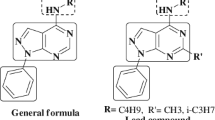
Abstract
A new class of pyrimidinyl bis(benzoxazoles/benzothiazoles/benzimidazoles) linked by amino sulfamido moieties were prepared and tested for antimicrobial activity. The bis(benzazolyl)pyrimidines (9, 10) displayed higher activity than the corresponding azolyl pyrimidines (4–7). In fact, bis(benzimidazolylamino)pyrimidinylsulfamide (9c), bis(benzothiazolylmethylamino)pyrimidinylsulfamide (10b), bis(benzimidazolylmethylamino)-pyrimidinylsulfamide (10c) were found to be potential antimicrobial agents against S. aureus and P. chrysogenum.



Similar content being viewed by others

References
Abdel-Wahab BF, Abdel-Aziz HA, Ahmed EM (2009) Synthesis and antimicrobial evaluation of 1-(benzofuran-2-yl)-4-nitro-3-arylbutan-1-ones and 3-(benzofuran-2-yl)-4,5-dihydro-5-aryl-1-[4-(aryl)-1,3-thiazol-2-yl]-1H-pyrazoles. Eur J Med Chem 44:2632–2635
Azoro C (2002) Antibacterial activity of crude aqeous extracts of Azadirachta indica on Salmonella typhi. World J Biotechnol 3:347–351
Balaswamy G, Srinivas K, Pradeep P, Sarangapani M (2012) Synthesis characterization and antimicrobial activity of novel substituted benzoxazole derivatives. Int J Chem Sci 10:619–626
Ban M, Taguchi H, Katsushima T, Aoki S, Watanabe A (1998) Novel antiallergic agents part I: synthesis and pharmacology of pyrimidine amide derivatives. Bioorg Med Chem 6:1057–1067
Bishnu J, Sunil L, Anuja S (2009) Antibacterial property of different medicinal plants: Ocimum sanctum, Cinnamomum zeylanicum, Xanthoxylum armatum and Origanum majorana. J Sci Eng Technol 5:143–150
Bradshaw TD, Westwell AD (2004) The development of the antitumour benzothiazole prodrug, phortress as a clinical candidate. Curr Med Chem 11:1009–1021
Chung KT, Thomasson WR, Wu-Yuan CD (1990) Growth inhibition of selected food-borne bacteria, particularly Listeria monocytogenes, by plant extracts. J Applied Bacteriol 69:498–503
Clinical and Laboratory Standards Institute (CLSI), reference method for broth dilution antifungal susceptibility testing of yeasts, approved standard—third Edition. CLSI document M27-A3; Wayne, PA 2008
Clinical and Laboratory Standards Institute, methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically, approved standard—ninth edition. CLSI document M07-A9; Wayne, PA 2012
Fache F, Schulz E, Tommasino ML, Lemaire M (2000) Nitrogen-containing ligands for asymmetric homogenous and heterogenous catalysis. Chem Rev 100:2159–2231
Gumus F, Algul O, Eren G, Eroglu H, Diril N, Gur S, Ozkul A (2003) Synthesis, cytotoxic activity on MCF-7 cell line and mutagenic activity of platinum(II) complexes with 2-substituted benzimidazole ligands. Eur J Med Chem 38:473–480
Gupta A, Rawat S (2010) Synthesis and cyclization of benzothiazole: review. J Curr Pharm Res 3:13–23
Hili R, Yudin AK (2006) Making carbon-nitrogen bonds in biological and chemical synthesis. Nat Chem Biol 2:284–287
Janovska D, Kubikov K, Kokoska L (2003) Screening for antimicrobial activity of some medicinal plants species of traditional chinese medicine. Czech J Food Sci 21:107–110
Jyothi M, Ramchander M (2013) Antibacterial and antifungal activity of some newly substituted benzoxazoles. Int J Chem Tech Res 5:2425–2428
Kamal A, Srinivasa Reddy K, Naseer A, Khan M, Rajesh VCRNCS, Janaki Ramaiah M, Pushpavalli SNCVL, Srinivas C, Pal-Bhadra M, Chourasia M, Narahari Sastry G, Juvekar A, Zingde S, Barkume M (2010) Synthesis, DNA-binding ability and anticancer activity of benzothiazole/benzoxazole–pyrrolo[2,1-c][1,4]benzo-diazepine conjugates. Bioorg Med Chem 18:4747–4761
Kaniwa K, Ohtsuki T, Yamamoto K, Ishibashi M (2006) Kehokorins A-C, novel cytotoxic dibenzofurans isolated from the myxomycete Trichia favogineavar. persimilis. Tetrahedron Lett 47:1505–1508
Kumar D, Jacob MR, Reynolds MB, Kerwin SM (2002) Synthesis and evaluation of anticancer benzoxazoles and benzimidazoles related to UK-1. Bioorg Med Chem 10:3997–4004
Mariappan G, Prabhat P, Sutharson L, Banerjee J, Patangia U, Nath S (2012) Synthesis and antidiabetic evaluation of benzothiazole derivatives. J Korean Chem Soc 56:251–256
Messina F, Botta M, Corelli F, Paladino A (2000) Chiral azole derivatives. Part 5: synthesis of enantiomerically pure 1-[(benzofuran-2-yl)arylmethyl]-1H-1,2,4-triazoles, antifungal and antiaromatase agents. Tetrahedron: Asymmetry 11:4895–4901
Miwa T, Hitaka T, Akimoto H, Nomurat H (1991) Novel Pyrrolo[2,3-d]pyrimidine antifolates: synthesis and antitumor activities. J Med Chem 34:555–560
Paget CJ, Kisner K, Stone RL, Delong DC (1969) Heterocyclic substiutued urease.II. Immunosuppressive and antiviral activity of benzothiazole and benzoxazoleureas. J Med Chem 12:1016–1018
Pilyugin V, Sapozhnikov Y, Davydov A, Chikisheve G, Vorobeva T, Klimakova E, Kiseleva G, Kuznetosova S, Davletov R, Sapozhnikov N, Yamadilov R (2006) 13C NMR spectra and biological activity of N-(1H-benzimidazolyl-2-yl)benzamides. Russ J Gen Chem 76:1653–1659
Pretorius SI, Breytenbach WJ, de Kock C, Smith PJ, N’Da DD (2013) Synthesis, characterization and antimalarial activity of quinoline-pyrimidine hybrids. Bioorg Med Chem 21:269–277
Rajyalakshmi G, Rama Narasimha Reddy A, Sarangapani M (2011) Synthesis, characterization, anticancer and antioxidant activities of some novel N-(benzo[d]oxazol-2-yl)-2-(7or5-substituted-2-oxoindolin-3-ylidene)hydrazine carbox- amide derivatives. J Enzyme Inhib Med Chem 26:813–818
Seenaiah D, Ramachandra Reddy P, Mallikarjuna Reddy G, Padmaja A, Padmavathi V, Siva krishna N (2014) Synthesis, antimicrobial and cytotoxic activities of pyrimidinyl benzoxazole, benzothiazole and benzimidazole. Eur J Med Chem 77:1–7
Severo S, Stella F, Claudio T, Frank P, Peg D, Yusuke S, Akihiro A, Ewa DM, Lawrence HL, Gianfranco B (2008) Role of benzimidazole (Bid) in the δ–opiod agonist pseudopeptide H-Dmt-Tic-NH-CH2-Bid (UFP-502). Bioorg Med Chem 16:3032–3038
Sommer PSM, Almeida RC, Schneider K, Beil W, Süssmuth RD, Fiedler HP (2008) Nataxazole, a new benzoxazole derivative with antitumor activity produced by streptomyces sp. Tü 6176. J Antibiot 61:683–686
Sondhi SM, Singh N, Johara M, Kumar A (2005) Synthesis, anti-inflammatory and analgesic activities evaluation of some mono, bi and tricyclic pyrimidine derivatives. Bioorg Med Chem 13:6158–6166
Srinivas A, Vidya Sagar J, Raju J, Rama G, Sarangapani M (2010) Design, synthesis and biological evaluation of benzoxazole derivatives as cyclooxygenase inhibitors. Der Pharma Chemica 2:189–199
Srinivas Rao N, Kistareddy C, Balram B, Ram B (2012) Synthesis and antibacterial activity of novel imidazo[1,2-a]pyrimidine and imidazo[1,2-a]pyridine chalcones derivatives. Der Pharma Chemica 4:2408–2415
Sun L, Wu J, Zhang L, Luo M, Sun D (2011) Synthesis and antifungal activity of some novel pyrimidine derivatives. Molecules 16:5618–5628
Surry DS, Buchwald SL (2011) Dialkylbiaryl phosphines in Pd-catalyzed amination: a user’s guide. Chem Sci 2:27–50
Wang SB, Deng XQ, Zheng Y, Yuan YP, Quan ZS, Guan LP (2012a) Synthesis and evaluation of anticonvulsant and antidepressant activities of 5-alkoxytetrazolo[1,5-c]thieno[2,3-e]pyrimidine derivatives. Eur J Med Chem 56:139–144
Wang Y, Song X, Shao S, Zhong H, Lin F (2012b) An efficient, soluble and recyclable multiwalled carbon nanotubes-supported TEMPO for oxidation of alcohols. Rsc Adv 2:7693–7698
Acknowledgements
One of the authors V. Padmavathi is grateful to Council of Scientific and Industrial Research (CSIR), New Delhi for financial assistance under major research project. The DST-PURSE, Sri Venkateswara University, Tirupati, India is gratefully acknowledged for antimicrobial activity.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Seenaiah, D., Rekha, T., Padmaja, A. et al. Synthesis and antimicrobial activity of pyrimidinyl bis(benzazoles). Med Chem Res 26, 431–441 (2017). https://doi.org/10.1007/s00044-016-1758-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-016-1758-9