Immunol 10:798
Krampera M, Glennie S, Dyson J et al (2003) Bone marrow mesenchymal stem cells inhibit the response of naive and memory antigen-specific T cells to their cognate peptide. Blood 101(9):3722–3729
Duffy MM, Pindjakova J, Hanley SA et al (2011) Mesenchymal stem cell inhibition of T-helper 17 cell- differentiation is triggered by cell-cell contact and mediated by prostaglandin E2 via the EP4 receptor. Eur J Immunol 41(10):2840–2851
Akbar AN, Henson SM, Lanna A (2016) Senescence of T Lymphocytes: implications for enhancing human immunity. Trends Immunol 37(12):866–876
Czesnikiewicz-Guzik M, Lee WW, Cui D et al (2008) T cell subset-specific susceptibility to aging. Clin Immunol 127(1):107–118
Xiong Y, Wang Y, Zhang J et al (2020) hPMSCs protects against D-galactose-induced oxidative damage of CD4(+) T cells through activating Akt-mediated Nrf2 antioxidant signaling. Stem Cell Res Ther 11(1):468
Zhou B, Zhao Z, Zhang X, Deng W, Li Y (2020) Effect of allogenic bone marrow mesenchymal stem cell transplantation on T cells of old mice. Cell Reprogram 22(1):30–35
Murphy KM, Stockinger B (2010) Effector T cell plasticity: flexibility in the face of changing circumstances. Nat Immunol 11(8):674–680
Hashimoto K, Kouno T, Ikawa T et al (2019) Single-cell transcriptomics reveals expansion of cytotoxic CD4 T cells in supercentenarians. Proc Natl Acad Sci U S A 116(48):24242–24251
Mogilenko DA, Shpynov O, Andhey PS et al (2021) Comprehensive profiling of an aging immune system reveals clonal GZMK(+) CD8(+) T cells as conserved hallmark of inflammaging. Immunity 54(1):99-115.e112
Hagmann S, Gotterbarm T, Müller T et al (2013) The influence of bone marrow- and synovium-derived mesenchymal stromal cells from osteoarthritis patients on regulatory T cells in co-culture. Clin Exp Immunol 173(3):454–462
Bolandi Z, Mokhberian N, Eftekhary M et al (2020) Adipose derived mesenchymal stem cell exosomes loaded with miR-10a promote the differentiation of Th17 and Treg from naive CD4(+) T cell. Life Sci 259:118218
Taechangam N, Walker NJ, Borjesson DL (2021) Feline adipose-derived mesenchymal stem cells induce effector phenotype and enhance cytolytic function of CD8+ T cells. Stem Cell Res Ther 12(1):495
Cen S, Wang P, Xie Z et al (2019) Autophagy enhances mesenchymal stem cell-mediated CD4(+) T cell migration and differentiation through CXCL8 and TGF-β1. Stem Cell Res Ther 10(1):265
Funding
This work was supported by the National Natural Science Foundation of China (No. 81972643, No. 82172962), the Sichuan Science and Technology Project (2021YJ0201), the Luxian People’s Government and Southwest Medical University Scientific and Technological Achievements Transfer and Transformation Strategic Cooperation Project (2019LXXNYKD-07), and the Science and Technology Program of Luzhou, China (No. 2017LZXNYD-Z04, No. 21CGZHPT0001).
Author information
Authors and Affiliations
Contributions
Yueshui Zhao and Jing Shen had the idea for the article. Jie Chen and Bo Wen performed the literature search and data analysis. Xianmei Zhong, Xu Wu, Mingxing Li, Fukuan Du, Yu Chen, and Shuai Deng drafted and critically revised the work. Zhangang Xiao acquired the funding for the work. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhong, X., Chen, J., Wen, B. et al. Potential role of mesenchymal stem cells in T cell aging. J Mol Med 101, 1365–1378 (2023). https://doi.org/10.1007/s00109-023-02371-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00109-023-02371-y