Abstract
Purpose
Complement is activated in hemorrhagic shock and protective effects by specific complement inhibition were shown. However, it remains unclear if complement activation contributes to the local tissue damage and organ failure. Zonulin is known to activate complement and affect organ failure. Therefore, local and systemic complement activation during hemorrhagic shock and its consequences on zonulin were examined.
Methods
Porcine hemorrhagic shock (n = 9) was initiated with mean arterial blood pressure maintained constant for 4 h before retransfusion. Before, 4 h after hemorrhage and 12 and 22 h after resuscitation, central and renal blood samples were drawn. Analysis included HMGB-1, C3a, and zonulin (blood and kidney homogenisates) as well as terminal complement complex (TCC) and CH50 (blood). Organ samples were taken for histological and immunohistochemical analyses (C3c).
Results
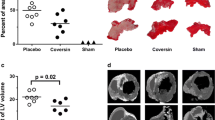
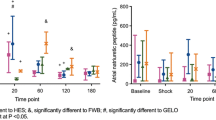
HMGB-1 was significantly elevated in plasma 4 h after hemorrhagic shock and in homogenized kidneys. TCC after 12 h was significantly elevated centrally, while renal levels were not altered. In contrast, CH50 showed diminished renal values, while normal central levels were observed. Local complement activation was observed with enhanced C3c deposition in kidneys. Zonulin showed significantly diminished levels at 12 and 22 h after hemorrhagic shock (central and renal) and significantly correlated with levels of CH50 and neutrophil gelatinase-associated lipocalin (NGAL).
Conclusion
The more pronounced complement activation centrally might indicate consumption of complement products in kidney tissue, which is underlined by C3c staining. Together with diminished levels of zonulin in both systemic and local samples, results could indicate the involvement of complement as well as zonulin in acute kidney failure.





Similar content being viewed by others
References
Ehrnthaller C, et al. New insights of an old defense system: structure, function, and clinical relevance of the complement system. Mol Med. 2011;17(3–4):317–29.
Huber-Lang M, Ignatius A, Brenner RE. Role of complement on broken surfaces after trauma. Adv Exp Med Biol. 2015;865:43–55.
Arumugam TV, et al. Complement mediators in ischemia-reperfusion injury. Clin Chim Acta. 2006;374(1–2):33–45.
Tschoeke SK, Ertel W. Immunoparalysis after multiple trauma. Injury. 2007;38(12):1346–57.
Huber-Lang MS, et al. Complement-induced impairment of innate immunity during sepsis. J Immunol. 2002;169(6):3223–31.
Ward PA. Sepsis, apoptosis and complement. Biochem Pharmacol. 2008;76(11):1383–8.
Huber-Lang MS, et al. Protection of innate immunity by C5aR antagonist in septic mice. FASEB J. 2002;16(12):1567–74.
Arumugam TV, et al. A small molecule C5a receptor antagonist protects kidneys from ischemia/reperfusion injury in rats. Kidney Int. 2003;63(1):134–42.
Ehrnthaller C, et al. Alteration of complement hemolytic activity in different trauma and sepsis models. J Inflamm Res. 2012;5:59–66.
Chen D, et al. Inhibition of complement C3 might rescue vascular hyporeactivity in a conscious hemorrhagic shock rat model. Microvasc Res. 2016;105:23–9.
Goering J, Pope MR, Fleming SD. TLR2 regulates complement-mediated inflammation induced by blood loss during hemorrhage. Shock. 2016;45(1):33–9.
Cunningham PN, et al. Complement is activated in kidney by endotoxin but does not cause the ensuing acute renal failure. Kidney Int. 2000;58(4):1580–7.
Dalle Lucca JJ, et al. Effects of C1 inhibitor on tissue damage in a porcine model of controlled hemorrhage. Shock. 2012;38(1):82–91.
Dalle Lucca JJ, et al. Decay-accelerating factor limits hemorrhage-instigated tissue injury and improves resuscitation clinical parameters. J Surg Res. 2013;179(1):153–67.
Rittirsch D, et al. Zonulin as prehaptoglobin2 regulates lung permeability and activates the complement system. Am J Physiol Lung Cell Mol Physiol. 2013;304(12):L863–72.
Klaus DA, et al. Increased plasma zonulin in patients with sepsis. Biochem Med (Zagreb). 2013;23(1):107–11.
Knoller E, et al. Effects of hyperoxia and mild therapeutic hypothermia during resuscitation from porcine hemorrhagic shock. Crit Care Med. 2016;44(5):e264–77.
van Vuuren BJ, et al. Electroluminescent TCC, C3dg and fB/Bb epitope assays for profiling complement cascade activation in vitro using an activated complement serum calibration standard. J Immunol Methods. 2014;402(1–2):50–6.
Venereau E, et al. HMGB1 and leukocyte migration during trauma and sterile inflammation. Mol Immunol. 2013;55(1):76–82.
Cohen MJ, et al. Early release of high mobility group box nuclear protein 1 after severe trauma in humans: role of injury severity and tissue hypoperfusion. Crit Care. 2009;13(6):R174.
Yang H, et al. The many faces of HMGB1: molecular structure-functional activity in inflammation, apoptosis, and chemotaxis. J Leukoc Biol. 2013;93(6):865–73.
Ganter MT, et al. Role of the alternative pathway in the early complement activation following major trauma. Shock. 2007;28(1):29–34.
Frey A, et al. Complement C3c as a biomarker in heart failure. Mediators Inflamm. 2013;2013:716902.
Ficek J, et al. Relationship between plasma levels of zonulin, bacterial lipopolysaccharides, D-lactate and markers of inflammation in haemodialysis patients. Int Urol Nephrol. 2017;49(4):717–25.
Dschietzig TB, et al. Plasma Zonulin and its association with kidney function, severity of heart failure, and metabolic inflammation. Clin Lab. 2016;62(12):2443–7.
Acknowledgements
This work is supported by grants from the German Research Foundation (DFG) to MH-L and P-R (SFB1149 A01, B03, Z02).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors report no conflict of interests.
Rights and permissions
About this article
Cite this article
Ehrnthaller, C., Schultze, A., Wakileh, G. et al. Hemorrhagic shock induces renal complement activation. Eur J Trauma Emerg Surg 47, 373–380 (2021). https://doi.org/10.1007/s00068-019-01187-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00068-019-01187-1