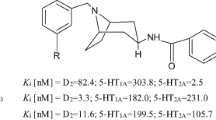
Abstract
Cocaine (1) use disorder (CUD) is a major, unmet medical need. This tropane alkaloid is one of the most commonly abused recreational drugs. In 2018, there were over 5.5 million cocaine users. Therapeutic options capable of addressing this issue are limited, as there are no approved pharmacotherapies. The D3 dopamine receptor is a potential CUD therapeutic target that remains under active investigation. We have recently disclosed a series of functionalized 5-(4-arylpiperazin-1-yl)-N-arylpentanamides typified by (8) that are potent D3 ligands that have moderate to high selectivity for D3 over D2. We have continued our exploration of this series of compounds in an effort to identify compounds with improved microsomal stability such as (12c).

Graphical Abstract





Similar content being viewed by others
References
Bose J, Hedden SL, Lipari RN, Park-Lee E, Key Substance Use and Mental Health Indicators in the United States: Results from the 2017 National Survey on Drug Use and Health, HHS Publication No. SMA 18-5068, NSDUH Series H-53, Rockville, MD: Center for Behavioral Health Statistics and Quality, Substance Abuse and Mental Health Services Administration, https://www.samhsa.gov/data/.
NIDA report Health Consequences of Drug Misuse Retrieved from https://www.drugabuse.gov/related-topics/health-consequences-drug-misuse 2019, April 2.
https://nida.nih.gov/drug-topics/trends-statistics/overdose-death-rates.
Balk JH. Dopamine signaling in reward-related behaviors. Front Neural Circuits. 2013;7:1–16. https://doi.org/10.3389/fncir.2013.00152.
Wise RA, Koob GF. The development and maintenance of drug addiction. Neuropsychopharmacology. 2014;39:254–62. https://doi.org/10.1038/npp.2013.261.
Alonso-Matias L, Reyes-Zamorano E, Gonzalez-Olvera JJ. Cognitive functions of subjects with cocaine and crack dependency disorder during early abstinence. Rev Neurol 2019;68:271–80. https://doi.org/10.33588/rn.6807.2018119.
Substance Abuse and Mental Health Services Administration, Drug Abuse Warning Network, 2011: National Estimates of Drug-Related Emergency Department Visits. HHS Publication No. (SMA) 13-4760, DAWN Series D-39. Rockville, MD: Substance Abuse and Mental Health Services Administration, 2013. https://www.samhsa.gov/data/sites/default/files/DAWN2k11ED/DAWN2k11ED/DAWN2k11ED.pdf.
Schierenberg A, van Amsterdam J, van den Brink W, Goudriaan AE. Efficacy of contingency management for cocaine dependence treatment: a review of the evidence. Curr Drug Abus Rev. 2012;5:320–31. https://doi.org/10.2174/1874473711205040006.
Penberthy JK, Ait-Daoud N, Vaughan M, Fanning T. Review of treatment for cocaine dependence. Curr Drug Abus Rev. 2010;3:49–62. https://doi.org/10.2174/1874473711003010049.
Johnson BA, Ait-Daoud N, Wang XQ, Penberthy JK, Javors MA, Seneviratne C, et al. Topiramate for the treatment of cocaine addiction: a randomized clinical trial. JAMA Psychiatry. 2013;70:338–1346. https://doi.org/10.1001/jamapsychiatry.2013.2295.
Somoza EC, Winship D, Gorodetzky CW, Lewis D, Ciraulo DA, Galloway GP, et al. A multisite, double-blind, placebo-controlled clinical trial to evaluate the safety and efficacy of vigabatrin for treating cocaine dependence. JAMA Psychiatry. 2013;70:630–7. https://doi.org/10.1001/jamapsychiatry.2013.872.
Somoza EC, Winship D, Gorodetzky CW, Lewis D, Ciraulo DA, Galloway GP, et al. Topiramate in the treatment of cocaine use disorder. Am J Health-Sys Pharm. 2018;75:e13–e22. https://doi.org/10.2146/ajhp160542.
Oliveto A, Poling J, Mancino MJ, Feldman Z, Cubells JF, Pruzinsky R, et al. Randomized, double-blind, placebo-controlled trial of disulfiram for the treatment of cocaine dependence in methadone-stabilized patients. Drug Alcohol Depend. 2011;113:184–91. https://doi.org/10.1016/j.drugalcdep.2010.07.022.
McCance-Katz EF, Kosten TR, Jatlow P. Disulfiram effects on acute cocaine administration. Drug Alcohol Depend. 1998;52:27–39. https://doi.org/10.1016/s0376-8716(98)00050-7.
Harvey-Lewis C, Li Z, Higgins GA, Fletcher PJ. The 5-HT2C receptor agonist lorcaserin reduces cocaine self-administration, reinstatement of cocaine-seeking and cocaine induced locomotor activity. Neuropharmacology. 2016;101:237–45. https://doi.org/10.1016/j.neuropharm.2015.09.028.
Baker DA, McFarland K, Lake RW, Shen H, Toda S, Kalivas PW. N‐Acetyl cysteine‐induced blockade of cocaine‐induced reinstatement. Ann N. Y Acad Sci. 2003;1003:349–51. https://doi.org/10.1196/annals.1300.023.
Placenza FM, Fletcher PJ, Vaccarino FJ, Erb S. Effects of central neurokinin-1 receptor antagonism on cocaine- and opiate-induced locomotor activity and self-administration behaviour in rats. Pharm Biochem Behav. 2006;84:94–101. https://doi.org/10.1016/j.pbb.2006.04.011.
Roeper J. Dissecting the diversity of midbrain dopamine neurons. Trends Neurosci. 2013;36:336–42. https://doi.org/10.1016/j.tins.2013.03.003.
Jaber M, Robinson SW, Missale C, Caron MG. Dopamine receptors and brain function. Neuropharmacology. 1996;35:1503–19. https://doi.org/10.1016/s0028-3908(96)00100-1.
Staley JK, Mash DC. Adaptive increase in D3 dopamine receptors in the brain reward circuits of human cocaine fatalities. J Neurosci. 1996;16:6100–6106. https://doi.org/10.1523/JNEUROSCI.16-19-06100.1996.
Segal DM, Moraes CT, Mash DC. Up-regulation of D3 dopamine receptor mRNA in the nucleus accumbens of human cocaine fatalities. Brain Res Mol Brain Res. 1997;45:335–339. https://doi.org/10.1016/s0169-328x(97)00025-9.
Mash DC, Staley JK. D3 dopamine and kappa opioid receptor alterations in human brain of cocaine-overdose victims. Ann N. Y Acad Sci. 1999;877:507–522. https://doi.org/10.1111/j.1749-6632.1999.tb09286.x.
Keck TM, John WS, Czoty PW, Nader MA, Newman AH. Identifying medication targets for psychostimulant addiction: unraveling the dopamine D3 receptor hypothesis. J Med Chem. 2015;58:5361–5380. https://doi.org/10.1021/jm501512b.
Newman AH, Blaylock BL, Nader MA, Bergman J, Sibley DR, Skolnick P. Medication discovery for addiction: translating the dopamine D3 receptor hypothesis. Biochem Pharm. 2012;84:882–90. https://doi.org/10.1016/j.bcp.2012.06.023.
Payer D, Balasubramaniam G, Boileau I. What is the role of the D3 receptor in addiction? A mini review of PET studies with [11C]-(+)-PHNO. Prog Neuropsychopharmacol Biol Psychiatry. 2014;52:4–8. https://doi.org/10.1016/j.pnpbp.2013.08.012.
Blaylock BL, Nader MA. Dopamine D3 receptor function and cocaine exposure. Neuropsychopharmacology. 2012;37:297–8. https://doi.org/10.1038/npp.2011.170.
Millan MJ, Loiseau F, Dekeyne A, Gobert A, Flik G, Cremers TI. et al. S33138 (N-[4-[2-[(3aS, 9bR)-8-cyano-1, 3a, 4, 9b-tetrahydro [1] benzopyrano [3, 4-c] pyrrol-2 (3H)-yl)-ethyl] phenyl-acetamide), a preferential dopamine D3 versus D2 receptor antagonist and potential antipsychotic agent: III. Actions in models of therapeutic activity and induction of side effects. J Pharmacol Exp Ther. 2008;324:1212–26.
Blass BE, Chen PJ, Taylor M, Griffin SA, Gordon JC, Luedtke RR, Design, synthesis, and evaluation of functionalized 5-(4-arylpiperazin-1-yl)-N-arylpentanamides as selective dopamine D3 receptor ligands, Med Chem Res, 2021. https://doi.org/10.1007/s00044-021-02825-3.
Hayatshahi HS, Xu K, Griffin SA, Taylor M, Mach RH, Liu J, et al. Analogues of arylamide phenylpiperazine ligands to investigate the factors influencing d3 dopamine receptor bitropic binding and receptor subtype selectivity. ACS Chem Neurosci. 2018;9:2972–2983. https://doi.org/10.1021/acschemneuro.8b00142.
Chen PJ, Taylor M, Griffin SA, Amani A, Hayatshahi H, Korzekwa K, et al. Design, synthesis, and evaluation of N-(4-(4-phenyl piperazin-1-yl)butyl)-4-(thiophen-3-yl)benzamides as selective dopamine D3 receptor ligands. Bioorg Med Chem Lett. 2019;29:2690–4. https://doi.org/10.1016/j.bmcl.2019.07.020.
Blass BE, Chen PJ, Taylor M, Griffin SA, Gordon JC, Luedtke RR, Design, synthesis, and evaluation of functionalized 5-(4-arylpiperazin-1-yl)-N-arylpentanamides as selective dopamine D3 receptor ligands, Med Chem Res, 2021. https://doi.org/10.1007/s00044-021-02825-3.
https://pdsp.unc.edu/pdspweb/content/PDSP%20Protocols%20II%202013-03-28.pdf.
Yang J, Jamei M, Yeo KR, Rostami-Hodjegan A, Tucker GT. Misuse of the well-stirred model of hepatic drug clearance. Drug Metab Dispos. 2007;35:501–2.
McMasters DR, Torres RA, Crathern SJ, Dooney D, Nachbar RB, Sheridan RP, et al. Inhibition of recombinant cytochrome P450 isoforms 2D6 and 2C9 by diverse drug-like molecules. J Med Chem. 2007;50:3205–13. https://doi.org/10.1021/jm0700060.
Besnard J, Ruda GF, Setola V, Abecassis K, Rodriguiz RM, Huang XP, et al. Automated design of ligands to polypharmacological profiles. Nature. 2012;492:215–20. https://doi.org/10.1038/nature11691.
Acknowledgements
D1, D2, D3, D4, and D5 Ki determinations were generously provided by the National Institute of Mental Health’s Psychoactive Drug Screening Program, Contract # HHSN-271-2018-00023-C (NIMH PDSP) [35]. The NIMH PDSP is Directed by Bryan L. Roth MD, PhD at the University of North Carolina at Chapel Hill and Project Officer Jamie Driscoll at NIMH, Bethesda MD, USA. For experimental details please refer to the PDSP web site https://pdsp.unc.edu/ims/investigator/web/. The content of this manuscript is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Blass, B.E., Chen, PJ. & Gordon, J.C. Synthesis of functionalized 5-(4-arylpiperazin-1-yl)-N-arylpentanamides and their evaluation as D3 receptor ligands. Med Chem Res 31, 735–748 (2022). https://doi.org/10.1007/s00044-022-02872-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-022-02872-4