Abstract
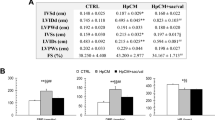
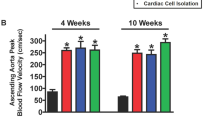
Left ventricular (LV) function influences outcomes in right ventricular (RV) failure. Carvedilol reduces mortality in LV failure and improves RV function in experimental pulmonary arterial hypertension (PAH). However, its impact on ventricular-ventricular interactions and LV function in RV afterload is unknown. We investigated effects of carvedilol on biventricular fibrosis and function in a rat model of persistent PAH. Rats were randomized into three groups: Sham controls, PAH, and PAH + carvedilol. Severe PAH was induced by 60 mg/kg subcutaneous monocrotaline. In the treatment group, oral carvedilol (15 mg/kg/day) was started 2 weeks after monocrotaline injection and continued for 3 weeks until the terminal experiment. Echocardiography and exercise performance were performed at baseline and repeated at the terminal experiment with hemodynamic measurements. LV and RV myocardium were analyzed for hypertrophy, fibrosis, and molecular signaling by protein and mRNA analysis. PAH and PAH + carvedilol rats experienced severely elevated pulmonary arterial pressures and RV hypertrophy. Despite similar RV systolic pressures, carvedilol reduced biventricular collagen content (RV fibrosis area; 13.4 ± 6.5 vs. 5.5 ± 2.7 %, p < 0.001) and expression of transforming growth factor-β1 (TGFβ1) (RV TGFβ1/glyceraldehyde 3-phosphate dehydrogenase (GAPDH) ratio; 1.16 ± 0.39 vs. 0.57 ± 0.22, p < 0.01) and connective tissue growth factor (CTGF) (RV CTGF/GAPDH ratio; 0.49 ± 0.06 vs. 0.35 ± 0.17, p < 0.05). RV pro-apoptotic caspase-8 was increased in PAH compared to controls and was significantly reduced in both ventricles compared to PAH animals by carvedilol. Tissue effects were accompanied by improved biventricular systolic and diastolic performance and exercise treadmill distance (36 ± 30 vs. 80 ± 33 m, p < 0.05). In RV pressure-load, carvedilol improves biventricular fibrosis and function through abrogation of TGFβ1-CTGF signaling.
Key message
• RV afterload caused biventricular injury and dysfunction through TGFβ1-CTGF signaling.
• Carvedilol reduced biventricular TGFβ1-CTGF signaling, fibrosis, and apoptosis.
• Carvedilol improved cardiac output and biventricular function.
• Improved fibrosis and hemodynamics occurred despite persistent RV afterload.








Similar content being viewed by others
References
Kitaori K, He H, Ayres SM, Kawata M, Cowan DB, Friehs I, del Nido PJ, McGowan FX (2009) Development of left ventricular diastolic dysfunction with preservation of ejection fraction during progression of infant right ventricular hypertrophy. Circ Heart Fail 2:599–607
Handoko ML, de Man FS, Allaart CP, Paulus WJ, Westerhof N, Vonk-Noordegraaf A (2010) Perspectives on novel therapeutic strategies for right heart failure in pulmonary arterial hypertension: lessons from the left heart. Eur Respir Rev 19:72–82
Gan CT, Lankhaar JW, Marcus JT, Westerhof N, Marques KM, Bronzwaer JG, Boonstra A, Postmus PE, Vonk-Noordegraaf A (2006) Impaired left ventricular filling due to right-to-left ventricular interaction in patients with pulmonary arterial hypertension. Am J Physiol Heart Circ Physiol 290:H1528–H1533
Alkon J, Humpl T, Manlhiot C, McCrindle BW, Reyes JT, Friedberg MK (2010) Usefulness of the right ventricular systolic to diastolic duration ratio to predict functional capacity and survival in children with pulmonary arterial hypertension. Am J Cardiol 106:430–436
Friedberg MK, Cho MY, Li J, Assad RS, Sun M, Rohailla S, Honjo O, Apitz C, Redington AN (2013) Adverse biventricular remodeling in isolated right ventricular hypertension is mediated by increased TGFbeta1 signaling and is abrogated by angiotensin receptor blockade. Am J Respir Cell Mol Biol 49:1019–1028
Apitz C, Honjo O, Humpl T, Li J, Assad RS, Cho MY, Hong J, Friedberg MK, Redington AN (2012) Biventricular structural and functional responses to aortic constriction in a rabbit model of chronic right ventricular pressure overload. J Thoracic Cardiovasc Surg 144:1494–1501
Nootens M, Kaufmann E, Rector T, Toher C, Judd D, Francis GS, Rich S (1995) Neurohormonal activation in patients with right ventricular failure from pulmonary hypertension: relation to hemodynamic variables and endothelin levels. J Am Coll Cardiol 26:1581–1585
Bogaard HJ, Natarajan R, Mizuno S, Abbate A, Chang PJ, Chau VQ, Hoke NN, Kraskauskas D, Kasper M, Salloum FN et al (2010) Adrenergic receptor blockade reverses right heart remodeling and dysfunction in pulmonary hypertensive rats. Am J Respir Crit Care Med 82:652–660
Tei C, Dujardin KS, Hodge DO, Bailey KR, McGoon MD, Tajik AJ, Seward SB (1996) Doppler echocardiographic index for assessment of global right ventricular function. J Am Soc Echocardiogr 9:838–847
Duan Y, Harada K, Wu W, Ishii H, Takada G (2008) Correlation between right ventricular Tei index by tissue Doppler imaging and pulsed Doppler imaging in fetuses. Pediatr Cardiol 29:739–743
Fukuda Y, Tanaka H, Sugiyama D, Ryo K, Onishi T, Fukuya H, Nogami M, Ohno Y, Emoto N, Kawai H et al (2011) Utility of right ventricular free wall speckle-tracking strain for evaluation of right ventricular performance in patients with pulmonary hypertension. J Am Soc Echocardiogr 24:1101–1108
Koopman LP, Slorach C, Manlhiot C, McCrindle BW, Jaeggi ET, Mertens L, Friedberg MK (2011) Assessment of myocardial deformation in children using Digital Imaging and Communications in Medicine (DICOM) data and vendor independent speckle tracking software. J Am Soc Echocardiogr 24:37–44
Hessel MH, Steendijk P, den Adel B, Schutte CI, van der Laarse A (2006) Characterization of right ventricular function after monocrotaline-induced pulmonary hypertension in the intact rat. Am J Physiol Heart Circ Physiol 291:H2424–H2430
Matute-Bello G, Downey G, Moore BB, Groshong SD, Matthay MA, Slutsky AS, Kuebler WM (2011) An official American Thoracic Society workshop report: features and measurements of experimental acute lung injury in animals. Am J Respir Cell Mol Biol 44:725–738
van Heeswijk RB, De Blois J, Kania G, Gonzales C, Blyszczuk P, Stuber M, Eriksson U, Schwitter J (2013) Selective in vivo visualization of immune-cell infiltration in a mouse model of autoimmune myocarditis by fluorine-19 cardiac magnetic resonance. Circ Cardiovasc Imaging 6:277–284
Sun M, Opavsky MA, Stewart DJ, Rabinovitch M, Dawood F, Wen WH, Liu PP (2003) Temporal response and localization of integrins beta1 and beta3 in the heart after myocardial infarction: regulation by cytokines. Circulation 107:1046–1052
Shehata ML, Lossnitzer D, Skrok J, Boyce D, Lechtzin N, Mathai SC, Girgis RE, Osman N, Lima JA, Bluemke DA et al (2011) Myocardial delayed enhancement in pulmonary hypertension: pulmonary hemodynamics, right ventricular function, and remodeling. AJR Am J Roentgenol 196:87–94
Frey N, Olson EN (2003) Cardiac hypertrophy: the good, the bad, and the ugly. Annu Rev Physiol 65:45–79
Villarreal FJ, Dillmann WH (1992) Cardiac hypertrophy-induced changes in mRNA levels for TGF-beta 1, fibronectin, and collagen. Am J Physiol 262:H1861–H1866
Drake JI, Gomez-Arroyo J, Dumur CI, Kraskauskas D, Natarajan R, Bogaard HJ, Fawcett P, Voelkel NF (2013) Chronic carvedilol treatment partially reverses the right ventricular failure transcriptional profile in experimental pulmonary hypertension. Physiol Genomics 45:449–461
Mori T, Kawara S, Shinozaki M, Hayashi N, Kakinuma T, Igarashi A, Takigawa M, Nakanishi T, Takehara K (1999) Role and interaction of connective tissue growth factor with transforming growth factor-beta in persistent fibrosis: a mouse fibrosis model. J Cell Physiol 181:153–159
Mu Y, Gudey SK, Landstrom M (2012) Non-Smad signaling pathways. Cell Tissue Res 347:11–20
Okada M, Kikuzuki R, Harada T, Hori Y, Yamawaki H, Hara Y (2008) Captopril attenuates matrix metalloproteinase-2 and -9 in monocrotaline-induced right ventricular hypertrophy in rats. J Pharmacol Sci 108:487–494
Stawowy P, Margeta C, Kallisch H, Seidah NG, Chretien M, Fleck E, Graf K (2004) Regulation of matrix metalloproteinase MT1-MMP/MMP-2 in cardiac fibroblasts by TGF-beta1 involves furin-convertase. Cardiovasc Res 63:87–97
Zhang YJ, Xiang MX, San J, Cheng G, Wang SS (2006) Effect of matrine and carvedilol on collagen and MMPs activity of hypertrophy myocardium induced by pressure overload. J Zhejiang Univ Sci B 7:245–250
Dewachter C, Dewachter L, Rondelet B, Fesler P, Brimioulle S, Kerbaul F, Naeije R (2010) Activation of apoptotic pathways in experimental acute afterload-induced right ventricular failure. Crit Care Med 38:1405–1413
Periasamy M, Bhupathy P, Babu GJ (2008) Regulation of sarcoplasmic reticulum Ca2+ ATPase pump expression and its relevance to cardiac muscle physiology and pathology. Cardiovasc Res 77:265–273
Derynck R, Zhang YE (2003) Smad-dependent and Smad-independent pathways in TGF-beta family signaling. Nature 425:577–584
Acknowledgments
The Canadian Institutes of Health Research supported this research.
Conflicts of interest
The authors declare that they have no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Okumura, K., Kato, H., Honjo, O. et al. Carvedilol improves biventricular fibrosis and function in experimental pulmonary hypertension. J Mol Med 93, 663–674 (2015). https://doi.org/10.1007/s00109-015-1251-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00109-015-1251-9