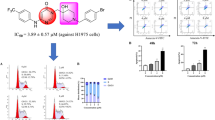
Abstract
A series of novel 5-cyano-2,4,6-substituted pyrimidine derivatives containing acrylamide group were designed, synthesized, and evaluated for their antitumor activity against four human cancer cell lines (MGC-803, PC-3, A549, and H1975) using the MTT assay. Among them, compound 20y exhibited the most potent cytotoxicity against PC-3 cells (IC50 = 2.75 ± 0.08 μM). Notably, compound 20y significantly inhibited the colony formation, migration, and invasion of PC-3 cells. Furthermore, compound 20y induced S-phase cell cycle arrest and apoptosis in PC-3 cells. These findings indicate that compound 20y might serve as a valuable lead compound for developing antitumor agents targeting prostate cancer cells.
Graphical Abstract







Similar content being viewed by others
References
Shi Z-D, Pang K, Wu Z-X, Dong Y, Hao L, Qin J-X, et al. Tumor cell plasticity in targeted therapy-induced resistance: mechanisms and new strategies. Signal Transduct Target Ther. 2023;8:113. https://doi.org/10.1038/s41392-023-01383-x
Zhang L, Ye B, Chen Z, Chen Z-S. Progress in the studies on the molecular mechanisms associated with multidrug resistance in cancers. Acta Pharm Sin B. 2023;13:982–97. https://doi.org/10.1016/j.apsb.2022.10.002
Marinescu M. Biginelli reaction mediated synthesis of antimicrobial pyrimidine derivatives and their therapeutic properties. Molecules. 2021;26:6022 https://doi.org/10.3390/molecules26196022
Kappe CO. Recent advances in the Biginelli dihydropyrimidine synthesis. New tricks from an old dog. Acc Chem Res. 2000;33:879–88. https://doi.org/10.1021/ar000048h
Tan L, Wu C, Zhang J, Yu Q, Wang X, Zhang L, et al. Design, synthesis, and biological evaluation of heterocyclic-fused pyrimidine chemotypes guided by x-ray crystal structure with potential antitumor and anti-multidrug resistance efficacy targeting the colchicine binding site. J Medicinal Chem. 2023;66:3588–620. https://doi.org/10.1021/acs.jmedchem.2c02115
Tian J, Zhang D, Kurbatov V, Wang Q, Wang Y, Fang D, et al. 5-Fluorouracil efficacy requires anti-tumor immunity triggered by cancer-cell-intrinsic STING. Embo J. 2021;40:e106065. https://doi.org/10.15252/embj.2020106065
Vala RM, Sharma MG, Patel DM, Puerta A, Padrón JM, Ramkumar V, et al. Synthesis and in vitro study of antiproliferative benzyloxy dihydropyrimidinones. Arch Pharm. 2021;354:e2000466. https://doi.org/10.1002/ardp.202000466
Prasad T, Mahapatra A, Sharma T, Sahoo CR, Padhy RN. Dihydropyrimidinones as potent anticancer agents: Insight into the structure-activity relationship. Arch Pharm. 2023:e2200664. https://doi.org/10.1002/ardp.202200664.
Lachhab S, El Mansouri AE, Mehdi A, Dennemont I, Neyts J, Jochmans D, et al. Synthesis of new 3-acetyl-1,3,4-oxadiazolines combined with pyrimidines as antileishmanial and antiviral agents. Mol Divers. 2022:1–13. https://doi.org/10.1007/s11030-022-10548-9.
Rane JS, Pandey P, Chatterjee A, Khan R, Kumar A, Prakash A, et al. Targeting virus-host interaction by novel pyrimidine derivative: an in silico approach towards discovery of potential drug against COVID-19. J Biomol Struct Dyn. 2021;39:5768–78. https://doi.org/10.1080/07391102.2020.1794969
Sharma P, Rane N, Gurram VK. Synthesis and QSAR studies of pyrimido[4,5-d]pyrimidine-2,5-dione derivatives as potential antimicrobial agents. Bioorg Med Chem Lett. 2004;14:4185–90. https://doi.org/10.1016/j.bmcl.2004.06.014
Prakash O, Bhardwaj V, Kumar R, Tyagi P, Aneja KR. Organoiodine (III) mediated synthesis of 3-aryl/hetryl-5,7-dimethyl-1,2,4-triazolo[4,3-a]pyrimidines as antibacterial agents. Eur J Med Chem. 2004;39:1073–7. https://doi.org/10.1016/j.ejmech.2004.06.011
Sharma MG, Vala RM, Rajani DP, Ramkumar V, Gardas RL, Banerjee S, et al. Crystal structure, antibacterial and antifungal evaluation of 5-bromothiophene based 3,4-dihydropyrimidin-2-(1H)-(thi)ones. Phosphorus, Sulfur, Silicon Relat Elem. 2023;198:145–53. https://doi.org/10.1080/10426507.2022.2121397
Agarwal N, Raghuwanshi SK, Upadhyay DN, Shukla PK, Ram VJ. Suitably functionalised pyrimidines as potential antimycotic agents. Bioorg Med Chem Lett. 2000;10:703–6. https://doi.org/10.1016/s0960-894x(00)00091-3
Shoichet BK, Stroud RM, Santi DV, Kuntz ID, Perry KM. Structure-based discovery of inhibitors of thymidylate synthase. Science. 1993;259:1445–50. https://doi.org/10.1126/science.8451640
Alam O, Khan SA, Siddiqui N, Ahsan W, Verma SP, Gilani SJ. Antihypertensive activity of newer 1,4-dihydro-5-pyrimidine carboxamides: synthesis and pharmacological evaluation. Eur J Med Chem. 2010;45:5113–9. https://doi.org/10.1016/j.ejmech.2010.08.022
Kumar V, Mahdi F, Khanna AK, Singh R, Chander R, Saxena JK, et al. Antidyslipidemic and antioxidant activities of hibiscus rosa sinensis root extract in alloxan induced diabetic rats. Indian J Clin Biochem. 2013;28:46–50. https://doi.org/10.1007/s12291-012-0223-x
Spasov AA, Babkov DA, Sysoeva VA, Litvinov RA, Shamshina DD, Ulomsky EN, et al. 6-Nitroazolo[1,5-a]pyrimidin-7(4H)-ones as antidiabetic agents. Arch Pharm. 2017;350. https://doi.org/10.1002/ardp.201700226.
Rahaman SA, Pasad YR, Kumar P, Kumar B. Synthesis and anti-histaminic activity of some novel pyrimidines. Saudi Pharm J. 2009;17. https://doi.org/10.1016/j.jsps.2009.08.001.
Finger V, Kufa M, Soukup O, Castagnolo D, Roh J, Korabecny J. Pyrimidine derivatives with antitubercular activity. Eur J Med Chem. 2023;246:114946. https://doi.org/10.1016/j.ejmech.2022.114946
Xu C, Shao T, Shao S, Jin G. High activity, high selectivity and high biocompatibility BODIPY-pyrimidine derivatives for fluorescence target recognition and evaluation of inhibitory activity. Bioorg Chem. 2021;114:105121. https://doi.org/10.1016/j.bioorg.2021.105121
Guo W, Xing Y, Zhang Q, Xie J, Huang D, Gu H, et al. Synthesis and biological evaluation of B-cell lymphoma 6 inhibitors of N-phenyl-4-pyrimidinamine derivatives bearing potent activities against tumor growth. J Med Chem. 2020;63:676–95. https://doi.org/10.1021/acs.jmedchem.9b01618
Metterle L, Nelson C, Patel N. Intralesional 5-fluorouracil (FU) as a treatment for nonmelanoma skin cancer (NMSC): a review. J Am Acad Dermatol. 2016;74:552–7. https://doi.org/10.1016/j.jaad.2015.09.040
David C, Naureen S, Sheela R, Timothy I, Marianne N, Fareeda C, et al. Capecitabine and oxaliplatin for advanced esophagogastric cancer. N Engl J Med. 2008;358. https://doi.org/10.1056/nejmoa073149.
Valle J, Wasan H, Palmer DH, Cunningham D, Anthoney A, Maraveyas A, et al. Cisplatin plus gemcitabine versus gemcitabine for biliary tract cancer. N Engl J Med. 2010;362:1273–81. https://doi.org/10.1056/NEJMoa0908721
Kelly C, Bhuva N, Harrison M, Buckley A, Saunders M. Use of raltitrexed as an alternative to 5-fluorouracil and capecitabine in cancer patients with cardiac history. Eur J Cancer. 2013;49:2303–10. https://doi.org/10.1016/j.ejca.2013.03.004
Morihiro K, Ishinabe T, Takatsu M, Osumi H, Osawa T, Okamoto A. Floxuridine oligomers activated under hypoxic environment. J Am Chem Soc. 2021;143:3340–7. https://doi.org/10.1021/jacs.0c10732
Dhillon S. Decitabine/cedazuridine: first approval. Drugs. 2020;80:1373–8. https://doi.org/10.1007/s40265-020-01389-7
Honglin D, Chao G, Xiaojie S, Yutong Z, Zhengjie W, Limin L, et al. Design, synthesis, and antitumor activity evaluation of 2,4,6-trisubstituted quinazoline derivatives containing acrylamide. Russ J Bioorg Chem. 2022;48:1089–100. https://doi.org/10.1134/S1068162022050090
HongWei J, HuaLi Y, ZhiLing X, MingHui D, Tong W, Yang L, et al. Design, synthesis and antitumor activity evaluation of novel indole acrylamide derivatives as IMPDH inhibitors. Bioorgan Chem. 2022;129. https://doi.org/10.1016/j.bioorg.2022.106213.
Hu X, Tang S, Yang F, Zheng P, Xu S, Pan Q, et al. Design, synthesis, and antitumor activity of olmutinib derivatives containing acrylamide moiety. Molecules. 2021;26:3041. https://doi.org/10.3390/molecules26103041
Enzalutamide in Castration-Resistant Prostate Cancer. N. Engl J Med. 2018;379:1380-1. https://doi.org/10.1056/NEJMc1810065.
Apperley JF, Byrne JL, Smith G, Claudiani S, Viqueira A, Ferdinand R, et al. The real world use of bosutinib in patients with chronic myeloid leukaemia. Blood. 2016;128:5435. https://doi.org/10.1182/blood.V128.22.5435.5435
Pingxian L, Dongmei F, Wenliang Q, Xinlian H, Lidan Z, Yunhan J, et al. SAR study and molecular mechanism investigation of novel naphthoquinone-furan-2-cyanoacryloyl hybrids with antitumor activity. Pharmaceutics. 2022;14. https://doi.org/10.3390/pharmaceutics14102104.
Lian-Fang Y, Yajing X, Jie-Xin X, Jia X, Wei G, Jiuqing X, et al. Synthesis of cyanoenone-modified diterpenoid analogs as novel Bmi-1-mediated antitumor agents. ACS Med Chem Lett. 2018;9. https://doi.org/10.1021/acsmedchemlett.8b00345.
Ester K, Supek F, Majsec K, Marjanović M, Lembo D, Donalisio M, et al. Putative mechanisms of antitumor activity of cyano-substituted heteroaryles in HeLa cells. Investig N Drugs. 2012;30:450–67. https://doi.org/10.1007/s10637-010-9571-7
Wang H, Zhang L, Zheng X, Zhang X, Si X, Wang M. The ability of avitinib to penetrate the blood brain barrier and its control of intra-/extra- cranial disease in patients of non-small cell lung cancer (NSCLC) harboring EGFR T790M mutation. J Clin Oncol. 2017;35:e20613-e. https://doi.org/10.1200/JCO.2017.35.15_suppl.e20613
Elias R, Pudusseri A, Akinboro O, White PS, Sarosiek S. Ibrutinib: a post-marketing safety analysis. J Clin Oncol. 2018;36:e19559-e. https://doi.org/10.1200/JCO.2018.36.15_suppl.e19559
Fleisher TA. Apoptosis. Ann Allergy, Asthma Immunol. 1997;78:245–50. https://doi.org/10.1016/S1081-1206(10)63176-6
Li S, Wu B, Zheng X, Wang C, Zhao J, Sun H, et al. Synthesis and biological activity of imidazole group-substituted arylaminopyrimidines (IAAPs) as potent BTK inhibitors against B-cell lymphoma and AML. Bioorg Chem. 2021;106:104385. https://doi.org/10.1016/j.bioorg.2020.104385
Jadhav CK, Nipate AS, Chate AV, Songire VD, Patil AP, Gill CH. Efficient rapid access to Biginelli for the multicomponent synthesis of 1,2,3,4-tetrahydropyrimidines in room-temperature diisopropyl ethyl ammonium acetate. ACS Omega. 2019;4:22313–24. https://doi.org/10.1021/acsomega.9b02286
Mao R, Shao J, Zhu K, Zhang Y, Ding H, Zhang C, et al. Potent, selective, and cell active protein arginine methyltransferase 5 (PRMT5) inhibitor developed by structure-based virtual screening and hit optimization. J Med Chem. 2017;60:6289–304. https://doi.org/10.1021/acs.jmedchem.7b00587
Said MM, Taher AT, El-Nassan HB, El-Khouly EA. Synthesis of novel S-acyl and S-alkylpyrimidinone derivatives as potential cytotoxic agents. Res Chem Intermed. 2016;42:6643–62. https://doi.org/10.1007/s11164-016-2487-x
El-Hameed RHA, Fatahala SS, Sayed AI. Synthesis of some novel benzimidazole derivatives as anticancer agent and evaluation for CDK2 inhibition activity. Med Chem. 2022;18:238–48. https://doi.org/10.2174/1573406417666210304100830
Zhu Y, Zheng X, Wang C, Sun X, Sun H, Ma T, et al. Synthesis and biological activity of thieno[3,2-d]pyrimidines as potent JAK3 inhibitors for the treatment of idiopathic pulmonary fibrosis. Bioorg Med Chem. 2020;28:115254. https://doi.org/10.1016/j.bmc.2019.115254
Rodriguez F, Saffon N, Sammartino JC, Degiacomi G, Pasca MR, Lherbet C. First triclosan-based macrocyclic inhibitors of InhA enzyme. Bioorg Chem. 2020;95:103498. https://doi.org/10.1016/j.bioorg.2019.103498
Sun B, Liu X, Zheng X, Wang C, Meng Q, Sun H, et al. Novel pyrimidines as multitarget protein tyrosine kinase inhibitors for the treatment of idiopathic pulmonary fibrosis (IPF). ChemMedChem. 2020;15:182–7. https://doi.org/10.1002/cmdc.201900606.
Mologni L, Dalla Via M, Chilin A, Palumbo M, Marzaro G. Discovery of (wt) RET and (V804M) RET inhibitors: from hit to lead. ChemMedChem. 2017;12:1390–8. https://doi.org/10.1002/cmdc.201700243
Ge Y, Yang H, Wang C, Meng Q, Li L, Sun H, et al. Design and synthesis of phosphoryl-substituted diphenylpyrimidines (Pho-DPPYs) as potent Bruton’s tyrosine kinase (BTK) inhibitors: targeted treatment of B lymphoblastic leukemia cell lines. Bioorg Med Chem. 2017;25:765–72. https://doi.org/10.1016/j.bmc.2016.11.054
Song Z, Jin Y, Ge Y, Wang C, Zhang J, Tang Z, et al. Synthesis and biological evaluation of azole-diphenylpyrimidine derivatives (AzDPPYs) as potent T790M mutant form of epidermal growth factor receptor inhibitors. Bioorg Med Chem. 2016;24:5505–12. https://doi.org/10.1016/j.bmc.2016.09.001
Fargualy AM, Habib NS, Ismail KA, Hassan AM, Sarg MT. Synthesis, biological evaluation and molecular docking studies of some pyrimidine derivatives. Eur J Med Chem. 2013;66:276–95. https://doi.org/10.1016/j.ejmech.2013.05.028
Acknowledgements
This work was supported by the National Natural Science Foundation of China (No. U21A20416).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yu, F., Chi, L., Wang, H. et al. Design, synthesis and antitumor activity evaluation of 5-cyano-2,4,6-substituted pyrimidine derivatives containing acrylamide group. Med Chem Res 32, 2116–2124 (2023). https://doi.org/10.1007/s00044-023-03099-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-023-03099-7