Abstract
The microbial control agent Bacillus thuringiensis var. israelensis (Bti) has been successfully used worldwide to reduce abundances of biting Nematocera (Diptera), often with little direct impact on non-target organisms observed. However, the potential for additional indirect effects on other ecosystem properties, including on trophic linkages within food webs, is poorly known. We investigated the effects of multiple-year mosquito control treatments using the Bti product VectoBac®-G on the stable isotope composition of epigeal and soil-based consumers inhabiting replicate floodplains along the River Dalälven, Sweden. We observed significant changes in the isotopic composition of detritivores feeding at the base of floodplain food webs. Enchytraeid worms were characterised by 3.5% higher δ13C values in treated floodplains, suggesting increased consumption of δ13C-enriched food. The overall range of community-wide δ15N values was 56% greater in the treated floodplains, whilst δ15N values of oribatid mites were elevated by 97%. These results suggest extra fractionation in the transfer of nitrogen through floodplain food chains. We conjecture that the ecological mechanisms driving these food web shifts are (1) the mass mortality of high δ13C A. sticticus larvae, which leaves high concentrations of dead mosquito biomass deposited on soils at local scales, after the floodwaters have receded and (2) incorporation of the very high δ13C-enriched corn particles comprising the bulk of the VectoBac®-G product into floodplain food webs. Our results suggest that repeated applications of Bti might have wider, still largely unknown implications for nutrient and energy cycles within floodplain ecosystems.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The microbial control agent Bacillus thuringiensis var. israelensis (Bti) has been successfully used worldwide to reduce emergence of adult mosquitoes (Culicidae) and other nuisance blood-feeding Nematocera (Diptera) from lakes, rivers and floodplains (Boisvert and Boisvert 2000; Brühl et al. 2020; Land et al. 2019; Schäfer and Lundström 2014). Unlike most other aquatic invertebrates, Nematoceran Diptera have an alkaline gut environment, within which Bti produces crystal aggregations containing toxins disrupting the gut wall (Boisvert and Boisvert 2000). This results in mortality of the target organisms in the larval stage, without the often severe environmental impacts of chemical insecticides (Becker et al. 2010; Boisvert and Boisvert 2000). However, impacts of Bti on potentially vulnerable non-target organisms (NTOs), primarily comprising other Nematocera such as the ecologically diverse non-biting midges (Chironomidae), have been observed in some field studies (Allgeier et al. 2019b; Hershey et al. 1998; Vaughan et al. 2008), though not others (Boisvert and Boisvert 2000; Lundstrom et al. 2010). Additionally, the potential for indirect effects on other ecosystem properties, including trophic linkages and the flow of carbon and nutrients through food webs, is increasingly raised as a possible risk associated with the use of Bti (Brühl et al. 2020; Hershey et al. 1998; Poulin et al. 2010), but has been little assessed in field research (Land et al. 2019).
Indirect effects on food webs might arise when Bti-induced changes in the density and/or behaviour of one or more organism groups result in alterations of food web structure (e.g. number of trophic levels), and/or function, e.g. processing of different resources or biomass production (Land et al. 2019; Truchy et al. 2015). In the case of Bti, both target organisms and the most vulnerable NTOs complete their larval development in aquatic or semi-aquatic habitats before emerging into the terrestrial environment as winged adults, sometimes at very high densities (Armitage et al. 1994; Becker et al. 2010; Carlson et al. 2016). Adult aquatic insects are often notably rich in polyunsaturated fatty acids which are essential for animal growth and development, and comprise an important resource for both vertebrate (birds, bats, amphibians) and invertebrate (spiders, insects) predators (Müller-Navarra et al. 2000; Ramberg et al. 2020). Accordingly, the massive (e.g. 90–100%, Schäfer and Lundström 2014) reductions in the biomass of adult mosquitoes following Bti treatment dramatically reduces a potentially abundant and nutrient-rich food source for terrestrial organisms. Indeed, 66% reductions in the biomass of emerging Nematoceran Dipterans (mosquitoes and chironomids) from French floodplains treated with Bti were associated with reduced breeding success and fitness of house martins (Delichon urbicum L.) (Poulin et al. 2010).
Control of mosquitoes using Bti has particularly strong potential to affect ground-based (epigeal) and soil-based food webs. Extreme densities of mosquitoes and other mass-emerging aquatic insects regularly exceed the feeding capacities of their main predators (Yang et al. 2010). As such, a large portion of aquatic insect productivity ends up deposited on riparian or floodplain soils as dead adults (Hoekman et al. 2012). Deposition of dead adult chironomids has been linked with an increase in the number of soil-based detrivores including Collembola and mites (Hoekman et al. 2011), and to increased soil nutrient availability (Gratton et al. 2017). Additional indirect and poorly studied effects on soil food webs might arise from the vector by which the Bti spores are delivered. For example, the widely used formulation VectoBac®-G (Valent BioSciences, USA) consists of pellets derived from the husks of corn (Zea mays) cobs treated with a dried culture of Bti, attached with the help of corn oil (Lundstrom et al. 2010). During mosquito control, these pellets are typically distributed by helicopter over water bodies or inundated floodplains, where they are either immediately consumed by biota or, alternatively, enter the pool of organic detritus available to microorganisms and detritivores.
In this study, we focus on the effects of mosquito control using Bti on epigeal and soil food webs of floodplains in the lower River Dalälven catchment, Sweden. Beginning in the final years of the twentieth century, this region suffered increasingly severe outbreaks of the floodplain mosquito Aedes sticticus Meigen (Schäfer and Lundström 2014). A Bti-based control program initiated in 2002 has been highly effective at reducing abundances of flying A. sticticus by 90–100%, with little evidence of negative outcomes for NTOs including Chironomidae (Lundstrom et al. 2010). Nevertheless, increases in abundance of Bti itself in treated floodplain soils (Schneider et al. 2017), along with substantial increases in density and taxonomic richness of heterotrophic protozoans (Östman et al. 2008), point to potential changes in the structure and function of soil and epigeal food webs.
We used stable isotopes to characterise shifts in the soil- and ground-based food webs of floodplain habitats of the lower Dalälven River catchment, following more than a decade of Bti application (as granular VectoBac®-G) for mosquito control. We studied the isotopic composition of various groups of ground- and soil-based detritivores, representing the probable first scavengers of dead mosquitos in floodplain soils and/or grazers of microbial decomposers of mosquito remains, and of key invertebrate predators (spiders and beetles). As a C4 plant, the corn comprising the bulk of the VectoBac®-G product has a strongly contrasting isotopic composition compared with the C3 plants that comprise the vast majority of terrestrial flora in Sweden (Pyankov et al. 2010). Based on this, we expected that any incorporation of these corn particles into the ground-based food webs might alter the C isotope values of consumers. We hypothesised that:
-
1)
Detritivore δ13C values are expected to be higher in the treated than control floodplains, in line with the deposition of C4-plant-derived particles used in delivery of Bti.
-
2)
Consumer δ15N isotopes will be enriched in the treated floodplains, if the mass accumulation of dead larvae acts as a significant nutrient subsidy in the treated floodplains, lengthening food chains (Hoekman et al. 2012).
We further analysed adult A. sticticus for their isotopic composition and polyunsaturated fatty acid (PUFA) content, to gain insight into their value as a food resource for higher consumers (Goedkoop et al. 2007; Müller-Navarra et al. 2000).
Materials and methods
Study sites and mosquito control
The lower River Dalälven region is characterised by a flat alluvial landscape, encompassing Scandinavia’s only true river Delta, and interspersed with extensive wet meadow habitats that are strongly shaped by recurring floods (Holmstedt and Linderheim 2019). High river discharges and subsequent flooding can occur several times over the course of a growing season. Conservation values for the region are high, due to a rich biodiversity of plant and animal life, patches of ancient forest, and the presence of many endangered species and habitat types. Much of the region has received European Union Natura 2000 designation, and is protected in national parks or nature reserves.
Since 2002, the Biological Mosquito Control Project (BMCP) (www.mygg.se) has repeatedly treated several of the floodplain areas to limit the mass emergence of the nuisance mosquito A. sticticus using Bti. Aedes sticticus is a relatively large-bodied and aggressive mosquito that attacks both humans and livestock, and which has had severe socioeconomic effects in the lower River Dalälven region, including on tourism (Schäfer and Lundström 2014). A. sticticus lays its eggs in wet-meadow and alder swamp habitats, where their eggs can lay dormant for many years. The eggs typically hatch when a dry period in spring or summer is followed by a flood with water temperature exceeding 8–10 degrees (Schäfer and Lundström 2009). After the floodwater recedes (often after only a few days), the larvae continue their development in remaining temporary standing water habitats. The exact diet of A. sticticus larvae in Sweden has not been characterised. However, larval floodplain mosquitoes elsewhere consume mostly particulate detritus and microbes, with algae important only where environmental conditions (light and nutrient levels) favour significant primary production (Merritt et al. 1992).
We sampled epigeal and soil invertebrates from five treated and five control floodplains that are included in the BMCP long-term monitoring program. All are located in and around Färnebofjärden National Park, in close proximity to the main Dalälven river stem (Fig. 1, Tables S1-2). All sampled floodplains are dominated by “wet meadow” habitats, with soils water saturated for much of the growing season and characterised by a cover of herbaceous sedges and grasses (Table 1). Treated floodplains have been subjected to mosquito control starting in 2002, using the granular formulation of VectoBac-G®, applied at a dosage of 13–15 kg per hectare (Schäfer and Lundström 2014). The continuous area of treated habitat encompassing our sampling sites ranged from 1.1 to 11.3 km2 (Table S2). Prior to 2014 (the year of our study), treatments had been applied during May–August in most years, and most often 2–3 times per year, excepting 2004 and 2013 (no applications) and 2009 and 2012 (four applications) (Schäfer and Lundström 2014, supplemented with information for 2013 from www.mygg.se). Reference floodplains have never been treated, and all are situated more than 1.75 km from the nearest treated area (Table S2).
The ten floodplains were divided into five treated–reference pairs based on their proximity to the main river, soil types and vegetation composition (Tables S1-2, Fig. 1). This paired design was used in our statistical models to account for background variation amongst the floodplains that might have affected our analyses of mosquito control effects. Information on soil type was obtained from the Swedish Geological Survey (SGU, www.sgu.se). Vegetation composition was assessed onsite during field sampling, with the percent cover of different grasses, herbs and woody vegetation recorded in four 5-m2 quadrats per floodplain. Pairs 1–3 were dominated by grasses and herbs, with pairs 1–2 also characterised by significant growth of Salix (willow) (Table S1). Pairs 4–5 are characterised by significant Sphagnum cover, which was lacking in pairs 1–3. Overall, neither the elevation nor ground cover of the vegetation categories differed significantly between treated and floodplain categories, except for Salix cover which was slightly greater in treated floodplains overall (Table S1).
Isotopic biplots (δ15N and δ13C) for reference (upper panel) and treated floodplains (lower panel) showing basal resources (colors), key taxa of detritivores (small letters) and predators (capital letters), as well as samples of Aedes sticticus and VectoBac®-G. Resources plotted as mean values ± 1 SD. For VectoBac®-G, means (± sd) are plotted for three technical subsamples, whilst for adult mosquitoes (A. sticticus), data for four individuals are plotted. Both panels are plotted using identical axes, to facilitate comparison
Field sampling
We collected organisms for stable isotope analysis during July 22–24, 2014, 2 weeks after the most recent mosquito control campaign (undertaken July 9–10 after the second mass emergence of A. sticticus during 2014; www.mygg.se). At the time of collection, the flood waters had receded and the floodplains were relatively dry. We focussed on key, predominantly saprophagous, invertebrate groups likely to respond to the deposition of dead mosquitoes and/corn particles in Vectobac G: (1) Annelid worms, including earthworms (Oligochaeta: Lumbricidae) and pot worms (Oligochaeta: Enchytraeidae), and (2) soil microarthropod detritivores, comprising springtails (Collembola) and moss mites (Acari: Oribatida). The two oligochaete families consume decaying plant material and other detritus and associated microflora (fungi, bacteria, actinomycetes, algae), whilst Lumbricidae also feed on living plant material and microfauna (Curry and Schmidt 2007; Didden 1993). The microarthropods Collembola and Oribatida similarly encompass species that predominantly consume decaying vegetation and associated microflora, but also include predacious and carrion-consuming scavenger species (Petersen 2002; Schneider et al. 2004). We further sampled ground-hunting predators, comprising wolf spiders (Lycosidae), fishing spiders (Pisauridae: Dolomedes fimbriatus) and rove beetles (Staphylinidae), to assess whether shifts in isotopic composition of detritivores and herbivores can be traced at higher trophic levels. Finally, when available we also collected semi-aquatic Diptera (Tipulidae, Chironomidae), which feed on particulate detritus and algae in moist floodplain habitats (Armitage et al. 1994), and predacious soil mites (Acari: Mesostigmata).
Wolf spiders and rove beetles were collected by sweep-netting vegetation near to ground level, whilst D. fimbriatus was hand collected by visually searching for individuals. We aimed to collect at least five individuals for each spider and beetle group in each floodplain. Captured spiders and beetles were kept alive in individual jars, cold and in the dark, until further processing in the laboratory.
All soil invertebrates, except Collembola, were collected from two types of habitats in the floodplains: raised tussocks formed of grasses and sedges, and damper depressions forming in between the tussocks. The dryer tussocks were targeted for collection of earthworms and oribatid and predacious mites, whilst pot worms and semi-aquatic Diptera were collected from the damp depressions. We sampled two tussocks and two damp depressions per floodplain. Tussocks were harder to locate in the Sphagnum-dominated pairs 4 and 5 (Table S1). In these floodplains we nevertheless divided our sampling between raised, drier areas of ground and lower, wetter depressions. To sample soil in the tussocks, we used a spade to first cut off the grassy top, and then dug a c. 800–1000 cm3 (18 × 18 x c. 2–3 cm depth) soil core. Damp depressions were sampled using an 8 (inner diameter) × 15 cm cylindrical soil corer (754 cm3). Larger earthworms were sorted immediately from the tussock core in the field and retained in jars with additional soil from the site. All soil core samples were then stored in plastic bags and returned in cool boxes to the laboratory for extraction of soil fauna on the same day. Biota from the tussock samples were extracted over Tullgren funnels for a total of 4 days (Tullgren 1971). Soil cores from the damp depressions were extracted over 3 days using a cold wet extraction technique, with the aid of modified Baermann funnels (Didden et al. 1995). Animals were picked daily during extraction and stored cold in the dark.
Springtails (Collembola) were sampled by collecting a larger soil volume (0.02 m3) with a spade from two tussock habitats per floodplain. In the field, this soil sample was placed in a bucket of river water, and individuals were picked as they floated to the surface. As we were unable to collect sufficient springtail biomass for isotopic analysis during our first sampling visit, we collected additional springtails during a second sampling trip to the floodplains on 11–12 August 2014. There were no new flood or mosquito control events between these two trips.
We also collected plant, biofilm and soil samples for isotopic analysis, to characterise the basal resources available to earthworms and microdetrivores in the floodplains. Based on our vegetation inventories, Cirsium palustre (marsh thistle) and Carex spp. were selected to represent common terrestrial litter types, occurring in all floodplains. We collected leaves from multiple specimens of these plants across a 50 m2 area per floodplain, and stored them cold for return to the laboratory. During our second field visit, we additionally collected samples for analysis of soil isotopes (five soil cores per floodplain) and biofilm from both aquatic and terrestrial plants. Biofilm from terrestrial plants was collected by cutting plants close to ground level, which were then placed in 11L buckets filled with distilled water. To collect aquatic biofilm, submersed fragments of aquatic reeds (Phragmites australis) were cut from the water surface down to a depth of 10 cm. Both aquatic and terrestrial plant samples were both placed directly into 11 L buckets filled with 2.5L distilled water. The plants were gently brushed in the distilled water to remove biofilm. The water (1.5–2.5 dL) containing biofilm was then filtered through pre-combusted Whatman glass fiber filters (GF/F, effective pore size = 0.7 µm) to capture biofilm-associated algae and other microbes. The filters were then inspected to remove any larger particles, and placed in 4-ml tubes for transport back to the laboratory in a cool box.
We successfully collected targeted consumer groups from all floodplains, except for lumbriculid earthworms and tipulid dipterans (Table S3). We also obtained four of five targeted resources from all floodplains (Table S3). The exception was biofilm from the adjacent river waters: in two reference and two treated floodplains, there was neither Phragmites nor other suitable water plants available for epiphytic biofilm collection.
Unfortunately, we were unable to obtain A. sticticus during our two field sampling visits. There was no extensive mosquito emergence during our study visits, which were conducted in between flood events when standing water on the flood plains was minimal. However, the BMCP were able to provide us with 12 adult A. sticticus hatched from mature (pre-pupal) larvae. The larvae were collected from temporary standing water habitats at one of our sampling areas (Ista) during an outbreak later in the season. Four of the reared adults were sent for analyses of stable isotopes, and the remainder were allocated for FA analyses. The isotope and PUFA composition of these individuals fully reflect resources consumed during larval development in their natal floodplain habitats, as they were newly emerged and had not fed since collection as mature larvae. Finally, the BMCP provided us with a sample of the control product VectoBac®, which we analysed for stable isotopes.
Stable isotope and fatty acid analyses
Following field work and subsequent laboratory extraction, collected consumer and resource samples were separated into the taxonomic and resource groups described above. After 24–48 h of cold storage, during which invertebrates were not supplied with food, all samples were snap frozen in liquid nitrogen (− 196 °C) and stored in a freezer at − 40 °C. At the time of freezing, all organisms except the staphylinid beetles were alive. After freeze drying, samples were weighed and packaged into 5 × 9 mm tin capsules. Larger animals and plants were homogenised prior to weighing to obtain a representative sub-sample. Target weights were 1.3 mg for invertebrates, 3.5 mg for plants and 6.0 mg for soil. For filters with biofilm, ten pieces of 4 mm discs were clipped out per filter with a hole puncher and stacked in tin capsules. Three samples with blank filters were weighed and sent for analysis, to obtain isotopic values for the filters themselves. Three replicates of each soil and terrestrial plant type were also analysed from each floodplain. Unfortunately, we had insufficient material to repeat this process for the biofilm samples.
Stable isotopes have long been used to provide a picture of an organism’s ecological niche, where enrichment in the 15N isotope indicates a higher trophic position, and variation in the 13C isotope indicates whether the basal resource has aquatic or terrestrial origins (e.g. Hoekman et al. 2012; Kupilas et al. 2016). Stable isotopes have been recommended as a tool in evaluation of whole-ecosystem effects of mosquito control using Bti (Boisvert and Boisvert 2000), but remain little utilised (but see Allgeier et al. 2019a). Our samples were analysed for stable isotopes by the Stable Isotope Facility at the University of California, Davis, as described in the supplementary online file S1.
Fatty acids (FA) that are specific for certain algal groups and conservatively incorporated into the tissues of consumers can be used as biomarkers, and certain polyunsaturated fatty acids (PUFA) are considered essential for the growth and development of consumers (Goedkoop et al. 2007; Müller-Navarra et al. 2000). We analysed our adult mosquitos their fatty acid composition following the protocol of Lau et al. (2014), as described in the supplementary online file S1.
Data analysis
Stable isotope data were first evaluated visually, using isotopic biplots and niche space estimation. Isotopic biplots, produced with the R package siar (siar: stable isotope analysis in R, Parnell and Jackson (2013)), allow for a visual assessment of the similarity of potential food sources to different groups of consumers. In the production of these isotope biplots, data from the five control areas and from the five Bti-treated areas, respectively, were merged. This resulted in two charts that summarise data for the replicate treated and reference floodplains.
We used the Bayesian framework developed by Jackson et al. (2011) to characterise differences in the dimensions of niche space occupied by consumers (i.e. excluding resources) between treated and reference floodplains. Use of a Bayesian framework facilitates estimation of community wide metrics that account for uncertainty in the isotopic composition of particular community members by incorporating information on their variability. Here, we used individual floodplains within Bti treatment categories as replicates for estimating uncertainty. Consequently, for all niche-related metrics, one value is derived for treated and another for untreated floodplain communities, together with a probabilistic distribution (the posterior) representing the range of possible estimates of the metric.
First, we plotted convex hulls for treated and floodplain communities to visually assess how much and in what direction the communities diverge from one another in the isotopic biplot. We then applied Jackson et al’s (2011) Bayesian framework to estimate the niche metrics proposed by Layman et al. (2012):
-
1)
Ranges of δ13C and δ15N in the niche space (two metrics: CR and NR, respectively)
-
2)
Total area (TA) of the convex hull, acting as a measure of the total amount of niche space occupied by the community.
-
3)
Mean distance to the centroid of points in two-dimensional space (CD), which acts as a measure of species spread;
-
4)
Mean nearest neighbor distance in two-dimensional space (MNDD), functioning as a measure of density of species packing;
-
5)
Standard deviation of nearest neighbor distance (SDNND), measuring evenness of species packing in two-dimensional space;
Overlap in posterior probability distributions was used to assess the percent probability that each Layman metric differed between reference and treated floodplains (Jackson et al. 2011). A probability value greater than 95% provides strong support for divergence. Uncertainty in each Layman metric estimate is visualised as credible intervals. All Layman metrics, convex hull estimates and probabilities of divergence between floodplains were generated using the SIBER package version 2.1.6 (Jackson et al. 2011) implemented in R version 4.1.1 (R core team, Vienna Austria, http://www.R-project.org/).
We supported our descriptions of isotopic niche space by analysing differences in isotope composition of consumers and resources between treated and reference floodplains (Table S3) using two-sided, paired t tests (JMP version 11.0, SAS institute). Two-sided t tests were followed by one sided t tests to further evaluate the direction (increase or decrease) of change.
Results
Isotopic biplots
The most marked difference in the isotopic biplot between floodplain types was the presence of corn-containing VectoBac®-G in the treated floodplains, which had far higher (less negative) δ13C values than the in situ resources (Fig. 2). Aquatic biofilm samples were characterised by lower (more negative) δ13C than the terrestrial plant samples in the reference but not in treated floodplains. Soil δ13C was also lower than all terrestrial plant samples. The large error bar for meadow algae δ13C reflects the wide range, from − 25.41 to − 33.13, of the replicates obtained. The δ13C of the four A. sticticus individuals analysed varied from − 27.3 to 28.71, spanning the range of most other detritivores. Finally, detritivore consumers, comprising worms, springtails, oribatid mites and semi-aquatic dipterans, all had lower δ15N values in the biplot space than the predominantly predacious groups, including predacious mites, spiders and beetles (Fig. 2).
Bayesian Layman metrics and niche ellipses
The convex hulls for the treated and reference floodplain communities (Fig. 3) overlapped and were similarly sized (Table 1). However, the reference hull was more spread out along the δ13C axis (Fig. 3a-b), with the treated community hull more spread out along the δ15N (Fig. 3a). Isotopic niche ellipses for individual floodplains are given in Supplementary Information, Fig. S1.
Amongst the Layman metrics, the range of δ15N in the niche space (NR) was 56% higher in treated compared with reference floodplains, with the probability of divergence greater than 95% (Table 1, Fig. 4a). Probabilities of divergence in the δ13C range (CR), total area of the convex hull (TA) and mean distance to the centroid (CD) were all > 75%, with trends for 31% lower CR, 39% higher TA and 18% higher CD in treated relative to reference floodplains (Table 1, Fig. 4b, d). Probabilities of divergence in the mean and standard deviation of nearest neighbor distance (MNND and SDNND) were less than 70% and 35%, respectively (Table 1, Fig. 4e, f).
Bayesian Layman metric modes plotted for each reference and treated floodplains with 95% credible intervals: ranges of a δ15N (NR) and b δ13C (CR) in the niche space, c total area (TA) of niche space occupied, based on convex hulls; d species spread, based on mean distance to centroid (CD); e species packing, based on mean nearest neighbor distance (MNDD); (f) evenness of species pacing, based on standard deviation of nearest neighbor distance (SDNND). Black dots represent the modal estimate, whilst shading in the density plot represents the 50% (dark gray), 75% (light gray) and 95% (white) credibility intervals. See Table 1 for probability of divergence estimates between wetland types
Consumers and resources
Values of δ15N were 97% higher for detrivorous oribatid mites (Fig. 5a) in treated compared with reference floodplains (Table 2). A similar trend for the pisaurid spider Dolomedes fimbriatus (9% higher in treated compared with untreated floodplains, Fig. 5a) was near significance at the 5% level in the two-sided t test (Table 2). Values of δ13C were 3.5% higher in reference than treated floodplains for Enchytraeidae (Fig. 5b, Table 2). There were several additional trends for lower δ13C and/or higher δ15N in other key consumer taxa in treated compared with reference floodplains (Fig. 5, Supplementary material Fig. S1), but these were either not significant in two-sided t tests, or could not be evaluated due to a lack of replication (Lumbricidae, Tipulidae) across all floodplains (Table S3).
Differences in mean ± 1 SE values for δ13C (a) and δ15N (b) of key consumer taxa in treated (orange) and reference floodplains (blue). Significance levels from two-sided paired t tests of differences (Table 2) indicated: *p < 0.05, •p < 0.1
Resources obtained from all sample sites (i.e. soil, meadow biofilm scrapes, Carex sp. and Cirsium palustre, Table S1) did not differ in either δ13C or δ15N values between treated and reference floodplains (all t <|1.3|, p > 0.3). The lowest C:N molar mass ratios were observed for A. sticticus (5.43 ± 0.51), and lake (9.44 ± 1.51) and meadow (12.07 ± 2.95) biofilms (Fig. 6). VectoBac®-G had the highest C:N ratio (126.7 ± 19.86, Fig. 6). C:N ratios were higher in treated floodplains for Carex sp. (27% higher; t = 4.43, p = 0.011). No other differences in C:N ratios between floodplain types were detected for the remaining resource types (all t <|1.4|, p > 0.23).
C:N molar mass ratios of seven resources found in the reference (blue/dark bars) and treated (orange/light bars) floodplains, ordered from lowest to highest. Biofilm, soil and plant samples were all collected directly from the floodplains, and the mean ± SE of these resources is calculated at the floodplain level. A. sticticus were sampled prior to pupation from one of the treated floodplains only (Ista mire), whilst the Vectobac®-G sample represents the product used for mosquito control in the floodplains. The mean ± SE for the A. sticticus and Vectobac®-G are calculated based on three and five analytical replicates. Significant differences between floodplain types based on paired t tests indicated: *p < 0.05
Fatty acid composition of A. sticticus
We detected 23 fatty acids (FA) with relative concentrations exceeding 0.05% in adult A. sticticus reared from field-collected fourth instar larvae (Table S3). The most abundant FA were the monounsaturated oleic acid (18:1ω9) and hypogeic acid (16:1ω9), and saturated FA palmitic acid (16:0), which accounted for 19–27.4% of total FA. Amongst PUFAs, eicosapentaenoic acid (20:5ω3, EPA) and arachidonic acid (20:4ω6) and their precursors α-linolenic acid (18:3ω3, ALA) and linoleic acid (18:2ω6, LA), respectively, comprised between 2.4 and 4.9%. Finally, adult A. sticticus also had detectable levels of docosahexaenoic acid (22:6ω3, DHA), i.e. 0.11% (Table S3).
Discussion
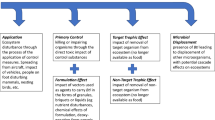
Our isotopic analyses of key invertebrate consumers indicate that more than a decade of mosquito control in the lower Dalälven using a granular formulation of Bti is causing significant changes in the isotopic composition of key organism groups feeding at the base of floodplain food webs. Enchytraeid worms were characterised by significantly higher δ13C values in treated relative to reference floodplains, and oribatid mites were significantly higher in δ15N values. Similar trends that were near significance were apparent in predacious staphylinid beetles and the raft spider D. fimbriatus. Finally, our Bayesian analyses of isotopic niche metrics indicate a 96.5% and 79% probability of divergence between floodplain types in the ranges of community δ15N and δ13C values. Variation in key soil characteristics (soil types, depth, isotopic signature) and the composition of ground cover amongst our floodplains were minimal, and did not differ systematically between floodplain types. Accordingly, we conclude these observed food web shifts are a consequence of the repeated, large-scale applications of VectoBac®-G in the treated floodplains, i.e. 3–5 times annually during all but 2 years over the period 2002–2014. Ecological mechanisms potentially explaining these results include (1) incorporation of the high δ13C value corn particles, used as a vector in the VectoBac®-G product, into floodplain food webs, and (2) the mass mortality of A. sticticus, which results in high concentrations of dead larvae deposited on floodplain soils at local scales.
In the absence of mosquito control, the vast majority of A. sticticus larvae will emerge as adults, and subsequently disperse in search of mates and (in the case of females) blood meals (Fig. S2). Those that escape predation can then be expected to die after swarming (for males) and oviposition (which occurs in moist soils), with their corpses being distributed over a wide geographic area. Dispersal distances > 11 km have been documented for female A. sticticus (Mirta et al. 2011). In contrast, under a scenario of repeated Bti treatment events, adult abundances are reduced by 90–100% in the lower River Dalälven (Schäfer and Lundström 2014), thus preventing wider dispersal of mosquito biomass (Fig. S3). Instead, dead larval biomass remains concentrated in the remaining, temporary, standing water habitats after the floodwater recedes (Fig. S2). This dead biomass likely enters soil- and ground-based food webs, either as a result of scavenging from detritivores, or as a result of microbial degradation (Gratton et al. 2017; Hoekman et al. 2012). Our results indicate that dead A. sticticus larvae represent a labile and relatively nutrient-rich resource, characterised by a low C:N ratio and moderate concentrations of long-chain PUFAs (Table S4). When made available, such high-quality food resources are preferentially consumed by soil detritivores, altering their isotopic composition (Scheu and Schaefer 1998; Tiunov and Scheu 2004). The dead A. sticticus larvae accumulating in floodplains might also be attractive to predators and/or scavengers, including oribatid mites and staphylinid beetles (Schneider et al. 2004; von Berg et al. 2012).
The ground corn particles used in VectoBac®-G also represent an additional C input to the epigeal and soil-based food webs of the treated floodplains. VectoBac®-G has an extremely high C:N ratio, similar to that of some of the most nutrient-poor, naturally occuring, plant litter species (Frainer et al. 2015; Handa et al. 2014). However, the percentage of C in corn cob comprised of refractory lignin is relatively low, varying from c. 5–15% depending on growing conditions (Demirbaş 2005; Mourtzinis et al. 2014; Pointner et al. 2014). This is similar to the lignin content of those plant litter species categorised as “fast-decomposing” by Handa et al. (2014), and is also at the lower end of the range observed previously for floodplain herbaceous vegetation (9.6–29.4%, Britson et al. 2016). Furthermore, the VectoBac®-G granules also incorporate an unknown quantity of corn oil, used to aid attachment of Bti (Lundstrom et al. 2010), which might enhance the resource quality of the particles. Thus, although nutrient-poor compared with dead A. sticticus and meadow biofilms, the C characterising the corn substrate used in VectoBac®-G is likely to be relatively labile for decomposing organisms inhabiting a soil matrix that is otherwise rich in highly refractory (Curry and Schmidt 2007).
Carbon characteristics of resources and consumers
Freshwater biofilms generally have lower δ13C values than terrestrial plants, reflecting the higher presence of light 12C isotopes taken up during algal photosynthesis (Marty and Planas 2008). However, our biofilm samples were not as 13C-depleted as expected, especially in the treated floodplains, given that clean algal samples often have δ13C values < − 30 (Marty and Planas 2008). It is, thus, likely that our aquatic biofilm samples also included particulate C of terrestrial origin. This “contamination” is a common problem in field-based aquatic isotope studies (Kupilas et al. 2016; Marty and Planas 2008). Interestingly, soil δ13C values were on average 4% more negative than those of terrestrial plants. This likely reflects the long-term influence of flooding on the soils of the wet meadows. Each time the floodwaters recede, carbon derived from aquatic primary producers and thus depleted in 13C is deposited on the floodplain (e.g. as stranded algae or submerged aquatic plants), thus reducing soil δ13C (Hein et al. 2003; Tockner et al. 1999). Finally, our analyses confirm that the corn used as a substrate in the Bti control agent VectoBac®-G consitutes a unique C-source for the River Dalälven floodplains, with δ13C values that are substantially higher than any other analysed resource.
Carbon isotopes of most consumers were similar to or lighter than the δ13C values of aquatic biofilm, and did not closely match with any of the terrestrial resources. This provides further support for the importance of C derived from aquatic primary producers in these floodplain food webs, and points towards the capacity of organisms to select for C of aquatic origin. Aquatic derived C is typically more palatable (i.e. characterised by more labile C compounds, lower amounts of secondary plant compounds and higher nutrient content) than that arising from terrestrial leaf litter. Indeed, Lau et al. (2014) showed that autochthonous resources are the main driver of secondary production even in dystrophic lakes, as algae supply consumers with fatty acids that are essential for consumer growth and reproduction. The higher palatability of aquatic-derived resources to terrestrial consumers is further seen in the low C:N ratios of lake biofilms. As expected, dead A. sticticus also had a low C:N ratio, which is likely to make it attractive to decomposers and scavengers alike.
The δ13C of newly emerged A. sticticus adults was highly variable amongst individuals, spanning the full range of terrestrial resources. Our PUFA analyses confirm A. sticticus consumes a mixed diet, with important long-chain PUFAs synthesised by algae (e.g. EPA 20:5ω3) making up around 5% of total FA, and the bacterial indicator FA vaccenic acid (18:1ω7) making up 2%. This is in line with values reported by Sushchik et al. (2013) for mosquito adults in floodplains in the Novosibirsk region in Russia, and those for detritivorous benthic invertebrates from boreal lakes, such as the isopod Asellus aquaticus (Lau et al. 2012). Finally, docosahexaenoic acid (DHA) is common in aquatic invertebrate predators (e.g. Lau et al. (2012)), and suggests that A. sticticus also preys on smaller invertebrates. Overall, these results suggest that much of the C ingested by the mosquitoes in their temporary floodplain habitats is terrestrial in origin, obtained through filtration of bacteria colonising particulate C (Merritt et al. 1992). However, the PUFA and isotope results indicate this diet is patchily supplemented with freshwater algal productivity, likely reflecting an uneven deposition of algal cells over the floodplains during flooding events (Hein et al. 2003; Tockner et al. 1999).
Consumer isotopes: comparing treated and reference floodplains
Transitions between trophic levels typically involve a δ15N increase of about 3‰ (from 2.2 to 3.4 ‰) (Vander Zanden and Rasmussen 2001). Accordingly, our isotopic analyses suggest our sampled invertebrates spanned at least three trophic levels. The overall increase in the range of δ15N values characterising consumers in the treated floodplains suggests that repeated applications of VectoBac®-G have increased the amount of fractionation involved in nutrient transfer through floodplain food webs, and hence increased food chain lengths. The marked increase in δ15N values of oribatid mites suggests that some of this extra fractionation occurs at the base of these food webs, possibly mediated by protozoans and other micro/meiofauna that feed on bacteria and decaying organic matter (Griffiths 1994). The use of Bti has led to an increase in single-celled protozoans in soils of Dalälven floodplains (Östman et al. 2008). This result was attributed to reduced feeding by mosquito larvae on these organisms (Östman et al. 2008), but might also reflect the ready availability of dead mosquito larvae for protozoans to colonise. We, thus, hypothesise that the observed additional N-fractionation for the oribatid mites in treated floodplains reflects an increased transfer of N from decaying mosquito larvae to higher trophic levels, mediated at least in part through protozoans.
Enchytraeid worms were significantly higher in δ13C in treated relative to reference floodplains. We sampled Enchytraeidae from the moist depressions in between grassy tussocks characterising our floodplains. These are the habitats where standing water is likely to persist longest after the floodwaters have receded, and hence are where both dead mosquito larvae and the corn particles comprising the bulk of the VectoBac®-G product are most likely to accumulate. Enchytraeidae are one of the main detritivore groups feeding on particulate C and its associated microbes in moist soils (Didden 1993), and are capable of searching out patches of more labile C within a soil matrix otherwise characterised by highly refractory C (Curry and Schmidt 2007). Accordingly, the increase in enchytraeid δ13C values in the treated floodplains might be especially attributable to consumption of the corn particles, given these are substantially richer in 13C values than any other floodplain resource. The trend for an overall reduced δ13C range in consumers of treated floodplains suggests a shift from consumption of a broader diversity of C sources towards the readily available C delivered in the form of the VectoBac®-G corn particles, and/or the abundant C introduced locally by the mass morality of larval mosquitoes. However, more research is required to confirm the evidence of divergence in not only the δ13C range but also total isotopic niche area (77% probability) and species spread (79% probability) between floodplain types observed in our study.
Quantities of VectoBac®-G compared with possible abundances of dead mosquito larvae
We are unable to definitively distinguish whether the shifts in the isotopic characteristics of soil- and ground-dwelling food webs between treated and untreated floodplains of the lower Dalälven are primarily attributable to the labile C introduced with VectoBac®-G, or the prevention of A. sticticus emergence and the subsequent accumulation of dead larvae on floodplain soils. An approach for separating these mechanisms is the use of isotopic mixing models for quantifying the contribution of different resources in consumer biomass (Layman et al. 2012; Phillips et al. 2014). However, isotopic mixing models are prone to strong biases when the isotopic range of potential resources falls well outside the niche space of consumers (Phillips et al. 2014), as is clearly the case with the corn particles comprising Vectobac-G. Nevertheless, the question of the impact of the corn particles is non-trivial, since alternative Bti products are available that do not use corn as the substrate. Here, we explore the potential for both mechanisms to change the isotopic composition of resources in floodplain soils using data on the C and N content of the corn granules and mosquitoes, respectively.
Our analyses reveal that VectoBac®-G consists on average of approximately 50% C (500 g/kg) and 0.5% (5 g/kg) N. At the typical application rate of 15 kg/ha (Schäfer and Lundström 2014), the spreading of VectoBac®-G in the lower Dalälven floodplains adds 7050 gC/ha, and 75 gN/ha per treatment event, all in a relatively labile, albeit nutrient poor, form. These figures accumulate with repeated applications in 1 year. After ten applications, 75 kg of C and 0.75 kg of N will have been added per hectare (though the final fate of these particles will depend on the influence of subsequent winds, rainfall and flooding). Estimates of mosquito biomass in treated and reference floodplains are more challenging, since we lack data on mosquito production per hectare. In one of the most extreme mass emergence years (2000), some 192,000 individual female mosquitoes were caught from June–August in carbon dioxide traps covering 99 ha of floodplains area. According to our analyses, an adult A. sticticus contains on average 0.416 mg C and 0.04 mg N. Upscaling these figures and assuming a sex ratio of 1:1 (Lounibos and Escher 2008), we estimate that the mosquito biomass captured in the traps in 2000 represented approximately 0.64 gC/ha and 0.14 gN/ha. This is, of course, an underestimate of emerging mosquitos, given that CO2 traps only capture a fraction of all flying adults and are particularly inefficient at attracting males (McPhatter and Gerry 2017). If we assume that similar-size populations of mosquitoes instead had been killed as larvae, this C and N would remain concentrated in temporary pools in treated floodplains. There are clearly multiple sources of uncertainty in these calculations, including the lack of data on abundances of males, and uncertainties in the sampling efficiency of the carbon dioxide traps. Nevertheless, these figures serve to demonstrate the potential impact of dead mosquitoes on soil- and ground-based food webs compared with that of the distribution of the corn particles in VectoBac®-G. Ultimately, it is likely that the two effects are partially linked, as consumers that feed on dead mosquito larvae will also consume carbon from any VectoBac®-G remaining in the mosquito’s digestive tract.
Conclusion
The findings of our spatially replicated field study point towards changes in the trophic structure of the soil- and ground-based food webs in the floodplains of the lower Dalälven, associated with the long-term, repeated applications (2002–2014) of the mosquito control agent VectoBac®-G. In a previous aquatic mesocosm experiment, Bti application as a suspension resulted in an increase in isotopic niche areas of predatory dragonflies (Allgeier et al. 2019a). In contrast, the effects of Bti on our terrestrial, floodplain communities were clearest at the base of the food web. This is exemplified by the increased δ13C and δ15N values of detrivorous enchytraeid worms and oribatid moss mites, respectively. Additionally, the increased δ15N range characterising treated floodplain consumer communities indicates an increase in the number of trophic fractionation steps involved in the processing of nitrogen. We conjecture that the increase in the overall δ15N range in the treated wetlands arises from an extra trophic step involved in decomposition of the dead mosquito larvae, which is likely to be mediated not only by the organism groups studied here, but also by other key organisms involved in the processing of detritus (e.g. protozoans, nematodes and microorganisms). In contrast, the increase in the δ13C of enchytraeid worms might be more likely to reflect integration of the more palatable C originating from the corn particles composing the VectoBac®-G product, into soil- and ground-based food webs. Further research is required to assess these hypotheses.
The potential ecological significance of our observed shifts in isotopic values of some key consumer groups can be assessed by comparing with previous studies of terrestrial and other invertebrates living in close association with aquatic ecosystems. Riparian spider δ15N values were 9% higher in an agricultural compared with forested catchment in New Zealand (Collier et al. 2002), and river restoration was associated with 7–11% increases in δ15N values of riparian spiders and beetles (Kupilas et al. 2020). More marked 70–400% shifts in δ15N values were observed in detritivorous millipedes, omnivorous beetles and predatory spiders living adjacent to rivers with marine-salmon-derived nutrient enrichment, relative to references (Hocking and Reimchen 2002; Rammell et al. 2021). Finally, 50–150% increases in δ15N values were observed in benthic macroinvertebrates along gradients of increasing anthropogenic nutrient enrichment in Sweden (Bergfur et al. 2009; Frainer and McKie 2015), similar to observations elsewhere (Clapcott et al. 2010; Smucker et al. 2018). Differences in δ13C values in the same studies ranged less widely, from 0 to 9% (anthropogenic nutrient enrichment, Bergfur et al. 2009; Frainer and McKie 2015), 0–4% (marine derived nutrients, Hocking and Reimchen 2002; Rammell et al. 2021) and 0–1% (river restoration, Kupilas et al. 2020). Collier et al (2002) observed a more marked 28–31% difference in δ13C values between an agricultural compared with forested catchment in New Zealand, which is attributable to strong differences in tree cover that were not a factor in our study. Finally, Tiunov and Scheu (2004) found that enrichment of soil with refractory C (glucose) increased earthworm δ13C values by 7%.
Accordingly, the 97% increase in δ15N values observed in oribatid detritivore mites and the 3.5% increase in δ13C observed for enchytraeid worms, fall within the mid-range of shifts observed previously in studies of key environmental drivers on riparian and floodplain invertebrate consumers. Furthermore, isotopic shifts in the range observed here have previously been linked to altered nutrient and energy flows, including across habit boundaries (Hladyz et al. 2011; Hocking and Reimchen 2002; Kupilas et al. 2016, 2020). On the other hand, statistical support for divergence in the isotopic characteristics of most other consumer groups (i.e. apart from Enchytraeidae and Oribatida) was weak, and the isotopic niche space occupied by our untreated and treated floodplain communities substantially overlapped. Thus, we suggest our results are best characterised as providing evidence that the food webs of our treated floodplains might be in transition, with effects of mosquito control manifesting unevenly amongst organism groups, trophic levels and floodplains. Further research with a greater level of replication and after more years of VectoBac®-G treatment is, therefore, required to establish how extensive and established the shifts observed here become, and to evaluate the potential wider implications for nutrient and energy cycles within floodplain ecosystems and linkages with adjacent habitats.
Data availability
The datasets used and/or analysed during the current study are available as supplementary material.
Code availability
Not applicable.
Change history
02 March 2023
A Correction to this paper has been published: https://doi.org/10.1007/s00027-023-00951-1
References
Allgeier S, Friedrich A, Brühl CA (2019a) Mosquito control based on Bacillus thuringiensis israelensis (Bti) interrupts artificial wetland food chains. Sci Total Environ 686:1173–1184. https://doi.org/10.1016/j.scitotenv.2019.05.358
Allgeier S, Kästel A, Brühl CA (2019b) Adverse effects of mosquito control using Bacillus thuringiensis var. israelensis: Reduced chironomid abundances in mesocosm, semi-field and field studies. Ecotoxicol Environ Saf 169:786–796. https://doi.org/10.1016/j.ecoenv.2018.11.050
Armitage P, Cranston PS, Pinder LCV (eds) (1994) Chironomidae: biology and ecology of non-biting midges. Chapman and Hall, London
Becker N et al (2010) Mosquitoes and their control. Springer-Verlag, Berlin
Bergfur J, Johnson RK, Sandin L, Goedkoop W (2009) Effects of nutrient enrichment on C and N stable isotope ratios of invertebrates, fish and their food resources in boreal streams. Hydrobiologia 628:67–79. https://doi.org/10.1007/s10750-009-9746-4
Boisvert M, Boisvert J (2000) Effects of Bacillus thuringiensis var. israelensis on target and nontarget organisms: a review of laboratory and field experiments. Biocontrol Sci Tech 10:517–561
Britson A, Wardrop D, Drohan P (2016) Plant community composition as a driver of decomposition dynamics in riparian wetlands. Wetlands Ecol Manage 24:335–346. https://doi.org/10.1007/s11273-015-9459-6
Brühl CA et al (2020) Environmental and socioeconomic effects of mosquito control in Europe using the biocide Bacillus thuringiensis subsp. israelensis (Bti). Sci Total Environ 724:137800. https://doi.org/10.1016/j.scitotenv.2020.137800
Carlson PE, McKie BG, Sandin L, Johnson RK (2016) Strong land-use effects on the dispersal patterns of adult stream insects: implications for transfers of aquatic subsidies to terrestrial consumers. Freshw Biol 61:848–861
Clapcott JE, Young RG, Goodwin EO, Leathwick JR (2010) APPLIED ISSUES: exploring the response of functional indicators of stream health to land-use gradients. Freshw Biol 55:2181–2199. https://doi.org/10.1111/j.1365-2427.2010.02463.x
Collier KJ, Bury S, Gibbs M (2002) A stable isotope study of linkages between stream and terrestrial food webs through spider predation. Freshw Biol 47:1651–1659
Curry JP, Schmidt O (2007) The feeding ecology of earthworms - a review. Pedobiologia 50:453–477
Demirbaş A (2005) Estimating of structural composition of wood and non-wood biomass samples. Energy Sources 27:761–767. https://doi.org/10.1080/00908310490450971
Didden W et al (1995) The relative efficiency of wet funnel techniques for the extraction of Enchytraeidae. Pedobiologia 39:52–57
Didden WAM (1993) Ecology of terrestrial Enchytraeidae. Pedobiologia 37:2–29
Frainer A, Jabiol J, Gessner MO, Bruder A, Chauvet E, McKie BG (2015) Stoichiometric imbalances between detritus and detritivores are related to shifts in ecosystem functioning. Oikos 125:861–871
Frainer A, McKie BG (2015) Shifts in the diversity and composition of consumer traits constrain the effects of land use on stream ecosystem functioning. Adv Ecol Res 52:169–200
Goedkoop W, Demandt M, Ahlgren G (2007) Interactions between food quantity and quality (long-chain polyunsaturated fatty acid concentrations) effects on growth and development of Chironomus riparius. Can J Fish Aquat Sci 64:425–436. https://doi.org/10.1139/f07-016
Gratton C, Hoekman D, Dreyer J, Jackson RD (2017) Increased duration of aquatic resource pulse alters community and ecosystem responses in a subarctic plant community. Ecology 98:2860–2872. https://doi.org/10.1002/ecy.1977
Griffiths BS (1994) Microbial-feeding nematodes and protozoa in soil: their effectson microbial activity and nitrogen mineralization in decomposition hotspots and the rhizosphere. Plant Soil 164:25–33
Handa IT et al (2014) Consequences of biodiversity loss for litter decomposition across biomes. Nature 509:218–221
Hein T, Baranyi C, Herndl GJ, Wanek W, Schiemer F (2003) Allochthonous and autochthonous particulate organic matter in floodplains of the River Danube: the importance of hydrological connectivity. Freshw Biol 48:220–232
Hershey AE, Lima AR, Niemi GJ, Regal RR (1998) Effects of Bacillus thuringiensis israelensis (Bti) and methoprene on nontarget macroinvertebrates in Minnesota wetlands. Ecol Appl 8:41–60
Hladyz S, Åbjörnsson K, Giller PS, Woodward G (2011) Impacts of an aggressive riparian invader on community structure and ecosystem functioning in stream food webs. J Appl Ecol 48:443–452
Hocking MD, Reimchen TE (2002) Salmon-derived nitrogen in terrestrial invertebrates from coniferous forests of the Pacific Northwest. BMC Ecol 2:4. https://doi.org/10.1186/1472-6785-2-4
Hoekman D, Bartrons M, Gratton C (2012) Ecosystem linkages revealed by experimental lake-derived isotope signal in heathland food webs. Oecologia 170:735–743
Hoekman D, Dreyer J, Jackson RD, Townsend PA, Gratton C (2011) Lake to land subsidies: experimental addition of aquatic insects increases terrestrial arthropod densities. Ecology 92:2063–2072
Holmstedt S, Linderheim A (2019) Färnebofjärden: the national park of lower Dalälven (Färnebofjärden : nationalparken vid Nedre Dalälven). Votum & Gullers, Örebro
Jackson AL, Inger R, Parnell AC, Bearhop S (2011) Comparing isotopic niche widths among and within communities: SIBER–Stable Isotope Bayesian Ellipses in R. J Anim Ecol 80:595–602
Kupilas B, Friberg N, McKie BG, Jochmann MA, Lorenz AW, Hering D (2016) River restoration and the trophic structure of benthic invertebrate communities across 16 European restoration projects. Hydrobiologia 769:105–120
Kupilas B, McKie BG, Januschke K, Friberg N, Hering D (2020) Stable isotope analysis indicates positive effects of river restoration on aquatic-terrestrial linkages. Ecol Ind 113:106242. https://doi.org/10.1016/j.ecolind.2020.106242
Land M, Bundschuh M, Hopkins RJ, Poulin B, McKie BG (2019) What are the effects of control of mosquitoes and other nematoceran Diptera using the microbial agent Bacillus thuringiensis israelensis (Bti) on aquatic and terrestrial ecosystems? A systematic review protocol. Environ Evid 8:32. https://doi.org/10.1186/s13750-019-0175-1
Lau DC, Sundh I, Vrede T, Pickova J, Goedkoop W (2014) Autochthonous resources are the main driver of consumer production in dystrophic boreal lakes. Ecology 95:1506–1519
Lau DCP, Vrede T, Pickova J, Goedkoop W (2012) Fatty acid composition of consumers in boreal lakes – variation across species, space and time. Freshw Biol 57:24–38. https://doi.org/10.1111/j.1365-2427.2011.02690.x
Layman CA et al (2012) Applying stable isotopes to examine food-web structure: an overview of analytical tools. Biol Rev 87:545–562. https://doi.org/10.1111/j.1469-185X.2011.00208.x
Lounibos LP, Escher RL (2008) Sex ratios of mosquitoes from long-term censuses of Florida tree holes. J Am Mosq Control Assoc 24:11–15. https://doi.org/10.2987/5656.1
Lundstrom JO, Schafer ML, Petersson E, Vinnersten TZP, Landin J, Brodin Y (2010) Production of wetland Chironomidae (Diptera) and the effects of using Bacillus thuringiensis israelensis for mosquito control. Bull Entomol Res 100:117–125. https://doi.org/10.1017/s0007485309990137
Marty J, Planas D (2008) Comparison of methods to determine algal δ13C in freshwater. Limnol Oceanogr Methods 6:51–63. https://doi.org/10.4319/lom.2008.6.51
McPhatter L, Gerry AC (2017) Effect of CO2 concentration on mosquito collection rate using odor-baited suction traps. J Vector Ecol 42:44–50. https://doi.org/10.1111/jvec.12238
Merritt RW, Dadd RH, Walker ED (1992) Feeding behavior, natural food, and nutritional relationships of larval mosquitoes. Ann Rev Entomol 37:349–376
Mirta B, Enrih M, Tomislav B (2011) The flight distances of floodwater mosquitoes (Aedes vexans, Ochlerotatus sticticus and Ochlerotatus caspius) in Osijek, Eastern Croatia. Biologia 66:678–683. https://doi.org/10.2478/s11756-011-0073-7
Mourtzinis S et al (2014) Distribution of structural carbohydrates in corn plants across the Southeastern USA. BioEnergy Res 7:551–558. https://doi.org/10.1007/s12155-014-9429-5
Müller-Navarra DC, Brett MT, Liston AM, Goldman CR (2000) A highly unsaturated fatty acid predicts carbon transfer between primary producers and consumers. Nature 403:74–77
Östman Ö, Lundström JO, Vinnersten TZP (2008) Effects of mosquito larvae removal with Bacillus thuringiensis israelensis (Bti) on natural protozoan communities. Hydrobiologia 607:231–235
Parnell A, Jackson A (2013) siar: Stable Isotope Analysis in R. R package version 4.2. https://CRAN.R-project.org/package=siar
Petersen H (2002) General aspects of collembolan ecology at the turn of the millennium. Pedobiologia 46:24–260
Phillips DL et al (2014) Best practices for use of stable isotope mixing models in food-web studies. Can J Zool 92:823–835. https://doi.org/10.1139/cjz-2014-0127
Pointner M, Kuttner P, Obrlik A, Jäger A, Kahr H (2014) Composition of corncobs as a substrate for fermentation of biofuels. Agron Res 12:391–396
Poulin B, Lefebvre G, Paz L (2010) Red flag for green spray: adverse trophic effects of Bti on breeding birds. J Appl Ecol 47:884–889
Pyankov VI, Ziegler H, Akhani H, Deigele C, LÜTtge U, (2010) European plants with C4 photosynthesis: geographical and taxonomic distribution and relations to climate parameters. Bot J Linn Soc 163:283–304. https://doi.org/10.1111/j.1095-8339.2010.01062.x
Ramberg E et al (2020) The structure of riparian vegetation in agricultural landscapes influences spider communities and aquatic-terrestrial linkages. Water. https://doi.org/10.3390/w12102855
Rammell NF, Dennert AA-O, Ernst CA-O, Reynolds JA-O (2021) Effects of spawning Pacific salmon on terrestrial invertebrates: Insects near spawning habitat are isotopically enriched with nitrogen-15 but display no differences in body size. Ecol Evol 11:2045–7758
Schäfer M, Lundström JO (2014) Efficiency of Bti-based floodwater mosquito control in Sweden–four examples. J Eur Mosquito Control Assoc 32:1–8
Schäfer ML, Lundström JO (2009) The present distribution and predicted geographic expansion of the floodwater mosquito Aedes sticticus in Sweden. J Vector Ecol 34:141–147
Scheu S, Schaefer M (1998) Bottom-up control of the soil macrofauna community in a beechwood on limestone: manipulation of food resources. Ecology 79:1573–1585
Schneider K, Renker C, Scheu S, Maraun M (2004) Feeding biology of oribatid mites : a minireview. Phytophaga 14:247–256
Schneider S, Tajrin T, Lundström JO, Hendriksen NB, Melin P, Sundh I (2017) Do multi-year applications of Bacillus thuringiensis subsp. israelensis for control of mosquito larvae affect the abundance of B. Cereus group populations in riparian wetland soils? Microb Ecol 74:901–909. https://doi.org/10.1007/s00248-017-1004-0
Smucker NJ, Kuhn A, Cruz-Quinones CJ, Serbst JR, Lake JL (2018) Stable isotopes of algae and macroinvertebrates in streams respond to watershed urbanization, inform management goals, and indicate food web relationships. Ecol Ind 90:295–304. https://doi.org/10.1016/j.ecolind.2018.03.024
Sushchik NN, Yurchenko YA, Gladyshev MI, Belevich OE, Kalachova GS, Kolmakova AA (2013) Comparison of fatty acid contents and composition in major lipid classes of larvae and adults of mosquitoes (Diptera: Culicidae) from a steppe region. Insect Sci 20:585–600. https://doi.org/10.1111/j.1744-7917.2012.01582.x
Tiunov AV, Scheu S (2004) Carbon availability controls the growth of detritivores (Lumbricidae) and their effect on nitrogen mineralization. Oecologia 138:83–90
Tockner K, Pennetzdorfer D, Reiner N, Schiemer F, Ward J (1999) Hydrological connectivity, and the exchange of organic matter and nutrients in a dynamic river–floodplain system (Danube, Austria). Freshw Biol 41:521–535
Truchy A, Angeler DG, Sponseller RA, Johnson RK, McKie BG (2015) Linking biodiversity, ecosystem functioning and services, and ecological resilience: towards an integrative framework for improved management. Adv Ecol Res 53:55–96
Tullgren A (1971) En enkel apparat för automatisk vittjande av sållgods (A simple apparatur for automatic yield of samples) Entom. Tidskr 72:91–100
Vander Zanden JM, Rasmussen JB (2001) Variation in δ15N and δ13C trophic fractionation: Implications for aquatic food web studies. Limnnol Oceanog 46:2061–2066
Vaughan IP, Newberry C, Hall DJ, Liggett JS, Ormerod SJ (2008) Evaluating large-scale effects of Bacillus thuringiensis var. israelensis on non-biting midges (Chironomidae) in a eutrophic urban lake. Freshw Biol 53:2117–2128. https://doi.org/10.1111/j.1365-2427.2008.02043.x
von Berg K, Traugott M, Scheu S (2012) Scavenging and active predation in generalist predators: a mesocosm study employing DNA-based gut content analysis. Pedobiologia 55:1–5. https://doi.org/10.1016/j.pedobi.2011.07.001
Yang LH, Edwards KF, Byrnes JE, Bastow JL, Wright AN, Spence KO (2010) A meta-analysis of resource pulse-consumer interactions. Ecol Monogr 80:125–151
Acknowledgements
We are grateful to Peter Carlson for assistance in the field, and to Tryggve Persson for advice on collection techniques of Enchytraeidae. We thank Martina Schäfer and Jan Lundström of the Swedish Biological Mosquito Control Project for advice on study site location and providing us with mosquito and Bti material for analysis.
Funding
Open access funding provided by Swedish University of Agricultural Sciences. This research was funded through a grant from the Swedish Environmental Protection Agency (Grant Number NV-04371–14) to BGM and WG.
Author information
Authors and Affiliations
Contributions
BGM, WG and AT conceived and designed the study. AF and TN conducted field sampling and laboratory analysis. The data were analysed by BGM and AF. The first draft was prepared by BGM and WG. All authors contributed to draft revision.
Corresponding author
Ethics declarations
Conflict of interest
The authors declared that they have no conflict of interest.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this article was revised due to incorrect figure 3.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
McKie, B.G., Taylor, A., Nilsson, T. et al. Ecological effects of mosquito control with Bti: evidence for shifts in the trophic structure of soil- and ground-based food webs. Aquat Sci 85, 47 (2023). https://doi.org/10.1007/s00027-023-00944-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00027-023-00944-0