Abstract
Aims
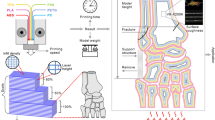
Fused filament fabrication 3D printing with polylactic acid filaments is the most widely used method to generate biomodels at hospitals throughout the world. The main limitation of this manufacturing system is related to the biomodels’ temperature sensitivity, which all but prevents them to be sterilized using conventional methods. The purpose of this study is to define an autoclave temperature-resistant FFF-PLA 3D printing protocol to print 3D fractures biomodels during preoperative planning.
Methods and results
Six different printing protocols were established, each with a different infill percentage. Ten distal radius biomodels were printed with each protocol and each biomodel was subject to 3D scanning. The biomodels were subsequently autoclave-sterilized at 134 °C and subjected to a new scanning process, which was followed by a calculation of changes in area, volume and deformity using the Hausdorff–Besicovitch method. Finally, 192 polylactic acid models were produced using the printing protocol offering the greatest resistance and were contaminated with 31 common nosocomial pathogens to evaluate the effectiveness of sterilizing the model printed using the said protocol. Sterilization resulted in a mean deformation of the biomodel of 0.14 mm, a maximum deformity of 0.75 mm, and a 1% area and a 3.6% volume reduction. Sterilization of the pieces printed using the analyzed protocol was 100% effective.
Conclusions
The analyzed 3D printing protocol may be applied with any FFF-PLA 3D printer, it is safe and does not significantly alter the morphology of biomodels. These results indicate that 3D printing is associated with significant advantages for health centers as it increases their autonomy, allowing them to easily produce 3D biomodels that can be used for the treatment of fractures.






Similar content being viewed by others
References
Tack P, Victor J, Gemmel P, Annemans L. 3D-printing techniques in a medical setting: a systematic literature review. Biomed Eng Online. 2016;15(1):115.
Auricchio F, Marconi S. 3D printing: clinical applications in orthopaedics and traumatology. EFORT Open Reviews. 2016;1(5):121–7.
Vaishya R, Patralekh MK, Vaish A, Agarwal AK, Vijay V. Publication trends and knowledge mapping in 3D printing in orthopaedics. J Clin Orthop Trauma. 2018;9(3):194–201.
Wong KC, Kumta SM, Geel NV, Demol J. One-step reconstruction with a 3D-printed, biomechanically evaluated custom implant after complex pelvic tumor resection. Computer Aided Surg. 2015;20(1):14–23 (New York, NY).
Bizzotto N, Tami I, Santucci A, Adani R, Poggi P, Romani D, et al. 3D printed replica of articular fractures for surgical planning and patient consent: a two years multi-centric experience. 3D Print Med. 2016;2(1):1–6.
Zheng W, Su J, Cai L, Lou Y, Wang J, Guo X, et al. Application of 3D-printing technology in the treatment of humeral intercondylar fractures. Orthop Traumatol Surg Res. 2018;104(1):83–8.
Wang Q, Hu J, Guan J, Chen Y, Wang L. Proximal third humeral shaft fractures fixed with long helical PHILOS plates in elderly patients: benefit of pre-contouring plates on a 3D-printed model—a retrospective study. J Orthop Surg Res. 2018;13(1):203.
Xie L, Chen C, Zhang Y, Zheng W, Chen H, Cai L. Three-dimensional printing assisted ORIF versus conventional ORIF for tibial plateau fractures: a systematic review and meta-analysis. Int J Surg. 2018;57:35–44.
Ferràs-Tarragó J, Sanchis-Alfonso V, Ramírez-Fuentes C, Roselló-Añón A, Baixauli-García F. A 3D-CT analysis of femoral symmetry—surgical implications. J Clin Med. 2020;9(11):3546.
Ferràs-Tarragó J, Sanchis-Alfonso V, Ramírez-Fuentes C, Roselló-Añón A, Baixauli-García F. Locating the origin of femoral maltorsion using 3D volumetric technology—the Hockey stick theory. J Clin Med. 2020;9(12):3835.
Schubert C, van Langeveld MC, Donoso LA. Innovations in 3D printing: a 3D overview from optics to organs. Br J Ophthalmol. 2014;98(2):159–61.
Liu S, Wu G, Chen X, Zhang X, Yu J, Liu M, et al. Degradation behavior in vitro of carbon nanotubes (CNTs)/poly(lactic acid) (PLA) composite suture. Polymers. 2019;11(6):1015.
Singhvi MS, Zinjarde SS, Gokhale DV. Polylactic acid: synthesis and biomedical applications. J Appl Microbiol. 2019;127(6):1612–26.
Liu S, Yu J, Li H, Wang K, Wu G, Wang B, et al. Controllable drug release behavior of polylactic acid (PLA) surgical suture coating with ciprofloxacin (CPFX)-polycaprolactone (PCL)/polyglycolide (PGA). Polymers. 2020;12(2):288.
Tyler B, Gullotti D, Mangraviti A, Utsuki T, Brem H. Polylactic acid (PLA) controlled delivery carriers for biomedical applications. Adv Drug Deliv Rev. 2016;107:163–75.
Shintani H. Ethylene oxide gas sterilization of medical devices. Biocontrol Sci. 2017;22(1):1–16.
Shaheen E, Alhelwani A, Van De Casteele E, Politis C, Jacobs R. Evaluation of dimensional changes of 3D printed models after sterilization: a pilot study. Open Dent J. 2018;12(1):72–9.
Zhao Y, Zhu B, Wang Y, Liu C, Shen C. Effect of different sterilization methods on the properties of commercial biodegradable polyesters for single-use, disposable medical devices. Mater Sci Eng C. 2019;105:110041.
Savaris M, Santos VD, Brandalise RN. Influence of different sterilization processes on the properties of commercial poly(lactic acid). Mater Sci Eng C. 2016;69:661–7.
Aguado-Maestro I, De Frutos-Serna M, González-Nava A, Merino-De Santos AB, García-Alonso M. Are the common sterilization methods completely effective for our in-house 3D printed biomodels and surgical guides? Injury 2020
Cignoni P, Callieri M, Corsini M, Dellepiane M, Ganovelli F, Ranzuglia G. MeshLab: an open-source mesh processing tool. 2008. Available at: https://search.datacite.org/works/https://doi.org/10.2312/localchapterevents/italchap/italianchapconf2008/129-136.
Cignoni P, Callieri M, Corsini M, Dellepiane M, Ganovelli F, Ranzuglia G. MeshLab: an open-source mesh processing tool. In: Eurographics Italian Chapter Conference 2008, pp. 129–136 (2008).
Foo GL, Kwek EBK Are three-dimensional printed models useful for preoperative planning of tibial plafond fractures? J Foot Ankle Surg. 2019;58(4):723–729.
Xiong L, Li X, Li H, Chen Z, Xiao T. The efficacy of 3D printing-assisted surgery for traumatic fracture: a meta-analysis. Postgrad Med J. 2019;95(1126):414–9.
Kang HJ, Kim BS, Kim SM, Kim YM, Kim HN, Park JY, et al. Can preoperative 3D printing change surgeon’s operative plan for distal tibia fracture? Biomed Res Int. 2019;2019:1–7.
Boursier J, Fournet A, Bassanino J, Manassero M, Bedu A, Leperlier D. Reproducibility, accuracy and effect of autoclave sterilization on a thermoplastic three-dimensional model printed by a desktop fused deposition modelling three-dimensional printer. Vet Comparative Orthop Traumatol. 2018;31(6):422–30.
Standau T, Long H, Murillo Castellón S, Brütting C, Bonten C, Altstädt V. Evaluation of the zero shear viscosity, the D-content and processing conditions as foam relevant parameters for autoclave foaming of standard polylactide (PLA). Materials. 2020;13(6):1371.
Zolfagharian A, Khosravani MR, Kaynak A. Fracture resistance analysis of 3D-printed polymers. Polymers. 2020;12(2):302.
Török G, Gombocz P, Bognár E, Nagy P, Dinya E, Kispélyi B, et al. Effects of disinfection and sterilization on the dimensional changes and mechanical properties of 3D printed surgical guides for implant therapy—pilot study. BMC Oral Health. 2020;20(1):19.
Ferràs-Tarragó J, Jordà-Gómez P, Català-de-las-Marinas J, Antequera-Cano JM, Barrés-Carsí M. A new universal 3D-printable device to prevent excessive drilling in orthopedic surgery. Euro J Trauma Emergency Surgery. 2020. https://doi.org/10.1007/s00068-020-01465-3.
Funding
This study was funded by a grant from the Spanish Society of Orthopedic and Trauma Surgery (Grant number 2020/1).
Author information
Authors and Affiliations
Contributions
JF-T: Development of the idea and 3D-printed samples. OS-B: Cultures and sterilization evaluation. JMS-A: Cultures and sterilization evaluation. MA-S: Statistical analyses. CDC: Evaluation of the results and critical analysis. JVA-V: Evaluation of the results and critical analysis. FB-G: Biomechanical analysis.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare they have no conflict of interest with respect to the present study.
Rights and permissions
About this article
Cite this article
Ferràs-Tarragó, J., Sabalza-Baztán, O., Sahuquillo-Arce, J.M. et al. Autoclave sterilization of an in-house 3D-printed polylactic acid piece: biological safety and heat-induced deformation. Eur J Trauma Emerg Surg 48, 3901–3910 (2022). https://doi.org/10.1007/s00068-021-01672-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00068-021-01672-6