Abstract
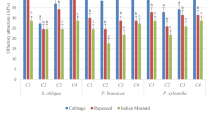
The importance of plant cuticular waxes in plant–herbivore interactions has become an area of increasing interest, but it has been poorly explored in chrysomelid beetles, one of the most important groups of insect pests. In this study, we investigated the effects of cuticular wax on feeding behaviors of Aulacophora femoralis (Chyrosomelidae), a pest of Cucurbitaceae crops. We analyzed chemical compositions of leaf cuticular waxes using gas chromatography–mass spectrometry. The compositions were significantly different among plant species; in particular, host (Cucumis maxima, C. pepo, C. sativus, Cucurbitaceae) and non-host (Pueraria lobata, Fabaceae) leaves had distinctive compositions of cuticular waxes. We examined if the cuticular wax induces feeding behaviors based on a bioassay using filter papers treated with cuticular wax extract from host or non-host leaves, or a synthetic mixture of straight-chain alkanes (n-alkanes), major components of the host cuticular waxes shared among different host species. We found that the beetles frequently left bite marks on the filter papers treated with n-alkanes but less frequently on those treated with cuticular wax extracts of host leaves. These results indicate that leaf cuticular wax n-alkanes stimulate feeding behavior, but are not cues for host selection.



Similar content being viewed by others
Data availability
Data supporting the findings of this study are available from the corresponding author I.K. on request.
References
Abe M, Matsuda K (2005) Chemical factors influencing the feeding preference of three Aulacophora leaf beetle species (Coleoptera: Chrysomelidae). Appl Entomol Zool 40:161–168. https://doi.org/10.1303/aez.2005.161
Adati T, Matsuda K (1993) Feeding stimulants for various leaf beetles (Coleoptera: Chrysomelidae) in the leaf surfae wax of their host plants. Appl Entomol Zool 28:319–324
Adati T, Matsuda K (2000) The effect of leaf surface wax on feeding of the strawberry leaf beetle, Galerucella vittaticollis, with reference to host plant preference. Tohoku J Agric Res 50:57–61
Agnihotri AR, Roy AA, Joshi RS (2016) Gustatory receptors in Lepidoptera: chemosensation and beyond. Insect Mol Biol 25:519–529. https://doi.org/10.1111/imb.12246
Arya GC, Sarkar S, Manasherova E et al (2021) The plant cuticle: an ancient guardian barrier set against long-standing rivals. Front Plant Sci. https://doi.org/10.3389/fpls.2021.663165
Bates D, Mächler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme. J Stat Softw 67:1–48
Bernaola L, Butterfield TS, Tai TH, Stout MJ (2021) Epicuticular wax rice mutants show reduced resistance to rice water weevil (Coleoptera: Curculionidae) and fall armyworm (Lepidoptera: Noctuidae). Environ Entomol 50:948–957. https://doi.org/10.1093/ee/nvab038
Buschhaus C, Jetter R (2011) Composition differences between epicuticular and intracuticular wax substructures: How do plants seal their epidermal surfaces? J Exp Bot 62:841–853. https://doi.org/10.1093/jxb/erq366
Bush RT, McInerney FA (2013) Leaf wax n-alkane distributions in and across modern plants: Implications for paleoecology and chemotaxonomy. Geochim Cosmochim Acta 117:161–179. https://doi.org/10.1016/j.gca.2013.04.016
Das S, Koner A, Barik A (2019) A beetle biocontrol agent of rice-field weeds recognizes its host plants by surface wax long-chain alkanes and free fatty acids. Chemoecology 29:155–170. https://doi.org/10.1007/s00049-019-00285-1
de Brito Sanchez MG (2011) Taste perception in honey bees. Chem Senses 36:675–692. https://doi.org/10.1093/chemse/bjr040
Diefendorf AF, Leslie AB, Wing SL (2015) Leaf wax composition and carbon isotopes vary among major conifer groups. Geochim Cosmochim Acta 170:145–156. https://doi.org/10.1016/j.gca.2015.08.018
Dodd RS, Afzal-Rafii Z (2000) Habitat-related adaptive properties of plant cuticular lipids. Evolution 54:1438. https://doi.org/10.1554/0014-3820(2000)054[1438:hrapop]2.0.co;2
Dodd RS, Poveda MM (2003) Environmental gradients and population divergence contribute to variation in cuticular wax composition in Juniperus communis. Biochem Syst Ecol 31:1257–1270. https://doi.org/10.1016/S0305-1978(03)00031-0
Dominguez E, Heredia-Guerrero JA, Heredia A (2011) The biophysical design of plant cuticles: an overview. New Phytol 189:938–949. https://doi.org/10.1111/j.1469-8137.2010.03553.x
Eigenbrode SD, Espelie KE (1995) Effects of plant epicuticular lipids on insect herbivores. Annu Rev Entomol 40:171–194. https://doi.org/10.1146/annurev.en.40.010195.001131
Gorski P, Jaworski A, Shannon S, Robinson R (1985) Rapid TLC and HPLC test for cucurbitacins. Cucurbit Genet Coop 25:1034–1026
Hauke V, Schreiber L (1998) Ontogenetic and seasonal development of wax composition and cuticular transpiration of ivy (Hedera helix L.) sun and shade leaves. Planta 207:67–75. https://doi.org/10.1007/s004250050456
Heisswolf A, Gabler D, Obermaier E, Müller C (2007) Olfactory versus contact cues in host plant recognition of a monophagous chrysomelid beetle. J Insect Behav 20:247–266. https://doi.org/10.1007/s10905-007-9078-z
Holm S (1979) A simple sequentially rejective multiple test procedure. Scand J Stat 6:65–70
Jetter R, Riederer M (2016) Localization of the transpiration barrier in the epi- and intracuticular waxes of eight plant species: Water transport resistances are associated with fatty acyl rather than alicyclic components. Plant Physiol 170:921–934. https://doi.org/10.1104/pp.15.01699
Lin S, Binder BF, Hart ER (1998) Insect feeding stimulants from the leaf surface of Populus. J Chem Ecol 24:1781–1790. https://doi.org/10.1023/A:1022351415467
Maffei M, Badino S, Bossi S (2004) Chemotaxonomic significance of leaf wax n-alkanes in the Pinales (Coniferales). J Biol Res 1:3–19
Müller C, Hilker M (2001) Host finding and oviposition behavior in a chrysomelid specialist - The importance of host plant surface waxes. J Chem Ecol 27:985–994. https://doi.org/10.1023/A:1010343205114
Müller C, Riederer M (2005) Plant surface properties in chemical ecology. J Chem Ecol 31:2621–2651. https://doi.org/10.1007/s10886-005-7617-7
Naß R, Markstädter C, Hauke V, Riederer M (1998) Quantitative gas chromatographic analysis of plant cuticular waxes containing long-chain aldehydes. Phytochem Anal 9:112–118
Oksanen J, Blanchet F, Kindt R, et al (2020) vegan: Community Ecology Package. https://CRAN.R-project.org/package=vegan
Piasentier E, Bovolenta S, Malossini F (2000) The n-alkane concentrations in buds and leaves of browsed broadleaf trees. J Agric Sci 135:311–320. https://doi.org/10.1017/S0021859699008230
R Core Team (2020). R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL https://www.R-project.org/
Roy N (2021) Synergism in host selection behavior of three generalist insects towards leaf cuticular wax of sesame cultivars. Neotrop Entomol 50:812–827. https://doi.org/10.1007/s13744-021-00892-0
Stocker RF (1994) The organization of the chemosensory system in Drosophila melanogaster: a rewiew. Cell Tissue Res 275:3–26. https://doi.org/10.1007/BF00305372
Tanabe C, Mishima R (1930) On the life-history and methods of control of Rhaphidopalpa femoralis Motsch. J Plant Prot 17:378–383
Yosano S, Kutsuwada Y, Akatsu M et al (2020) Taste recognition through tarsal gustatory sensilla potentially important for host selection in leaf beetles (Coleoptera: Chrysomelidae). Sci Rep 10:1–11. https://doi.org/10.1038/s41598-020-61935-x
Zhu X, Zhang Y, Du Z et al (2018) Tender leaf and fully-expanded leaf exhibited distinct cuticle structure and wax lipid composition in Camellia sinensis cv Fuyun 6. Sci Rep 8:1–12. https://doi.org/10.1038/s41598-018-33344-8
Acknowledgements
The authors are grateful to Akira Matumoto, Satomi Yoshinami for assisting with growing plants in the experimental field at CER; and Yuka Yahiro, Ayao Hori and Kaori Shiojiri for providing insects for the experiments. This study was financially supported by JSPS KAKENHI, grant nos. 16H04830 and 20H03324.
Funding
This work was supported by JSPS KAKENHI to Shoko Sakai (grant nos. 16H04830, 16K07516 and 20H03324).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Data collection of the cuticular waxes was performed by IK and YI. Feeding experiments were done by IK. The first draft of the manuscript was written by IK and SS. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Communicated by Günther Raspotnig.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kamata, I., Inui, Y. & Sakai, S. Straight-chain alkanes derived from leaf waxes stimulate feeding behaviors in a cucurbit leaf beetle, Aulacophora femolaris (Chrysomelidae, Coleoptera). Chemoecology 33, 55–61 (2023). https://doi.org/10.1007/s00049-023-00384-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00049-023-00384-0