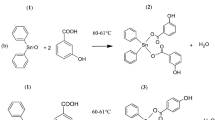
Abstract
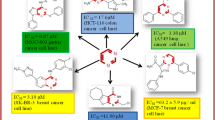
A series of novel 1,2,3-triazole-2H-benzo[b][1,4]oxazin-3(4H)-ones (4a–4k and 5a–5g) were designed, synthesized and screened for their anticancer activity against MCF-7 (breast) and HeLa (cervical) cell lines using [3-(4,5-Dimethylthiazol-2-yl)-2,5-Diphenyltetrazolium Bromide] assay. Among all the tested compounds, 5b and 5c exhibited good cytotoxic activity against two cancer cell lines with IC50 values nearer to the standard drug, cisplatin. The most active compounds 5b and 5c were further evaluated for their in vivo anticancer activity against Ehrlich Ascites Carcinoma-bearing mice and compound 5c found to possess potential cytotoxic activity as compared with the reference drug cisplatin. Molecular docking studies were also carried out to complement the experimental results.







Similar content being viewed by others
References
Bach PB, Jett JR, Pastorino U, Tockman MS, Swensen SJ, Begg CB (2007) Computed tomography screening and lung cancer outcomes. JAMA 297:953–961
Bennet IS, Brooks G, Broom NJP, Calvert SH, Coleman K, Francois I (1991) 6-(substituted methylene)penems, potent broad spectrum inhibitors of bacterial β-lactamase v. chiral 1, 2, 3-triazolyl derivatives. J Antibiot 44:969–978
Buckle DR, Rockell CJM, Smith H, Spicer BA (1986) Studies on 1,2,3-triazoles. 13. (Piperazinylalkoxy)-[1]benzopyrano[2,3-d]-1,2,3-triazol-9(1H)-ones with combined H1-antihistamine and mast cell stabilizing properties. J Med Chem 29:2262–2267
Buckman BO, Mohan R, Koovakkat S (1998) Design, synthesis, and biological activity of novel purine and bicyclic pyrimidine factor Xa inhibitors. Bioorg Med Chem Lett 8:2235–2240
Brockunier LL, Parmee ER, Ok HO, Candelore MR, Cascieri MA, Colwell LF, Deng L, Feeney WP, Forrest MJ, Hom GJ, MacIntyre DE, Tota L, Wyvratt MJ, Fisher MH, Weber AE (2000) Human β3-adrenergic receptor agonists containing 1,2,3-triazole-substituted benzenesulfonamides. Bioorg Med Chem Lett 10:2111–2114
Chen Y, Lopez-Sanchez M, Savoy DN, Billadeau DD, Dow GS, Kozikowski AP (2008) A series of potent and selective, triazolylphenyl-based histone deacetylases inhibitors with activity against pancreatic cancer cells and Plasmodium falciparum. J Med Chem 51:3437–3448
Chiu HI, Lin YC, Cheng CY, Tsai MC, Yu HC (2001) N-acyl-1,2,3,4a,5,10b-hexa hydro-[1]benzopyrano-[3,4-b][1,4]oxazine-9-carbonitriles as bladder-selective potassium channel openers. Bioorg Med Chem 9:383–393
Cindy JC, Gregory MR, Janina M, Laurent FB, Kasiram K, Julia M, Susan AC, Daniela V, Claudiu TS, Sally-Ann P (2013) A prodrug approach toward cancer-related carbonic anhydrase inhibition. J Med Chem 56:9623–9634
Cornelia MG, Qingzhong H, Sabrina R, Christina Z, Rolf WH (2015) Heteroatom insertion into 3,4-dihydro-1H-quinolin-2-ones leads to potent and selective inhibitors of human and rat aldosterone synthase. Eur J Med Chem 90:788–796
Gibbs JB (2000) Mechanism-based target identification and drug discovery in cancer research. Science 287:1969–1973
Haider S, Sarwar AM, Hamid H, Shafi S, Nargotra A, Mahajan P, Nazreen S, Kalle AM, Kharbanda C, Yakub A, Alam A, Panda AK (2013) Synthesis of novel 1,2,3-triazole based benzoxazolinones: their TNF-α based molecular docking with in-vivo anti-inflammatory, antinociceptive activities and ulcerogenic risk evaluation. Eur J Med Chem 70:579–588
Hazxa B, Sarkar R, Bhattacharya S, Roy P (2002) Tumour inhibitory activity of chicory root extract against Ehrlich ascites carcinoma in mice. Fitoterapia 73:730–733
Holla BS, Mahalinga M, Karthikeyan MS, Poojary B, Akberali PM, Kumari NS (2005) Synthesis, characterization and antimicrobial activity of some substituted 1,2,3-triazoles. Eur J Med Chem 40:1173–1178
Himo F, Lovell T, Hilgraf R, Rostovtsev VV, Noodleman L, Sharpless KB, Fokin VV (2005) Copper (I)-catalyzed synthesis of azoles. DFT study predicts unprecedented reactivity and intermediates. J Am Chem Soc 127:210–216
Jones JH, Anderson PS, Baldwin JJ, Clineschmidt BV, McClure DE, Lundell GF, Randall WC, Martin GE, Williams M, Hirshfield JM, Smith G, Lumma PK (1984) Synthesis of 4-substituted 2H-naphth[1,2-b]-l,4-oxazines, a new class of dopamine agonists. J Med Chem 27:1607–1613
Kelley JL, Koble CS, Davis RG, McLean EW, Soroko FE, Cooper BR (1995) 1-(Fluoro benzyl)–4–amino-1H-1,2,3-triazolo [4,5-c] pyridines: synthesis and anti convulsant activity. J Med Chem 38:4131–4134
Krasinski A, Radic Z, Manetsch R, Raushel J, Taylor P, Sharpless BK, Kolb HC (2005) In situ selection of lead compounds by click chemistry: target-guided optimization of acetylcholinesterase inhibitors. J Am Chem Soc 127:6686–6692
Kumaraswamy B, Narsimha S, Vasudeva RN, Priyanka B, Srinivasa Rao M (2016) Synthesis and antimicrobial evaluation of some novel thiomorpholine derived 1,4-disubstituted 1,2,3-triazoles. J Serb Chem Soc 81:233–242
Lanni TB, Greene KL, Kolz CN, Para KS, Visnick M, Mobley JL, Dudley DT, Baginskib TJ, Liimattab MB (2007) Design and synthesis of phenethyl benzo[1,4]oxazine-3-ones as potent inhibitors of PI3kinaseγ. Bioorg Med Chem Lett 17:756–760
Monks A, Scudiero D, Shekan P, Shoemaker R, Pamil K, Vistica D, Hose C, Langley J, Cromise P (1991) Feasibility of a high-flux anticancer drug screen using a diverse panel of cultured human tumor cell lines. J Natl Cancer Inst 83:757–766
Morris GM, Goodsell DS, Halliday RS, Huey R, Hart WE, Belew RK, Olson AJ (1998) Automated docking using a Lamarckian genetic algorithm and an empirical binding free energy function. J Comput Chem 19:1639–1662
Narsimha S, Kumar NS, Kumaraswamy B, Vasudeva RN, Hussain AS, Srinivasa Rao M (2016) Indole-2-carboxylic acid derived mono and bis 1,4-disubstituted 1,2,3-triazoles: synthesis, characterization and evaluation of anticancer, antibacterial, and DNA- cleavage activities. Bioorg Med Chem Lett 26:1639–1644
Narsimha S, Kumar TR, Kumar NS, Yakub S, Vasudeva RN (2014) Synthesis and antibacterial activity of (1-aryl-1, 2, 3-triazol-4-yl) methyl esters of morpholine-3-carboxylic acid. Med Chem Res 23:5321–5327
Narsimha S, Kumaraswamy B, Sudhakar L, Vasudeva RN, Hussain AS (2016) N,N’-(hexane-1,6-diyl)bis(N-((1-aryl/alkyl-1H-1,2,3-triazol-4-yl)methyl)-4-methyl benzenesulfonamide): synthesis, antibacterial, antioxidant, and DNA-cleavage activities. Phosphorus Sulfur Silicon Relat Elements. doi: 10.1080/10426507.2016.1146273
Ohno M, Tanaka Y, Miyamoto M, Takeda T, Hoshi K, Yamada N, Ohtake A (2006) Development of 3,4-dihydro-2H-benzo[1,4]oxazine derivatives as dual thromboxane A2 receptor antagonists and prostacyclin receptor agonists. Bioorg Med Chem 14:2005–2021
Olesen PH, Sørensen AR, Urso B, Kurtzhals P, Bowler AN, Ehrbar U, Hansen BF (2003) Synthesis and in vitro characterization of 1-(4-aminofurazan-3-yl)-5-dialkylaminomethyl-1H-[1,2,3]triazole-4-carboxylic acid derivatives. A new class of selective GSK-3 inhibitors. J Med Chem 46:3333–3341
Rajanarendar E, Raju S, Nagi Reddy M, Rama Krishna S, Hari Kiran L, Ram Narasimha Reddy A, Narasimha Reddy Y (2012) Multi-component synthesis and in vitro and in vivo anticancer activity of novel arylmethylene bis-isoxazolo[4,5-b]pyridine-N-oxides. Eur J Med Chem 50:274–279
Rajitha C, Dubey PK, Venkataiah S, Javier PF, Venugopal RV, Manojit P (2011) Synthesis and pharmacological evaluations of novel 2H-benzo[b][1,4]oxazin-3(4H)-one derivatives as a new class of anti-cancer agents. Eur J Med Chem 46:4887–4896
Rao PS, Kurumurthy C, Veeraswamy B, Kumar GS, Poornachandra Y, Kumar CG, Babu VS, Kotamraju S, Narsaiah B (2014) Synthesis of novel 1,2,3-triazole substituted-N-alkyl/aryl nitrone derivatives, their anti-inflammatory and anticancer activity. Eur J Med Chem 80:184–191
Reddy DM, Srinivas J, Chashoo G, Saxena AK, Kumar HMS (2011) 4β-[(4-Alkyl)-1,2,3-triazol-1-yl] podophyllotoxins as anticancer compounds: design, synthesis and biological evaluation. Eur J Med Chem 46:1983–1991
Ranjith KT, Narsimha S, Kumaraswamy B, Rajendra CV, Estari M, Vasudeva RN (2015) Synthesis, anticancer and antibacterial evaluation of novel (isopropylidene) uridine-[1,2,3]triazole hybrids. J Saud Chem Soc. doi: 10.1016/j.jscs.2015.12.001
Seruga JA, Barbero LG, Marquez J (2000) Ehrlich ascites tumour unbalances splenic cell populations and reduces responsiveness of T cells to Staphylococcus aureus enterotoxin B stimulation. Immunol Lett 74:111–115
Shekan P, Storeng R, Scudiero D, Monks A, Mc Mohan J, Vistica D, Warren JT, Bokesch H, Kenncy S, Boyd MR (1990) New colorimetric cytotoxicity assay for anticancer-drug screening. J Natl Cancer Inst 82:1107–1112
Shimura N, Musya A, Hashimoto T, Kojima S, Kubodera A, Sasaki T (2000) Usefulness of 99mTc-d,l-HMPAO for estimation of GSH content in tumor tissues. Nuclear Med Biol 27:577–580
Siegel R, Naishadham D, Jemal A (2013) Cancer statistics, 2013. CA Cancer J Clin 63:11–30
Singh P, Raj R, Kumar V, Mahajan MP, Bedi PMS, Kaur T, Saxena AK (2012) 1,2,3-Triazole tethered β-lactam-chalcone bifunctional hybrids: synthesis and anticancer evaluation. Eur J Med Chem 47:594–600
Soltis MJ, Yeh HJ, Cole KA, Whittaker N, Wersto RP, Kohn EC (1996) Identification and characterization of human metabolites of CAI [5-amino-1-1(4’-chlorobenzoyl-3,5-dichlorobenzyl)-1,2,3-triazole-4-carboxamide). Drug Metab Dispos 24:799–806
Stefely JA, Palchaudhuri R, Miller PA, Peterson RJ, Morask GC, Hergenrother PJ, Miller MJ (2010) N-((1-Benzyl-1H-1,2,3-triazol-4-yl)methyl)arylamide as a new scaffold that provides rapid access to antimicrotubule agents: synthesis and evaluation of antiproliferative activity against select cancer cell lines. J Med Chem 53:3389–3395
Stilwell GA, Adams HG, Turck M (1975) In vitro evaluation of a new oral cephalosporin, cefatrizine (BL-S640). Antimicrob Agents Chemother 8:751–753
Sugimoto Y, Otani T, Oie S, Wierzba K, Yamada Y (1990) Mechanism of action of a new macromolecular antitumor antibiotic, C-1027. J Antibiot 43:417–421
Sunny M, Shabana IK, Diwan SR (2011) Synthesis of 4-aminoquinoline-1,2,3-triazole and 4-aminoquinoline-1,2,3-triazole-1,3,5-triazine hybrids as potential antimalarial agents. Chem Biol Drug Des 78:124–136
Suresh S, Narsimha S, Kumaraswamy B, Hussain AS, Tangeda SJ, Vasudeva RN (2015) One pot synthesis of 1-((1-aryl-1H-1,2,3-triazol-4-yl)methyl)-1H-benzo[d] imidazoles in ionic liquids: evaluation of antioxidant and antimicrobial activities. Eur J Chem 6:482–487
Velazquez S, Alvarez R, Perez C, Gago F, Clercq E, De, Balzarini J, Camarasa MJ (1998) Regiospecific synthesis and anti-human immunodeficiency virus activity of novel 5-substituted N-alkylcarbamoyl and N,N-dialkyl carbamoyl 1,2,3-triazole-TSAO analogues. Antiviral Chem Chemother 9:481–489
Xue-Wen Z, Han-Lin M, Xuan Z, Shi-Yao J, Jun-Ying M, Bao-Xiang Z (2014) Synthesis of 6-cinnamoyl-2H-benzo[b][1,4]oxazin-3(4H)-ones and their effects on A549 lung cancer cell growth. Eur J Med Chem 79:95–101
Zhao SH, Berger J, Clark RD, Sethofer SG, Krauss NE, Brothers JM, Martin RS, Misner DL, Schwabd D, Alexandrovad L (2007) 3,4-Dihydro-2H-benzo[1,4]oxazine derivatives as 5-HT6 receptor antagonists. Bioorg Med Chem Lett 17:3504–3507
Zhong-Tai P, Li-Ping G, Li-Ming Z, Hu-Ri P, Zhe-Shan Q (2008) Synthesis of novel 7-benzylamino-2H-1,4-benzoxazin-3(4H)-ones as anticonvulsant agents. Eur J Med Chem 43:1216–1221
Zhou DH, Harrison BL, Shah U, Andree TH, Hornby GA, Scerni R, Schechter LE, Smith DL, Sullivanb KM, Mewshawa RE (2006) Studies toward the discovery of the next generation of antidepressants. Part 5: 3,4-dihydro-2H-benzo[1,4]oxazine derivatives with dual 5-HT1A receptor and serotonin transporter aff inity. Bioorg Med Chem Lett 16:1338–1341
Acknowledgments
Financial assistance from the UGC-MRP (F.No. 42-372/2013), New Delhi, India, is greatly acknowledged. Narsimha and Satheesh are thankful to the Council of Scientific and Industrial Research-University Grant Commission (CSIR-UGC, New Delhi) for the award of senior research fellowship.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interests.
Rights and permissions
About this article
Cite this article
Nagavelli, V.R., Nukala, S.K., Narsimha, S. et al. Synthesis, characterization and biological evaluation of 7-substituted- 4-((1-aryl-1H-1,2,3-triazol-4-yl) methyl)-2H-benzo[b][1,4]oxazin- 3(4H)-ones as anticancer agents. Med Chem Res 25, 1781–1793 (2016). https://doi.org/10.1007/s00044-016-1616-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-016-1616-9