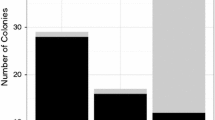
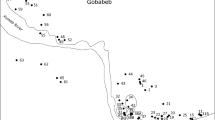
Abstract
Social insect colonies are among the most complex social organisations in nature, with reproductive and non-reproductive individuals co-ordinating to maintain the survival of the colony. Multiple reproductive schemes occur in social insects, from simple schemes with one founding reproductive pair, to more complex ones involving within-colony inbreeding and more than two unrelated reproductives. Colony breeding schemes and genetic structure remain understudied in termites, compared to the Hymenoptera. In this study, we performed the first genetic characterisation of the colony breeding structure of Nasutitermes exitiosus (Blattodea: Termitidae), an endemic Australian termite with a broad distribution across southern mainland Australia. We analysed the genetic structure of 60 N. exitiosus colonies from the Eastern part of its distribution, using microsatellites and mitochondrial sequence data. We found that most colonies were headed by one founding pair of reproductives, although some colonies exhibited a more complex breeding structure, including within-colony inbreeding and the presence of multiple unrelated reproductives. We found evidence for the presence of seven unrelated queens in one colony, to our knowledge, the highest number of unrelated queens yet found in a termite from the family Termitidae. We found some evidence for genetic isolation by distance, indicating that the species is a relatively poor disperser over long ranges.

Similar content being viewed by others
References
Ab Majid AH, Kamble S, Chen H (2014) Colony genetic organization and breeding pattern of subterranean termite (Reticulitermes flavipes) over the three field seasons in Nebraska, USA. Entomol Ornithol Herpetol Curr Res 3:1000133. https://doi.org/10.4172/2161-0983.1000133
Adams ES, Atkinson L, Bulmer MS (2007) Relatedness, recognition errors, and colony fusion in the termite Nasutitermes corniger. Behav Ecol Sociobiol 61:1195–1201. https://doi.org/10.1007/s00265-007-0349-7
Aldrich BT, Kambhampati S (2007) Population structure and colony composition of two Zootermopsis nevadensis subspecies. Heredity 99:443. https://doi.org/10.1038/sj.hdy.6801022
Arab DA, Namyatova A, Evans TA, Cameron SL, Yeates DK, Ho SYW, Lo N (2017) Parallel evolution of mound-building and grass-feeding in Australian nasute termites. Biol Lett 13:20160665. https://doi.org/10.1098/rsbl.2016.0665
Atkinson L, Adams ES (1997) The origins and relatedness of multiple reproductives in colonies of the termite Nasutitermes corniger. Proc R Soc B Biol Sci 264:1131–1136. https://doi.org/10.1098/rspb.1997.0156
Atkinson L, Adams ES, Crozier RH (2007) Microsatellite markers for the polygamous termites Nasutitermes corniger (Isoptera: Termitidae). Mol Ecol Notes 7:299–301. https://doi.org/10.1111/j.1471-8286.2006.01586.x
Atlas of Living Australia (2019) Nasutitermes exisiosus (Hill, 1925). https://bie.ala.org.au/species/urn:lsid:biodiversity.org.au:afd.taxon:c413e82b-40ae-4473-8461-f80ab97f9342. Accessed 5th July 2019
Beasley-Hall PG, Chui J, Arab DA, Lo N (2018a) Evidence for a complex evolutionary history of mound building in the Australian nasute termites (Nasutitermitinae). Biol J Linnean Soc 126:304–314. https://doi.org/10.1093/biolinnean/bly187%J
Beasley-Hall PG, Lee TRC, Rose HA, Lo N (2018b) Multiple abiotic factors correlate with parallel evolution in Australian soil burrowing cockroaches. J Biogeogr 45:1515–1528. https://doi.org/10.1111/jbi.13233
Brandl R, Hacker M, Bagine RKN, Kaib M (2004) Yearly variation in polygyny in the termite Macrotermes michaelseni (Sjöstedt). Insectes Soc 51:294–298. https://doi.org/10.1007/s00040-004-0747-z
Broughton RE (1995) Mitochondrial DNA variation within and among species of termites in the genus Zootermopsis (Isoptera: Termopsidae). Ann Entomol Soc Am 88:120–128. https://doi.org/10.1093/aesa/88.2.120
Bulmer MS, Adams ES, Traniello JFA (2001) Variation in colony structure in the subterranean termite Reticulitermes flavipes. Behav Ecol Sociobiol 49:236–243. https://doi.org/10.1007/s002650000304
Chiu C-I, Neoh K-B, Li H-F (2018) Colony-founding success of pleometrosis in a fungus-growing termite Odontotermes formosanus. Behav Ecol Sociobiol 72:13. https://doi.org/10.1007/s00265-017-2429-7
Cole BJ, Wiernasz DC (1999) The selective advantage of low relatedness. Science 285:891. https://doi.org/10.1126/science.285.5429.891
Darlington JPEC (1988) The structure of mature mounds of the termite Macrotermes herus in Kenya. Int J Trop Insect Sci 9:339–345. https://doi.org/10.1017/S1742758400006305
Deheer CJ, Vargo EL (2004) Colony genetic organization and colony fusion in the termite Reticulitermes flavipes as revealed by foraging patterns over time and space. Mol Ecol 13:431–441
DeHeer CJ, Vargo EL (2008) Strong mitochondrial DNA similarity but low relatedness at microsatellite loci among families within fused colonies of the termite Reticulitermes flavipes. Insectes Soc 55:190–199. https://doi.org/10.1007/s00040-008-0999-0
Evans TA, Lenz M, Gleeson PV (1998) Testing assumptions of mark-recapture protocols for estimating population size using Australian mound-building, subterranean termites. Ecol Entomol 23:139–159
Gay FJ, Calaby JH (1970) Termites of the Australian Region. In: Krishna K, Weesner FM (eds) Biology of termites, vol 2. Academic Press, London, pp 393–448
Gay FJ, Wetherly AH (1970) The population of a large mound of Nasutitiermes exitiosus (Hill) Isoptera: Termitidae. J Aust Entomol Soc 9
Hacker M, Kaib M, Bagine RK, Epplen JT, Brandl R (2005) Unrelated queens coexist in colonies of the termite Macrotermes michaelseni. Mol Ecol 14:1527–1532. https://doi.org/10.1111/j.1365-294X.2005.02507.x
Hartke TR, Baer B (2011) The mating biology of termites: a comparative review. Anim Behav 82:927–936. https://doi.org/10.1016/j.anbehav.2011.07.022
Hartke TR, Rosengaus RB (2013) Costs of pleometrosis in a polygamous termite. Proc R Soc B Biol Sci 280:20122563–20122563. https://doi.org/10.1098/rspb.2012.2563
Hochberg Y (1988) A sharper bonferroni procedure for multiple tests of significance. Biometrika 75:800–802
Holdaway FG, Gay FJ, Greaves T (1935) The termite population of a mound colony of Eutermes exitiosus Hill. J Council Sci Ind Res 8:42–46
Howard KJ, Johns PM, Breisch NL, Thorne BL (2013) Frequent colony fusions provide opportunities for helpers to become reproductives in the termite Zootermopsis nevadensis. Behav Ecol Sociobiol 67:1575–1585. https://doi.org/10.1007/s00265-013-1569-7
Hu J, Zhong JH, Guo MF, Li QJ, Liu BR (2007) Alate dispersal distances of the black-winged subterranean termite Odontotermes formosanus (Isoptera: Termitidae) in southern China. Sociobiology 50:1–8
Huang Q, Li G, Husseneder C, Lei C (2013) Genetic analysis of population structure and reproductive mode of the termite Reticulitermes chinensis Snyder. PLoS ONE 8:e69070. https://doi.org/10.1371/journal.pone.0069070
Jones JC, Myerscough MR, Graham S, Oldroyd BP (2004) Honey bee nest thermoregulation: diversity promotes stability. Science 305:402–404. https://doi.org/10.1126/science.1096340%JScience
Krishna K (1969) Biology of termites, vol 1. Academic Press, London
Lee TRC, Bourguignon T, Lo N (2017a) Female-only workers and soldiers in Schedorhinotermes intermedius are not produced by parthenogenesis. Insectes Soc 64:133–139. https://doi.org/10.1007/s00040-016-0523-x
Lee TRC, Evans TA, Cameron SL, Hochuli DF, Ho SYW, Lo N (2017b) Ecological diversification of the Australian Coptotermes termites and the evolution of mound building. J Biogeogr 44:1405–1417. https://doi.org/10.1111/jbi.12878
Luchetti A, Dedeine F, Velona A, Mantovani B (2013) Extreme genetic mixing within colonies of the wood-dwelling termite Kalotermes flavicollis (Isoptera, Kalotermitidae). Mol Ecol 22:3391–3402. https://doi.org/10.1111/mec.12302
Mattila HR, Seeley TD (2014) Extreme polyandry improves a honey bee colony’s ability to track dynamic foraging opportunities via greater activity of inspecting bees. Apidologie 45:347–363. https://doi.org/10.1007/s13592-013-0252-3
Messenger MT, Mullins AJ (2005) New flight distance recorded for Coptotermes formosanus (Isoptera: Rhinotermitidae). Florida Entomologist 88:99–100. https://doi.org/10.1653/0015-4040(2005)088%5b0099:NFDRFC%5d2.0.CO;2
Miller MP (2005) Alleles in space (AIS): computer software for the joint analysis of interindividual spatial and genetic information. J Hered 96:722–724. https://doi.org/10.1093/jhered/esi119
Noirot C (1990) Sexual castes and reproductive strategies in termites. In: Engels W (ed) Social insects: an evolutionary approach to castes and reproduction. Springer, Berlin, pp 5–35
Peakall R, Smouse PE (2006) genalex 6: genetic analysis in Excel. Population genetic software for teaching and research. Mol Ecol Resour 6:288–295. https://doi.org/10.1111/j.1471-8286.2005.01155.x
Peakall R, Smouse PE (2012) GenAlEx 6.5: genetic analysis in Excel. Population genetic software for teaching and research—an update. Bioinformatics 28:2537–2539. https://doi.org/10.1093/bioinformatics/bts460
Queller DC, Goodnight KF (1989) Estimating relatedness using genetic markers. Evolution 43:258–275. https://doi.org/10.1111/j.1558-5646.1989.tb04226.x
Queller DC, Strassmann JE (1998) Kin Selection and Social Insects: Social insects provide the most surprising predictions and satisfying tests of kin selection BioScience 48:165–175. https://doi.org/10.2307/1313262%JBioScience
Roisin Y (1993) Selective pressure on pleometrosis and secondary polygyny: a comparison of termites and ants. In: Keller L (ed) Queen number and sociality in insects. Oxford University Press, New York, pp 402–421
Roisin Y, Pasteels JM (1986) Replacement of reproductives in Nasutitermes princeps (Desneux) (Isoptera: Termitidae). Behav Ecol Sociobiol 18:437–442. https://doi.org/10.1007/bf00300519
Snyder TE (1926) The biology of the termite castes. Q Rev Biol 1:522–552
Su XH, Xue W, Liu H, Chen JL, Zhang XJ, Xing LX, Liu MH (2015) The development of adultoid reproductives and brachypterous neotenic reproductives from the last instar nymphs in Reticulitermes labralis (Isoptera: Rhinotermitidae): a comparative study. J Insect Sci 15:147. https://doi.org/10.1093/jisesa/iev130
Tarpy DR (2003) Genetic diversity within honeybee colonies prevents severe infections and promotes colony growth. Proc Biol Sci 270:99–103. https://doi.org/10.1098/rspb.2002.2199
Thompson GJ, Hebert PDN (1998) Probing termite social systems through allozyme and mtDNA analysis: a case study of Nasutitermes nigriceps and Nasutitermes costalis (Isoptera, Termitidae). Insectes Soc 45:289–299. https://doi.org/10.1007/s000400050089
Thompson GJ, Lenz M, Crozier RH, Crespi BJ (2007) Molecular-genetic analyses of dispersal and breeding behaviour in the Australian termite Coptotermes lacteus: evidence for non-random mating in a swarm-dispersal mating system. Aust J Zool 55:219–227. https://doi.org/10.1071/ZO07023
Thorne BL (1984) Polygyny in the Neotropical termite Nasutitermes corniger: life history consequences of queen mutualism. Behav Ecol Sociobiol 14:117–136. https://doi.org/10.1007/bf00291903
Thorne B, Traniello J, Adams E, Bulmer M (1999) Reproductive dynamics and colony structure of subterranean termites of the genus Reticulitermes (Isoptera Rhinotermitidae): a review of the evidence from behavioral, ecological, and genetic studies. Ethol Ecol Evol 11:149–169. https://doi.org/10.1080/08927014.1999.9522833
Vargo EL (2019) Diversity of termite breeding systems. Insects 10:52. https://doi.org/10.3390/insects10020052
Vargo EL, Husseneder C (2011) Genetic structure of termite colonies and populations. In: Bignell DE, Roisin Y, Lo N (eds) Biology of termites: a modern synthesis. Springer, Dordrecht
Wiernasz DC, Hines J, Parker DG, Cole BJ (2008) Mating for variety increases foraging activity in the harvester ant, Pogonomyrmex occidentalis. Mol Ecol 17:1137–1144. https://doi.org/10.1111/j.1365-294X.2007.03646.x
Acknowledgements
The authors would like to thank Patrick Gleeson and Thomas Madsen for their assistance in collecting termite specimens. We are grateful to Soren and Sonya Lunoe from Turon Gates Mountain Retreat for allowing us to collect termites on their property.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Montagu, A., Lee, T.R.C., Ujvari, B. et al. High numbers of unrelated reproductives in the Australian ‘higher’ termite Nasutitermes exitiosus (Blattodea: Termitidae). Insect. Soc. 67, 281–294 (2020). https://doi.org/10.1007/s00040-020-00764-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00040-020-00764-7