Abstract
Background
Acute large vessel occlusion due to underlying intracranial atherosclerotic stenosis (ICAS-LVO) increases the difficulty of revascularization, resulting in frequent re-occlusion. The establishment of its pathogenesis before endovascular treatment (EVT) is beneficial for patients. We aimed at developing and validating a clinical prediction model for ICAS-LVO patients before EVT.
Methods
Patients with acute large vessel occlusion at Jining No. 1 People’s Hospital from January 2019 to September 2021 were retrospectively included as the training cohort. The 70 patients who met the inclusion and exclusion criteria were included in the validation cohort (October 2021 to May 2022). Demographics, onset form, medical history, digital subtraction angiography (DSA) imaging data, and laboratory test data were collected. Preprocedural parameters for the ICAS-LVO risk prediction model were established by stepwise logistic regression controlling for the confounding effects. Then, we constructed a nomogram model and evaluated its performance via the Hosmer-Lemeshow goodness-of-fit test, area under the ROC curve (AUC) analysis.
Results
The 231 acute LVO patients were included in the final analysis, 74 (32.3%) patients were ICAS-LVO. A preoperative diagnosis prediction model consisting of five predictors for ICAS-LVO, including fluctuating symptoms, NIHSS < 16, atrial fibrillation, tapered sign, and ASITN/SIR score ≥ 2. The model depicted an acceptable calibration (Hosmer-Lemeshow test, p = 0.451) and good discrimination (AUC, 0.941; 95% confidence interval, 0.910–0.971). The optimal cut-off value for the ICAS-LVO scale was 2 points, with 86.5% sensitivity, 91.1% specificity, and 90.5% accuracy. In the validation cohort, the discriminative ability was promising with an AUC value of 0.897, implying a good predictive performance.
Conclusion
The established ICAS-LVO scale, which is composed of five predictors: fluctuating symptoms, NIHSS < 16, atrial fibrillation, tapered sign, and ASITN/SIR score ≥ 2, has a good predictive value for ICAS-LVO in Chinese populations.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Globally, acute ischemic stroke (AIS) due to large vessel occlusion (LVO) is among the leading causes of mortality and disability. Several randomized controlled trials [1,2,3,4,5] have shown that mechanical thrombectomy (MT) can effectively and safely improve the 90-day clinical outcomes for patients with anterior circulation acute ischemic stroke caused by LVO. It has been reported that MT is a potential option for treatment of acute vertebrobasilar artery occlusion [6,7,8]. Currently, endovascular treatment (EVT) has become the standard treatment for acute ischemic stroke in patients with LVO [9]; however, treatment outcomes of EVT for acute ischemic stroke with LVO are not always ideal. Shorter times to recanalization and higher recanalization rates are important for favorable prognostic outcomes [10].
Large vessel occlusion due to intracranial atherosclerosis (ICAS-LVO) is prevalent among Asians, and the incidences are higher than those found in other areas of the world [11]. Compared to LVO caused by embolism, stent–retriever and aspiration thrombectomy techniques are less efficacious in ICAS-LVO [12], with a markedly low recanalization rate, high re-occlusion rate and longer puncture-to-reperfusion time during endovascular procedures. Successful revascularization requires rescue treatment with intra-arterial thrombolysis, balloon angioplasty, or stenting [13,14,15]. Establishment of its pathogenesis before interventional surgery will inform on targeted treatment and help to have a smooth procedure.
Unique pathogenesis is associated with more complex and time-consuming surgical procedures, which present significant challenges in ICAS-LVO intervention [16, 17]. Therefore, a new, accurate and comprehensive predictive modality is required for patients with AIS due to ICAS-LVO. In this study, we developed and validated a clinical prediction model for EVT patients.
Methods
Patient Selection
We retrospectively evaluated patients who had been subjected to emergency EVT due to acute large vessel ischemic stroke at Jining No. 1 People’s Hospital stroke center from January 2019 to September 2021. The EVT procedures included stent retrieval, aspiration, angioplasty, stenting or a combination of these techniques.
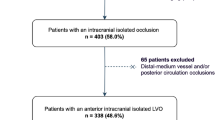
ICAS-LVO was defined as: (1) intracranial artery fixed stenosis of > 70% when successful reperfusion was achieved and (2) degree of intracranial artery stenosis > 50% in addition to either flow and perfusion impairment on angiography or evident re-occlusion tendency even after adequate treatment with stent retrievers. Embolic-LVO was defined as the absence of residual stenosis in occluded segments after successful reperfusion of the occlusion vessel. The exclusion criteria were: (1) the pathogenesis of occluded vessels due to failed recanalization cannot be reliably assessed, (2) when the cause of vascular occlusion was vasculitis, or moyamoya disease, (3) presence of tandem lesions associated with the carotid or vertebral arteries in the extracranial segment (including extracranial stenosis of the vertebral or carotid artery, carotid dissection) and (4) DSA image information is missing. This retrospective, observational study was approved by the Ethical Committee of No. 1 People’s Hospital of Jining.
Data Collection
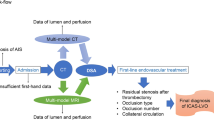
Immediately after admission, all patients were subjected to routine blood tests, serum biochemistry tests, blood coagulation tests, electrocardiogram, brain CT NIHSS, mRS, and GCS. Data on demographics, onset form, medical history, digital subtraction angiography (DSA) imaging and laboratory tests were retrospectively collected. Then, data were double entered using the EpiData Entry software v3.1 (EpiData Association, Odense, Denmark). To eliminate selection bias, radiologic assessments were conducted by two neuroradiologists and one neurologist.
Predictive Factors
Fluctuating symptoms are defined as symptomatic fluctuations and progressive aggravation during the early course of neurologic deficits [18]. The tapered sign denotes occlusive clot signs, described as the appearance of a tapered beak-like or flame-like sign [19, 20]. The American Society of Interventional and Therapeutic Neuroradiology/Society of Interventional Radiology (ASITN/SIR) developed the collateral flow grading scale for DSA [21] to evaluate collateral circulation. The National Institute of Health stroke scale (NIHSS) is a comprehensive stroke scale for evaluating neurological deficits.
Statistical Analyses
Multiple sixfold imputation was performed using the chained equations method to fill the missing blood test results (missing values < 5%). Baseline characteristics were compared between the ICAS-LVO and embolic LVO groups. Categorical variables are presented as numbers and percentages N (%) and compared using the χ2-testand Fisherʼs, exact test. Continuous variables are presented as mean ± standard deviation if normally distributed and as medians and interquartile ranges (IQR), if not normally distributed. The Mann-Whitney U test and the t‑test were used to compare continuous variables between groups.
Multivariable logistic regression analysis was performed for variables with p < 0.05 in the univariate regression analysis. Cut-off values were determined using the maximum of Youden index. Continuous variables were translated into categorical variables. Collinearity diagnosis was performed using the variance inflation factor (VIF). Binary logistic regression (forward, backward) was used to evaluate the predictors and to generate the regression model as well as its related odds ratio (OR), 95% confidence interval (CI), beta coefficient and p values. The beta coefficients were rounded to the closest integer to generate a brief ICAS-LVO scale. The ROC curve was used to calculate the optimal cut-off value of the area under the curve (AUC) to assess the predictive accuracy of the scale and discriminatory ability of the model. The Hosmer-Lemeshow test was performed to assess calibration. A nomogram was built on the brief predictive model as a graphical presentation.
Predictive Models
Analyses were performed using the SPSS software package, version 22 (IBM, Armonk, NY, USA) and R software, version 4.1.3 (R Statistical Software, R Foundation for Statistical Computing, Vienna, Austria).
Results
Tables 1, 2 and 4; Figs. 2 and 4.
Schematic presentation of the ICAS-LVO prediction model. NIHSS National Institutes of Health stroke scale, AF atrial fibrillation, ASITN/SIR score American Society of Interventional and Therapeutic Neuroradiology/Society of Interventional Radiology, β‑coefficient the regression coefficient, OR odds ratios, CI confidence interval
Nomogram for predicting the probability of ICAS-LVO in patients with acute ischemic stroke. NIHSS National Institutes of Health stroke scale, AF atrial fibrillation, ASITN/SIR score American Society of Interventional and Therapeutic Neuroradiology/Society of Interventional Radiology, ICAS-LVO acute large vessel occlusion due to intracranial atherosclerosis
Patient Baseline Characteristics
From January 2019 to September 2021, a total of 311 consecutive AIS patients with LVO were enrolled in the modelling group. Of the patients 32 were excluded because the pathogenesis of occluded vessels could not be reliably assessed due to unsuccessful revascularization, while 3 were excluded because of missing DSA image information. Then, 272 patients were subjected to DSA analyses. Based on vascular occlusive mechanisms, 45 patients (moyamaya disease n = 2, tandem lesions associated with the carotid or vertebral arteries in the extracranial segment n = 42, including extracranial stenosis of the vertebral or carotid artery carotid dissection n = 35, extracranial carotid dissection n = 7) were excluded. Finally, 231 patients were analyzed (Fig. 1).
Baseline characteristics for the training cohort are presented in Table 1. The median age for all participants was 65 ± 11.54 years and the number of males was 163 (70.56%); baseline NIHSS 18.0 (IQR 15.0, 21.0); GCS score 9.0 (IQR 7, 12); mRS score on admission 5.0 (IQR 4.0, 5.0); ASPECTS (pr-ASPECTS) 7 (IQR 7.0, 8.0); ASITN/SIR score 1.0 (IQR 0.0, 2.0). Risk factors included history of hypertension (n = 122, 52.81%); diabetes (n = 65, 28.14%); CHD n = 67 (29.00%); AF n = 87 (37.66%); VHD n = 19 (8.25%); ICAS n = 14 (6.06%); ischemic stroke n = 50 (21.65%); ICH, n = 6 (2.60%); smoking n = 72 (31.17%); and drinking 72 (n = 31.17%). Based on findings from DSA, patients were divided into two groups: ICAS-LVO group 74 (32.04%) and embolic-LVO group 157 (67.96%).
The ICAS-LVO and embolic-LVO groups differed in various aspects (Tables 1 and 2). Compared to the embolic-LVO group, patients with ICAS-LVO were younger, male, had higher blood pressure and blood lipid levels at admission (SBP:157.53 mmHg ± 23.78 mmHg vs. 149.41 mmHg ± 23.44 mmHg, p = 0.016, DBP: 93.50 mmHg [IQR 80.00 mmHg, 102.00 mmHg] vs. 85.00 mmHg [IQR 75.00 mmHg, 95.00 mmHg] p = 0.001; VLDL, very-low-density lipoprotein (0.71 mmol/L [IQR 0.43 mmol/L, 1.10 mmol/L] vs. 0.52 mmol/L [IQR 0.31 mmol/L, 0.73 mmol/L], p = 0.002; LDL, low density lipoproteins (2.79 mmol/L ± 1.14 mmol/L vs. 2.33 mmol/L ± 0.85 mmol/L, p = 0.010); TG, triglyceride (1.60 mmol/L [IQR 1.03 mmol/L, 2.41 mmol/L] vs. 1.12 mmol/L [IQR 0.73 mmol/L, 1.63 mmol/L]), p = 0.001) current smoking and drinking (40.50% vs. 26.90%), p = 0.037; 40.50% vs. 26.90%, p = 0.035), and had lower baseline NIHSS scores 15[IQR 12.00, 20.00] vs. 19[IQR 16.00, 21.50] p = 0.001; better collateral circulation ASITN/SIR score 2.00[IQR 1.00, 3.00] vs. 1.00[IQR 0.00, 1.00], p < 0.001. The embolic-LVO group had more cases of atrial fibrillation (52.90% vs. 5.40%, p < 0.001) and heart valve disease (11.5% vs. 1.40%, p = 0.09); INR (1.07[IQR 1.01, 1.15] vs. 1.01 [IQR 0.94, 1.08] p < 0.001); PT, (12.40 [IQR 11.25, 14.20] vs. 11.55 [IQR 10.90, 12.40], p = 0.001); WBC, (9.53×109/L [IQR 7.02×109/L, 12.51×109/L] vs. 11.19×109/L [IQR 8.29×109/L, 13.57×109/L], p = 0.035); neutrophils, (7.49×109/L [IQR 5.56×109/L, 11.40×109/L] vs. 9.80×109/L[IQR 6.45×109/L, 11.1×109/L], p = 0.040); NLR (6.40 [IQR 3.80, 10.96] vs. 8.47 [IQR 5.07, 11.83], p = 0.037) and a lower proportion of ICAS history (3.80% vs. 10.80%, p = 0.003) when compared to the ICAS-LVO group.
The internal carotid artery siphon, and the trunks before bifurcation in the M1 segment of the middle cerebral artery (MCA), mid-lower segment of the basilar artery are the most common intracranial sites of stenosis [22]. In this study, there was a predominance of anterior circulation LVO (163, 70.6%) and ICAS (42, 25.74%). Of the 68 (29.4%) posterior circulation stroke patients, 32 (47.06%) had intracranial stenosis. Specific sites for stenosis among the 74 ICAS patients were: M1 segments of the MCA (34, 45.95%), communicating segment of the internal carotid artery (3, 4.04%), ophthalmic segment of internal carotid artery (5, 6.76%), middle and lower segments of basilar artery (19, 25.68%) and V4 segment of the vertebral artery (13, 17.57%).
After excluding factors with collinearity or clinical relations with others, there were five more variables in the prediction model: fluctuating symptoms, NIHSS < 16 (cut-off value, 16), AF, tapered, ASITN/SIR score ≥ 2 (cut-off value, 2). The model depicted acceptable calibration (Hosmer-Lemeshow test, p = 0.451) and good discrimination (AUC, 0.941; 95% CI, 0.910–0.971). The β‑coefficients of the five predictors are shown in Table 3. The optimal cut-off value for the ICAS-LVO scale was 2 points with 86.5% sensitivity, 91.1% specificity, and 90.5% accuracy (Fig. 2). ICAS-LVO scale was developed as a nomogram (Fig. 4).
Validation Group
A total of 70 patients were included in the final analysis, ICAS-LVO (19, 27.14%) the discrimination ability was still promising with an AUC value of 0.897 and a good predictive performance (Fig. 3; Table 4).
Discussion
Early and accurate diagnosis of ICAS-LVO before interventional surgery is particularly important to help select the most appropriate device [23]. It is well known that preoperative etiology is relatively difficult to predict. In particular, vascular occlusion of intracranial segments. Tandem occlusion due to atherosclerotic stenosis of extracranial arteries or a carotid dissection is easily recognized early based on anatomical structures and unique imaging [24,25,26]. Currently, some predictors have been shown to distinguish between ICAS-LVO and embolism LVO with acute ischemic stroke patients. The high-resolution vessel wall magnetic resonance imaging (MRI) has limited applications in assessment of LVO type due to delays in treatment [27,28,29]. Some of the predictive studies were based on intraprocedural angiographic signs (IPASs) [19]. Although microcatheter “first-pass effects” exhibit reliable outcomes in identification of ICAS, they need a microcatheter through the area of total occlusion, which seems lacking in predicting models [30].
Tapered sign is defined as the appearance of a tapered beak-like or flame-like sign on DSA imaging [31,32,33]. Tapered signs are vital for identifying ICAS before an operation, but also present in occlusions due to arterial dissection [34]. Tapered signs in the petrocavernous segment of ICA or the origin of basilar artery are poorly predictive of ICAS-LVO [20]. ICAS-LVO patients exhibit a unique clinical history, including frequently present progressive or fluctuating symptoms [18], better collateral circulation [23], hypertension, diabetes, smoking [22], lower admission NIHSS score [35] and are younger than embolic-LVO group. A pre-EVT in situ atherosclerotic thrombosis (ISAT) predictive model formulated by Xing Jin et al. consists of three predictive factors: history of hypertension, atrial fibrillation rhythm, and dichotomous serum glucose levels. The ISAT scale is only applicable to patients with acute vertebrobasilar arterial occlusion [36]. Compared with previous studies, our study focused on the preoperative judgment of ICAS-LVO. The higher predictive value of the model helps in the accurate identification of ICAS in intracranial occluded segments, ease to implement and promotion.
Collateral circulation plays an important role in preserving perfusion and stabilizing cerebral blood flow in acute occlusion [37]. Chronic atherosclerotic intracranial arterial stenosis may lead to a compensatory adjustment in the brain. Collateral circulation has been shown to be better in patients with ICAS-LVO than in those without chronic stenosis, implying that good collateral circulation is a predictor of ICAS-LVO [37,38,39]. There is a significant association between angiographic collateral scores and baseline NIHSS scores [40].
We included variables that were associated with ICAS-LVO as predictors into the analysis. Binary logistic stepwise regression analysis was performed to evaluate the predictors and generate the ICAS-LVO scale, which had 5 predictors; fluctuating symptoms, NIHSS < 16, AF, tapered sign, and ASITN/SIR score ≥ 2. These 5 predictors are easily available clinically. This scale was shown to have the ability to identify the pathogenesis of intracranial vascular occlusion before interventional therapy and to enhance the identification of acute cerebral infarction due to ICAS-LVO. It is important for neurointerventionalists to perform optimal revascularization strategies to reduce the difficulty of surgery and shorten the vascular recanalization time, although, the preferred initial treatment during interventional therapy for ICAS-LVO patients has yet to be established; however, the identification of ICAS-LVO before EVT may improve recanalization rates with more targeted revascularization techniques and shorten recanalization times with early remedial measures [20, 36, 41]. The primary goal of recanalization is to rapidly open the occluded artery and remove the clot. The structure of intracranial atherosclerosis occlusive lesions is different from cardiogenic emboli [23]. Aspiration catheter recanalization for recanalization of ICAS-LVO exhibited poor outcomes [42, 43]. Stent retrievers as a first-line device may achieve a higher success rate than suction catheters in patients with ICAS-LVO [41]; however, repeated stent retrievers thrombectomy is associated with the possibility of intima injuries around the ICAS, which activates the platelets, easily resulting in occlusion again [44]. To eliminate potential stenosis and prevent re-occlusion, nearly half of ICAS-LVO patients require angioplasty with or without stent implement rescue treatment. Rescue treatment is a complex and time-consuming process that requires individualized treatment strategies [45,46,47].
Limitations
There are several limitations in this study. First, as a retrospective observational study conducted in a single center, there may be information bias. Second, the clinical prediction models drawn from this study are limited by the relatively small sample size. Our findings should be verified via further external validation and their generalizability evaluated using large sample, multicenter datasets.
Conclusion
The predictive scale comprising of fluctuating symptoms, NIHSS < 16, atrial fibrillation, tapered sign, and ASITN/SIR score ≥ 2 has a promising predictive value for ICAS-LVO before EVT in ischemic stroke due to acute large-vessel occlusion patients.
References
Berkhemer OA, Fransen PS, Beumer D, van den Berg LA, Lingsma HF, Yoo AJ, Schonewille WJ, Vos JA, Nederkoorn PJ, Wermer MJ, van Walderveen MA, Staals J, Hofmeijer J, van Oostayen JA, Lycklama à Nijeholt GJ, Boiten J, Brouwer PA, Emmer BJ, de Bruijn SF, van Dijk LC, Kappelle LJ, Lo RH, van Dijk EJ, de Vries J, de Kort PL, van Rooij WJ, van den Berg JS, van Hasselt BA, Aerden LA, Dallinga RJ, Visser MC, Bot JC, Vroomen PC, Eshghi O, Schreuder TH, Heijboer RJ, Keizer K, Tielbeek AV, den Hertog HM, Gerrits DG, van den Berg-Vos RM, Karas GB, Steyerberg EW, Flach HZ, Marquering HA, Sprengers ME, Jenniskens SF, Beenen LF, van den Berg R, Koudstaal PJ, van Zwam WH, Roos YB, van der Lugt A, van Oostenbrugge RJ, Majoie CB, Dippel DW, MR CLEAN Investigators. A randomized trial of intraarterial treatment for acute ischemic stroke. N Engl J Med. 2015;372:11–20. Erratum in: N Engl J Med. 2015;372:394.
Goyal M, Demchuk AM, Menon BK, Eesa M, Rempel JL, Thornton J, Roy D, Jovin TG, Willinsky RA, Sapkota BL, Dowlatshahi D, Frei DF, Kamal NR, Montanera WJ, Poppe AY, Ryckborst KJ, Silver FL, Shuaib A, Tampieri D, Williams D, Bang OY, Baxter BW, Burns PA, Choe H, Heo JH, Holmstedt CA, Jankowitz B, Kelly M, Linares G, Mandzia JL, Shankar J, Sohn SI, Swartz RH, Barber PA, Coutts SB, Smith EE, Morrish WF, Weill A, Subramaniam S, Mitha AP, Wong JH, Lowerison MW, Sajobi TT, Hill MD, ESCAPE Trial Investigators. Randomized assessment of rapid endovascular treatment of ischemic stroke. N Engl J Med. 2015;372:1019–30.
Saver JL, Goyal M, Bonafe A, Diener HC, Levy EI, Pereira VM, Albers GW, Cognard C, Cohen DJ, Hacke W, Jansen O, Jovin TG, Mattle HP, Nogueira RG, Siddiqui AH, Yavagal DR, Baxter BW, Devlin TG, Lopes DK, Reddy VK, du Mesnil de Rochemont R, Singer OC, Jahan R, SWIFT PRIME Investigators. Stent-retriever thrombectomy after intravenous t-PA vs. t-PA alone in stroke. N Engl J Med. 2015;372:2285–95.
Campbell BC, Mitchell PJ, Kleinig TJ, Dewey HM, Churilov L, Yassi N, Yan B, Dowling RJ, Parsons MW, Oxley TJ, Wu TY, Brooks M, Simpson MA, Miteff F, Levi CR, Krause M, Harrington TJ, Faulder KC, Steinfort BS, Priglinger M, Ang T, Scroop R, Barber PA, McGuinness B, Wijeratne T, Phan TG, Chong W, Chandra RV, Bladin CF, Badve M, Rice H, de Villiers L, Ma H, Desmond PM, Donnan GA, Davis SM, EXTEND-IA Investigators. Endovascular therapy for ischemic stroke with perfusion-imaging selection. N Engl J Med. 2015;372:1009–18.
Jovin TG, Chamorro A, Cobo E, de Miquel MA, Molina CA, Rovira A, San Román L, Serena J, Abilleira S, Ribó M, Millán M, Urra X, Cardona P, López-Cancio E, Tomasello A, Castaño C, Blasco J, Aja L, Dorado L, Quesada H, Rubiera M, Hernandez-Pérez M, Goyal M, Demchuk AM, von Kummer R, Gallofré M, Dávalos A, REVASCAT Trial Investigators. Thrombectomy within 8 hours after symptom onset in ischemic stroke. N Engl J Med. 2015;372:2296–306.
van Houwelingen RC, Luijckx GJ, Uyttenboogaart M. Intra-arterial treatment for basilar artery occlusion-reply. JAMA Neurol. 2017;74:130–1.
Kayan Y, Meyers PM, Prestigiacomo CJ, Kan P, Fraser JF, Society of NeuroInterventional Surgery. Current endovascular strategies for posterior circulation large vessel occlusion stroke: report of the Society of NeuroInterventional Surgery Standards and Guidelines Committee. J Neurointerv Surg. 2019;11:1055–62.
Writing Group for the BASILAR Group, Zi W, Qiu Z, Wu D, Li F, Liu H, Liu W, Huang W, Shi Z, Bai Y, Liu Z, Wang L, Yang S, Pu J, Wen C, Wang S, Zhu Q, Chen W, Yin C, Lin M, Qi L, Zhong Y, Wang Z, Wu W, Chen H, Yao X, Xiong F, Zeng G, Zhou Z, Wu Z, Wan Y, Peng H, Li B, Hu X, Wen H, Zhong W, Wang L, Jin P, Guo F, Han J, Fu X, Ai Z, Tian X, Feng X, Sun B, Huang Z, Li W, Zhou P, Tu M, Sun X, Li H, He W, Qiu T, Yuan Z, Yue C, Yang J, Luo W, Gong Z, Shuai J, Nogueira RG, Yang Q. Assessment of endovascular treatment for acute basilar artery occlusion via a nationwide prospective registry. JAMA Neurol. 2020;77:561–73.
Goyal M, Menon BK, van Zwam WH, Dippel DW, Mitchell PJ, Demchuk AM, Dávalos A, Majoie CB, van der Lugt A, de Miquel MA, Donnan GA, Roos YB, Bonafe A, Jahan R, Diener HC, van den Berg LA, Levy EI, Berkhemer OA, Pereira VM, Rempel J, Millán M, Davis SM, Roy D, Thornton J, Román LS, Ribó M, Beumer D, Stouch B, Brown S, Campbell BC, van Oostenbrugge RJ, Saver JL, Hill MD, Jovin TG, HERMES collaborators. Endovascular thrombectomy after large-vessel ischaemic stroke: a meta-analysis of individual patient data from five randomised trials. Lancet. 2016;387:1723–31.
Lapergue B, Blanc R, Gory B, Labreuche J, Duhamel A, Marnat G, Saleme S, Costalat V, Bracard S, Desal H, Mazighi M, Consoli A, Piotin M, ASTER Trial Investigators. Effect of endovascular contact aspiration vs stent retriever on revascularization in patients with acute Ischemic stroke and large vessel occlusion: the ASTER randomized clinical trial. JAMA. 2017;318:443–52.
Bang OY, Kim BM, Seo WK, Jeon P. Endovascular therapy for acute ischemic stroke of intracranial atherosclerotic origin-neuroimaging perspectives. Front Neurol. 2019;10:269.
Tsang COA, Cheung IHW, Lau KK, Brinjikji W, Kallmes DF, Krings T. Outcomes of stent retriever versus aspiration-first thrombectomy in Ischemic stroke: a systematic review and meta-analysis. Ajnr Am J Neuroradiol. 2018;39:2070–6.
Tsang ACO, Orru E, Klostranec JM, Yang IH, Lau KK, Tsang FCP, Lui WM, Pereira VM, Krings T. Thrombectomy outcomes of intracranial atherosclerosis-related occlusions. Stroke. 2019;50:1460–6.
Kang DH, Yoon W, Kim SK, Baek BH, Lee YY, Kim YW, Kim YS, Hwang YH, Kim JT, Park MS. Endovascular treatment for emergent large vessel occlusion due to severe intracranial atherosclerotic stenosis. J Neurosurg. 2018; https://doi.org/10.3171/2018.1.JNS172350.
Gorelick PB, Wong KS, Bae HJ, Pandey DK. Large artery intracranial occlusive disease: a large worldwide burden but a relatively neglected frontier. Stroke. 2008;39:2396–9.
Marto JP, Strambo D, Hajdu SD, Eskandari A, Nannoni S, Sirimarco G, Bartolini B, Puccinelli F, Maeder P, Saliou G, Michel P. Twenty-four-hour reocclusion after successful mechanical thrombectomy: associated factors and long-term prognosis. Stroke. 2019;50:2960–3.
Mosimann PJ, Kaesmacher J, Gautschi D, Bellwald S, Panos L, Piechowiak E, Dobrocky T, Zibold F, Mordasini P, El-Koussy M, Wiest R, Bervini D, Wagner F, Arnold M, Jung S, Galimanis A, Gralla J, Fischer U. Predictors of Unexpected Early Reocclusion After Successful Mechanical Thrombectomy in Acute Ischemic Stroke Patients. Stroke. 2018;49:2643–51. Erratum in: Stroke. 2018;49:e343.
Adams HP Jr, Bendixen BH, Kappelle LJ, Biller J, Love BB, Gordon DL, Marsh EE 3rd. Classification of subtype of acute ischemic stroke. Definitions for use in a multicenter clinical trial. TOAST. Trial of Org 10172 in Acute Stroke Treatment. Stroke. 1993;24:35–41.
Liang W, Wang Y, Du Z, Mang J, Wang J. Intraprocedural angiographic signs observed during endovascular thrombectomy in patients with acute Ischemic stroke: a systematic review. Neurology. 2021;96:1080–90.
Jin X, Shi F, Chen Y, Zheng X, Zhang J. Jet-like appearance in angiography as a predictive image marker for the occlusion of intracranial atherosclerotic stenosis. Front Neurol. 2020;11:575567.
Hassen BW, Malley C, Boulouis G, Clarençon F, Bartolini B, Bourcier R, Rodriguez Régent C, Bricout N, Labeyrie MA, Gentric JC, Rouchaud A, Soize S, Saleme S, Raoult H, Gallas S, Eugène F, Anxionnat R, Herbreteau D, Bracard S, Naggara O. Inter- and intraobserver reliability for angiographic leptomeningeal collateral flow assessment by the American Society of Interventional and Therapeutic Neuroradiology/Society of Interventional Radiology (ASITN/SIR) scale. J Neurointerv Surg. 2019;11:338–41.
Holmstedt CA, Turan TN, Chimowitz MI. Atherosclerotic intracranial arterial stenosis: risk factors, diagnosis, and treatment. Lancet Neurol. 2013;12:1106–14.
Kuribara T, Sato H, Iihoshi S, Tsukagoshi E, Teranishi A, Kinoshita Y, Sugasawa S, Kohyama S, Takahashi S, Kurita H. Preprocedural prediction of underlying atherosclerotic lesions in cerebral large-vessel occlusions: clinical backgrounds, radiological findings, and treatment outcomes. J Atheroscler Thromb. 2022;29:1613–24.
Provenzale JM, Sarikaya B. Comparison of test performance characteristics of MRI, MR angiography, and CT angiography in the diagnosis of carotid and vertebral artery dissection: a review of the medical literature. AJR Am J Roentgenol. 2009;193:1167–74.
Bonati LH, Jansen O, de Borst GJ, Brown MM. Management of atherosclerotic extracranial carotid artery stenosis. Lancet Neurol. 2022;21:273–83.
Poppe AY, Jacquin G, Roy D, Stapf C, Derex L. Tandem carotid lesions in acute Ischemic stroke: mechanisms, therapeutic challenges, and future directions. AJNR Am J Neuroradiol. 2020;41:1142–8.
Natori T, Sasaki M, Miyoshi M, Ohba H, Katsura N, Yamaguchi M, Narumi S, Kabasawa H, Kudo K, Ito K, Terayama Y. Evaluating middle cerebral artery atherosclerotic lesions in acute ischemic stroke using magnetic resonance T1-weighted 3-dimensional vessel wall imaging. J Stroke Cerebrovasc Dis. 2014;23:706–11.
Teng Z, Peng W, Zhan Q, Zhang X, Liu Q, Chen S, Tian X, Chen L, Brown AJ, Graves MJ, Gillard JH, Lu J. An assessment on the incremental value of high-resolution magnetic resonance imaging to identify culprit plaques in atherosclerotic disease of the middle cerebral artery. Eur Radiol. 2016;26:2206–14.
Zhai SJ, Jia L, Kukun HJ, Wang YL, Wang H, Ding S, Jia WX. Predictive power of high-resolution vessel wall magnetic resonance imaging in ischemic stroke. Am J Transl Res. 2022;14:664–71.
Yi TY, Chen WH, Wu YM, Zhang MF, Zhan AL, Chen YH, Wu ZZ, Shi YC, Chen BL. Microcatheter “first-pass effect” predicts acute Intracranial artery atherosclerotic disease-related occlusion. Neurosurgery. 2019;84:1296–305.
Pillai JJ, Lanzieri CF, Trinidad SB, Tarr RW, Sunshine JL, Lewin JS. Initial angiographic appearance of intracranial vascular occlusions in acute stroke as a predictor of outcome of thrombolysis: initial experience. Radiology. 2001;218:733–8.
Garcia-Bermejo P, Patro SN, Ahmed AZ, Al Rumaihi G, Akhtar N, Kamran S, Salam A, Own A, Saqqur M, Shuaib A. Baseline occlusion angiographic appearance on mechanical thrombectomy suggests underlying etiology and outcome. Front Neurol. 2019;10:499.
Mönch S, Boeckh-Behrens T, Berndt M, Maegerlein C, Wunderlich S, Zimmer C, Friedrich B. Angiographic baseline proximal thrombus appearance of M1/M2 occlusions in mechanical thrombectomy. Clin Neuroradiol. 2021;31:189–96.
Rodallec MH, Marteau V, Gerber S, Desmottes L, Zins M. Craniocervical arterial dissection: spectrum of imaging findings and differential diagnosis. Radiographics. 2008;28:1711–28.
Kim SJ, Ryoo S, Kim GM, Chung CS, Lee KH, Bang OY. Clinical and radiological outcomes after intracranial atherosclerotic stroke: a comprehensive approach comparing stroke subtypes. Cerebrovasc Dis. 2011;31:427–34.
Zha M, Wu M, Huang X, Zhang X, Huang K, Yang Q, Cai H, Ji Y, Lv Q, Yang D, Dai Q, Liu R, Liu X. A pre-Interventional scale to predict in situ atherosclerotic thrombosis in acute vertebrobasilar artery occlusion patients. Front Neurol. 2021;12:648081.
Lau AY, Wong EH, Wong A, Mok VC, Leung TW, Wong KS. Significance of good collateral compensation in symptomatic intracranial atherosclerosis. Cerebrovasc Dis. 2012;33:517–24.
Lan L, Leng X, Ip V, Soo Y, Abrigo J, Liu H, Fan F, Ma SH, Ma K, Ip BY, Chan KL, Mok VC, Liebeskind DS, Wong KS, Leung TW. Sustaining cerebral perfusion in intracranial atherosclerotic stenosis: The roles of antegrade residual flow and leptomeningeal collateral flow. J Cereb Blood Flow Metab. 2020;40:126–34.
Kim HJ, Lee SJ, Jung KO. Association of concomitant Intracranial atherosclerotic stenosis with collateral status in patients with emergent large vessel occlusion. World Neurosurg. 2019;128:e1102–e8.
Marks MP, Lansberg MG, Mlynash M, Olivot JM, Straka M, Kemp S, McTaggart R, Inoue M, Zaharchuk G, Bammer R, Albers GW. Diffusion and perfusion imaging evaluation for understanding stroke evolution 2 investigators. Effect of collateral blood flow on patients undergoing endovascular therapy for acute ischemic stroke. Stroke. 2014;45:1035–9.
Yoo J, Lee SJ, Hong JH, Kim YW, Hong JM, Kim CH, Kang DH, Choi JW, Kim YS, Sohn SI, Hwang YH, Lee JS. Immediate effects of first-line thrombectomy devices for intracranial atherosclerosis-related occlusion: stent retriever versus contact aspiration. BMC Neurol. 2020;20:283.
Liao G, Zhang Z, Zhang G, Du W, Li C, Liang H. Efficacy of a direct aspiration first-pass technique (ADAPT) for endovascular treatment in different etiologies of large vessel occlusion: embolism vs. intracranial atherosclerotic stenosis. Front Neurol. 2021;12:695085.
Park H, Baek JH, Kim BM. Endovascular treatment of acute stroke due to Intracranial atherosclerotic stenosis-related large vessel occlusion. Front Neurol. 2019;10:308.
Gascou G, Lobotesis K, Machi P, Maldonado I, Vendrell JF, Riquelme C, Eker O, Mercier G, Mourand I, Arquizan C, Bonafé A, Costalat V. Stent retrievers in acute ischemic stroke: complications and failures during the perioperative period. AJNR Am J Neuroradiol. 2014;35:734–40.
Yoon W, Kim SK, Park MS, Kim BC, Kang HK. Endovascular treatment and the outcomes of atherosclerotic intracranial stenosis in patients with hyperacute stroke. Neurosurgery. 2015;76:680–6. discussion 686.
Al Kasab S, Almadidy Z, Spiotta AM, Turk AS, Chaudry MI, Hungerford JP, Turner RD 4th. Endovascular treatment for AIS with underlying ICAD. J Neurointerv Surg. 2017;9:948–51.
Chuming H, Yifan H, Ke X, Chukai X, Weijie Z, Hui L, Guoyi P, Xiaoyong Z, Peifeng Z, Chuwei C. Association Between Occlusion Type and Etiology of Acute Intracranial Large Artery Occlusion. Front Neurol. 2020;11:582388.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Y. Cai, Y. Gu, Y. Wang, P. Wang, L. Zhang, C. Liu, J. Chu, H. Li, Z. Lu, Y. Zhou and H. Liu declare that they have no competing interests.
Ethical standards
This retrospective, observational study was approved by the Ethics Committee of No. 1 People’s Hospital of Jining. Number:2021 the Ethics Committee of the No.1 People’s Hospital of Jining (107). The study was performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Cai, Y., Gu, Y., Wang, Y. et al. A Clinical Prediction Model for Patients with Acute Large Vessel Occlusion Due to Underlying Intracranial Atherosclerotic Stenosis. Clin Neuroradiol 33, 519–528 (2023). https://doi.org/10.1007/s00062-022-01241-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00062-022-01241-3