Abstract
Purpose
To evaluate the efficacy and safety of ablative dose hypofractionated proton beam therapy (PBT) for patients with stage I and recurrent non-small cell lung carcinoma (NSCLC).
Patients and methods
A total of 55 patients with stage I (n = 42) and recurrent (n = 13) NSCLC underwent hypofractionated PBT and were retrospectively reviewed. A total dose of 50–72 CGE (cobalt gray equivalent) in 5–12 fractions was delivered.
Results
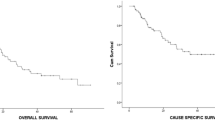
The median follow-up duration was 29 months (range 4–95 months). There were 24 deaths (43.6%) during the follow-up period: 11 died of disease progression and 13 from other causes. Kaplan–Meier overall survival rate (OS) at 3 years was 54.9% and the median OS was 48.6 months (range 4–95 months). Local progression was observed in 7 patients and the median time to local progression was 9.3 months (range 5–14 months). Cumulative actuarial local control rate (LCR), lymph node metastasis-free survival, and distant metastasis-free survival rates at 3 years were 85.4, 78.4, and 76.5%, respectively. Larger tumor diameter was significantly associated with poorer LCR (3-year: 94% for ≤3 cm vs. 65% for >3 cm, p = 0.006) on univariate analysis and also an independent prognostic factor for LCR (HR 6.9, 95% CI = 1.3–37.8, p = 0.026) on multivariate analysis. No grade 3 or 4 treatment-related toxicities developed. One grade 5 treatment-related adverse event occurred in a patient with symptomatic idiopathic pulmonary fibrosis.
Conclusions
Ablative dose hypofractionated PBT was safe and promising for stage I and recurrent NSCLC.
Zusammenfassung
Zweck
Beurteilung von Wirksamkeit und Sicherheit hypofraktionierter Protonentherapie (PBT) mit ablativen Dosen für nichtkleinzellige Lungenkarzinome (NSCLC) im Stadium I und rekurrierende NSCLC.
Methode und Materialien
Retrospektiv wurden insgesamt 55 NSCLC-Patienten (Stadium I: n = 42; rekurrierender Tumor: n = 13), analysiert. Sie waren mit einer Gesamtdosis von 50–72 CGE („cobalt gray equivalent“) in 5–12 Fraktionen behandelt worden.
Ergebnisse
Der Median der Follow-Up-Dauer lag bei 29 Monaten (4–95). Während der Follow-Up-Periode verstarben 24 (43,6%) Patienten, 11 an einem Erkrankungsprogress, 13 an anderen Ursachen. Die 3‑Jahre-Kaplan-Meier-Gesamtüberlebensrate (OS) war 54,9%, der Median lag bei 48,6 Monaten (4–95). Ein lokaler Progress zeigte sich bei 7 Patienten, der Median der Zeit bis zum lokalen Progress lag bei 9,3 Monaten (5–14). Die aktuarische kumulative lokale Kontrollrate (LCR) und die lymphknoten- bzw. die fernmetastasenfreie Überlebensraten betrugen 85,4, 78,4, bzw. 76,5%. Ein größerer Tumordurchmesser war in der univariaten Analyse statistisch signifikant mit einer ungünstigeren LCR assoziiert (3-Jahre: 94% für ≤3 cm vs. 65% für >3 cm, p = 0,006) und erwies sich auch in der multivariaten Analyse als unabhängiger Prognosefaktor für die LCR (HR 6,9, 95%-KI 1,3–37,8, p = 0,026) ist. Therapiebedingte Grad-3- oder -4-Toxizitäten traten nicht auf, bei einem Patienten mit symptomatischer idiopathischer Lungenfibrose kam es zu einer unerwünschten Therapiewirkung Grad 5.
Schlussfolgerung
Die hypofraktionierte PBT mit ablativen Dosen hat sich als sicheres und erfolgversprechendes Therapieverfahren für NSCLC im Stadium I und für rekurrierenden NSCLC erwiesen.

Similar content being viewed by others
References
Ferlay J, Shin HR, Bray F et al (2010) Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int J Cancer 127(12):2893–2917
Mountain CF (1997) Revisions in the international system for staging lung cancer. Chest J 111(6):1710–1717
Qiao X, Tullgren O, Lax I et al (2003) The role of radiotherapy in treatment of stage I non-small cell lung cancer. Lung Cancer 41(1):1–11
Nyman J, Johansson K‑A, Hultén U (2006) Stereotactic hypofractionated radiotherapy for stage I non-small cell lung cancer – mature results for medically inoperable patients. Lung Cancer 51(1):97–103
Soldà F, Lodge M, Ashley S et al (2013) Stereotactic radiotherapy (SABR) for the treatment of primary non-small cell lung cancer; systematic review and comparison with a surgical cohort. Radiother Oncol 109(1):1–7
Guckenberger M, Allgäuer M, Appold S et al (2013) Safety and efficacy of stereotactic body radiotherapy for stage I non-small-cell lung cancer in routine clinical practice: a patterns-of-care and outcome analysis. J Thorac Oncol 8(8):1050–1058
Auberger T, Seydl K, Futschek T et al (2007) Photons or protons: precision radiotherapy of lung cancer. Strahlenther Onkol 183:3–6
Oshiro Y, Mizumoto M, Okumura T et al (2012) Results of proton beam therapy without concurrent chemotherapy for patients with unresectable stage III non-small cell lung cancer. J Thorac Oncol 7(2):370–375
Hata M, Tokuuye K, Kagei K et al (2007) Hypofractionated high-dose proton beam therapy for stage I non-small-cell lung cancer: preliminary results of a phase I/II clinical study. Int J Radiat Oncol Biol Phys 68(3):786–793
Chang JY, Komaki R, Wen HY et al (2011) Toxicity and patterns of failure of adaptive/ablative proton therapy for early-stage, medically inoperable non-small cell lung cancer. Int J Radiat Oncol Biol Phys 80(5):1350–1357
Nihei K, Ogino T, Ishikura S, Nishimura H (2006) High-dose proton beam therapy for stage I non-small-cell lung cancer. Int J Radiat Oncol Biol Phys 65(1):107–111
Nakayama H, Sugahara S, Tokita M et al (2010) Proton beam therapy for patients with medically inoperable stage I non-small-cell lung cancer at the University of Tsukuba. Int J Radiat Oncol Biol Phys 78(2):467–471
Iwata H, Murakami M, Demizu Y et al (2010) High dose proton therapy and carbon ion therapy for stage I nonsmall cell lung cancer. Cancer 116(10):2476–2485
Kanemoto A, Okumura T, Ishikawa H et al (2014) Outcomes and prognostic factors for recurrence after high-dose proton beam therapy for centrally and peripherally located stage I non-small-cell lung cancer. Clin Lung Cancer 15(2):e7–e12
Bush DA, Cheek G, Zaheer S et al (2013) High-dose hypofractionated proton beam radiation therapy is safe and effective for central and peripheral early-stage non-small cell lung cancer: results of a 12-year experience at Loma Linda University Medical Center. Int J Radiat Oncol Biol Phys 86(5):964–968
Timmerman R, Galvin J, Michalski J et al (2006) Accreditation and quality assurance for Radiation Therapy Oncology Group: multicenter clinical trials using stereotactic body radiation therapy in lung cancer. Acta Oncol 45(7):779–786
Timmerman R, McGarry R, Yiannoutsos C et al (2006) Excessive toxicity when treating central tumors in a phase II study of stereotactic body radiation therapy for medically inoperable early-stage lung cancer. J Clin Oncol 24(30):4833–4839
Baek H‑J, Kim T‑H, Shin D et al (2008) Radiobiological characterization of proton beam at the National Cancer Center in Korea. J Radiat Res 49(5):509–515
Huang K, Senthi S, Palma DA et al (2013) High-risk CT features for detection of local recurrence after stereotactic ablative radiotherapy for lung cancer. Radiother Oncol 109(1):51–57
RTOG 0813 protocal information: Seamless phase I/II study of stereotactic lung radiotherapy (SBRT) for early stage, centrally located, non-small cell lung cancer (NSCLC) in medically inoperable patients. 2014.https://www.rtog.org/ClinicalTrials/ProtocolTable/StudyDetails.aspx?study=0813
Charlson M, Szatrowski TP, Peterson J et al (1994) Validation of a combined comorbidity index. J Clin Epidemiol 47(11):1245–1251
Onishi H, Shirato H, Nagata Y et al (2007) Hypofractionated stereotactic radiotherapy (HypoFXSRT) for stage I non-small cell lung cancer: updated results of 257 patients in a Japanese multi-institutional study. J Thorac Oncol 2(7):S94–S100
Westover KD, Seco J, Adams JA et al (2012) Proton SBRT for medically inoperable stage I NSCLC. J Thorac Oncol 7(6):1021
Onimaru R, Fujino M, Yamazaki K et al (2008) Steep dose-response relationship for stage I non-small-cell lung cancer using hypofractionated high-dose irradiation by real-time tumor-tracking radiotherapy. Int J Radiat Oncol Biol Phys 70(2):374–381
Olsen JR, Robinson CG, El Naqa I et al (2011) Dose-response for stereotactic body radiotherapy in early-stage non-small-cell lung cancer. Int J Radiat Oncol Biol Phys 81(4):e299–e303
Underberg RW, Lagerwaard FJ, Slotman BJ et al (2005) Use of maximum intensity projections (MIP) for target volume generation in 4DCT scans for lung cancer. Int J Radiat Oncol Biol Phys 63(1):253–260
Seppenwoolde Y, Shirato H, Kitamura K et al (2002) Precise and real-time measurement of 3D tumor motion in lung due to breathing and heartbeat, measured during radiotherapy. Int J Radiat Oncol Biol Phys 4:822–834
Liu HH, Balter P, Tutt T et al (2007) Assessing respiration-induced tumor motion and internal target volume using four-dimensional computed tomography for radiotherapy of lung cancer. Int J Radiat Oncol Biol Phys 68(2):531–540
Guckenberger M, Wilbert J, Meyer J et al (2007) Is a single respiratory correlated 4D-CT study sufficient for evaluation of breathing motion? Int J Radiat Oncol Biol Phys 67(5):1352–1359
Kang Y, Zhang X, Chang JY et al (2007) 4D Proton treatment planning strategy for mobile lung tumors. Int J Radiat Oncol Biol Phys 67(3):906–914
Brandner ED, Wu A, Chen H et al (2006) Abdominal organ motion measured using 4D CT. Int J Radiat Oncol Biol Phys 65(2):554–560
Berman AT, James SS, Rengan R (2015) Proton beam therapy for non-small cell lung cancer: current clinical evidence and future directions. Cancers (Basel) 7(3):1178–1190
Ohara K, Okumura T, Akisada M et al (1989) Irradiation synchronized with respiration gate. Int J Radiat Oncol Biol Phys 17(4):853–857
Heinzerling JH, Anderson JF, Papiez L et al (2008) Four-dimensional computed tomography scan analysis of tumor and organ motion at varying levels of abdominal compression during stereotactic treatment of lung and liver. Int J Radiat Oncol Biol Phys 70(5):1571–1578
Clarke K, Taremi M, Dahele M et al (2012) Stereotactic body radiotherapy (SBRT) for non-small cell lung cancer (NSCLC): is FDG-PET a predictor of outcome? Radiother Oncol 104(1):62–66
Takeda A, Yokosuka N, Ohashi T et al (2011) The maximum standardized uptake value (SUVmax) on FDG-PET is a strong predictor of local recurrence for localized non-small-cell lung cancer after stereotactic body radiotherapy (SBRT). Radiother Oncol 101(2):291–297
Hoopes DJ, Tann M, Fletcher JW et al (2007) FDG-PET and stereotactic body radiotherapy (SBRT) for stage I non-small-cell lung cancer. Lung Cancer 56(2):229–234
Onishi H, Shirato H, Nagata Y et al (2011) Stereotactic body radiotherapy (SBRT) for operable stage I non-small-cell lung cancer: Can SBRT be comparable to surgery? Int J Radiat Oncol Biol Phys 81(5):1352–1358
Hoppe BS, Huh S, Flampouri S et al (2010) Double-scattered proton-based stereotactic body radiotherapy for stage I lung cancer: a dosimetric comparison with photon-based stereotactic body radiotherapy. Radiother Oncol 97(3):425–430
Takeda A, Enomoto T, Sanuki N et al (2008) Acute exacerbation of subclinical idiopathic pulmonary fibrosis triggered by hypofractionated stereotactic body radiotherapy in a patient with primary lung cancer and slightly focal honeycombing. Radiat Med 26(8):504–507
Acknowledgement
This study was supported by a National Cancer Center Grant (NCCCTS10494).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
S.U. Lee, S.H. Moon, K.H. Cho, H.R. Pyo, J.Y. Kim, D.Y. Kim, T.H. Kim, Y.-G. Suh, and Y.-J. Kim state, that there are no conflicts of interest.Acknowledgement This study was supported by a National Cancer Center Grant (NCCCTS10494).
The study was performed in accordance with the guidelines of the institutional review board, which waived the requirements of informed consent due to the retrospective nature of this study.
Rights and permissions
About this article
Cite this article
Lee, S.U., Moon, S.H., Cho, K.H. et al. Ablative dose proton beam therapy for stage I and recurrent non-small cell lung carcinomas. Strahlenther Onkol 192, 649–657 (2016). https://doi.org/10.1007/s00066-016-0985-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00066-016-0985-9