Abstract
The paper presents the results of research into toxic metal concentrations in the surface layer of bottom sediments in Lake Gopło. The research objectives were to identify the levels and spatial variability of Cu, Pb, Cd, Zn, Ni, Cr, As and Hg concentrations, their potential sources and the determinants of pollution levels. Metal contamination of the sediments was assessed using the geoaccumulation index (Igeo), pollution load index (PLI) and ecological risk index (RI). Chemometric methods (Pearson correlation, principal component analysis (PCA) and cluster analysis (CA) were used to determine the relationship between sampling sites and concentrations of toxic metals, thereby identifying the sources of contamination. The research found that grain-size composition, carbonate content and organic matter content in the bottom surface sediments of Lake Gopło are all characterised by low diversity. Therefore, the lithological features of the sediments are not a major factor in the concentrations and spatial variability of the metals. It was found that the metal concentrations in the great majority of samples were above regional geochemical background levels. The geochemical indices (Igeo, PLI, RI) indicate that the degree of toxic metal pollution in the sediments is slight in the central and southern parts of the lake and high in the northern part. The chemical analysis results showed that the samples in the central and southern parts of the lake differ little in their shares and concentrations of individual metals. This provides evidence that, as well as geogenic sources, their presence in sediments can be associated with non-point sources related to agricultural activities and with atmospheric sources (mainly the products of fossil fuel combustion). The higher concentrations of metals (especially Ni, Cd, Cr and Hg) in the northern part of the lake are influenced by the supply of industrial and communal pollutants from the lakeside town of Kruszwica. A factor limiting the migration of pollutants from the northern part of the lake towards the south is the lake’s morphology of the lake, which hinders water exchange between the northern part and the rest of the lake.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The toxic metal pollution of aquatic ecosystems is a global problem that results from increasing industrialisation, urbanisation, and the intensification and chemicalisation of agriculture (Foster and Charlesworth 1996; He et al. 2005; Häder et al. 2020; Xia et al. 2020). The changes caused by the industrial revolution, and especially the significant increase in extraction and processing of metal ores in the early twentieth century, were marked by, among other things, a significant increase in metal concentrations in soil profiles and sub-aqueous sediment cores (Nriagu 1996; De Vleeschouwer et al. 2009).
The toxic metal pollution of the environment, and especially of aquatic ecosystems, is dangerous because of the environmental persistence of toxic metals, their toxicity and their potential for biological accumulation (Varol 2011; Copat et al. 2012; Jiang et al. 2012; Singh and Kumar 2017; Zhao et al. 2017; Chen et al. 2018). Toxic metals pass through the environment in different ways to ultimately reach water bodies, where they accumulate in bottom surface sediments (Foster and Charlesworth 1996; Cai et al. 2015). Their path from source of emission to water body depends on the form in which they are introduced into the environment. Toxic metals can enter the hydrological network directly in soluble form (Wang et al. 2018; Wilson 2018). They can also be emitted to the atmosphere as dust or gases, and then fall to the surface as liquid or solid precipitation (Gunawardena et al. 2013; Sarkar et al. 2015; Chang and Yingxia 2020).
Toxic metals contained in soil, both those of anthropic origin and those that are a natural component of the substrate, participate in adsorption and desorption processes, which determine their bioavailability and mobility in the environment (Violante et al. 2010). They can then be leached by infiltrating precipitation water or pass in insoluble form with mineral–organic suspended matter and in surface runoff to reach the channels of watercourses and onwards to natural and artificial water bodies (Foster and Charlesworth 1996; Garrett 2000; Sharma 2001; Siegel 2002).
Toxic metal concentrations in bottom surface sediments are conditioned by volume of supply, but also by their physical properties and mineral composition, the dynamics of the aquatic environment, and the physico-chemical properties of the water (Förstner 1982; Dickinson et al. 1996). In this case, the sediment properties that are of fundamental importance in affecting the efficiency of sorption processes are: the grain size of clastic material; the mineral composition of the sedimenting suspension and the sediment; the organic matter and carbonate contents in the sediment material; and their concentration of Al, Fe, Mn and P compounds (Helios-Rybicka 1986, 1997; Foster and Charlesworth 1996; Kabata-Pendias and Pendias 1999; Brekhovskikh et al. 2001; Canavan et al. 2007). Large water bodies usually exhibit significant spatial differentiation in the metal content in the bottom surface sediment layer (Håkansson and Jansson 1983). This reflects not only the location of the sources supplying pollutants to the water body, but also the effects of internal hydrodynamic processes that cause both the re-suspension of accumulated sediment and post-depositional bioturbations (Zhang et al. 2014). These processes change Eh and pH values, potentially causing mobile metal forms to be released from bottom surface sediments into the water (Eggleton and Thomas 2004; Atkinson et al. 2007; Cantwell et al. 2008).
Previous studies indicate that the scale and spatial variability of toxic metal concentrations in the bottom surface sediments of water bodies are influenced by many factors and processes:
-
(1)
The environmental diversity of the catchment area (e.g. relief, substrate lithology, soil type), (Abraham et al. 1998; Wiechuła 2003; Wang et al. 2014; Sojka et al. 2019; Kostka and Leśniak 2021),
-
(2)
The form of land use (Lindström 2001; Skwierawski, Sidoruk 2014; Zemełka et al. 2019; Zeng et al., 2020),
-
(3)
The location and type of point pollution sources (Cobelo-Garcia, Prego 2004; Sojka et al. 2013; Wu et al. 2014; Smal et al. 2015; Kuriata-Potasznik et al. 2016),
-
(4)
Water body characteristics (shape, morphology, depth, hydrodynamics), (Smal et al. 2015; Zhu et al. 2017; Gierszewski 2018; Sojka et al. 2018; Sahoo et al. 2019),
-
(5)
Bottom surface sediment properties (grain sizes, mineral composition, organic matter content) (Foster and Charlesworth 1996; Kabata-Pendias and Pendias 1999; Tao et al. 2015; Lin et al. 2016; Gierszewski 2018; Kostka and Leśniak 2021),
-
(6)
Chemical and biochemical processes in bottom surface sediments (Salomons and Stigliani 1995; Zhang et al. 2014),
-
(7)
Climate change (Zhang et al. 2018).
Due to the multiplicity of these factors, analyses of the factors determining the variation in toxic metal contamination of bottom surface sediments should consider the specific features of individual water bodies and their catchments (Sojka et al. 2019).
Here, we present the results of studies on toxic metal concentrations in the surface layer of bottom surface sediments in Lake Gopło. It is one of the Poland’s largest lakes by surface area and lies in a region of highly developed agriculture and extensive agricultural-food industry. It also lies within a zone of influence of lignite mines and coal-fired power plants. Despite the lake's catchment area containing many sources of supply of hazardous pollutants, no comprehensive studies have yet been undertaken to identify the sources of supply of toxic metals or the spatial distribution of toxic metals in the bottom surface sediments, nor to assess pollution. Therefore, the aim of the research was: (i) to determine the spatial distributions of selected toxic metals (Cu, Pb, Cd, Zn, Ni, Cr, As and Hg) in surface sediments of Gopło Lake; (ii) to assess the degree of contamination of the toxic metals using indices of contamination; and (iii) to evaluate the relationship between toxic metals and define their possible sources (natural and/or anthropogenic) by the application of chemometric analysis. Chemometric methods such as Pearson's correlation analysis, principal component analysis (PCA) and cluster analysis (CA) are considered to be effective at discovering sources of contamination and have been used successfully in many studies of the contamination of bottom sediments with toxic metals (e.g. Loska and Wiechuła 2003; Larrose et al. 2010; Chabukdhara and Nema 2012). The authors’ use of a complementary approach that integrates chemometric analyses and geochemical methods increased the accuracy of the assessment of sediment contamination with metals and helped determine the sources of their supply.
Materials and methods
Study area
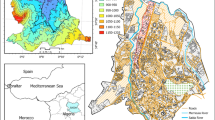
Located in the Kuyavian lake district (central Poland), Gopło Lake has an area of 21.5 km2, placing it ninth in Poland by area. It occupies a 25-km-long channel that splits into two arms in the middle of the lake. The shape of the lake and the presence of numerous islands and inlets mean that its shoreline is over 90 km long (Fig. 1). The lake depth is 16.6 m at its deepest point and averages 3.6 m. Its different depths mean that summer thermal stratification occurs only in its deepest, southern part (Goszczyński, Jutrowska 1996).
The relief of the lake’s 1342 km2 catchment area was formed during the Weichselian glaciation. It consists of morphologically diverse post-glacial moraine plateaus. In the northern part, these are mainly flat surfaces of sandy clay ground moraine. In the southern part, the relief is more diverse, consisting of an undulating moraine plateau, marginal hillocks and ridges, and sandy outwash plains. The moraine plateau is incised in many places by subglacial channels, the centrally located Gopło channel, and marginal valleys (Molewski 1999). The predominance of poorly permeable sediments limits the migration of pollutants to the groundwater, and the relatively low energy of the relief restricts surface runoff (Fig. 1). The dominant land-use type in the lake’s catchment area is agricultural, covering as much as 82.6% of its area. The share of other forms of land use is: 11.4% forests; 1.5% urban and industrial areas; 4.5% water and wetlands (Fig. 1). The lake area and adjacent farmland, meadows and pastures, forests, swamps, reeds and other wastelands constitute a protected area that functions partly as a landscape park and partly as a nature reserve (Gopło Millennium Landscape Park). It is also a Natura 2000 area (an area of EU nature protection; SPA PLB040004 Ostoja Nadgoplańska and SAC PLH040007 Jezioro Gopło, established pursuant to Council Directive 2009/147/EC on the conservation of wild birds (EU 2019) and Council Directive 92/43/EEC on the Conservation of natural habitats and of wild fauna and flora (EU 2013)). The toxic metal content in soils and sediments in the lake's catchment area does not exceed geochemical background values (Atlas Geochemiczny Polski 2012). Concentrations of Ni and Pb can be locally elevated (Cieśla et al. 1994).
The shallow depths of the lake, the large area of the bottom that lies within the range of the epilimnion, the large size of the catchment area relative to the lake area, and the catchment relief and land management all make Gopło Lake highly susceptible to the supply of pollutants from the catchment area and to the redistribution of autochthonous matter (Goszczyński and Jutrowska 1996). Some pollution is brought to lakes in the waters of tributaries. In this case, this is the Noteć River, which flows into Lake Gopło from the south and whose average flow at the Łysek gauge was 0.89 m3s˗1 in 1961–2000 (Graf 2003). In addition to the main tributary, the lake is supplied with water from 72 small streams and drainage channels and ditches, though these do not flow for most of the year (Juśkiewicz 2014).
Large-scale drainage works at the turn of the twentieth century were fundamental to the economic development of this part of Poland (Kaniecki 2011). They allowed intensive agriculture to develop in the Gopło Lake catchment, this practice being associated with the use of crop protection products and large amounts of mineral and organic fertilisers. The consumption of the latter is equivalent to 170–232 kg of NPK per 1 ha of agricultural land, which is among the highest such consumptions in Poland and Europe (GUS 2008; IERiGŻ 2015). This agricultural activity led not only to the rapid eutrophication of the lake (Wiśniewski 1992), but also to the supply of many toxic substances, including toxic metals. Since the early twentieth century, agricultural-food industries based on a range of crops has been established in the catchment area; these, together with industrial animal husbandry centres and domestic and municipal pollutants, constitute the main point sources of pollution. At various times, over 38 such sources have existed in the lake catchment area (Fig. 1). A large number of food industry plants (fat, wine, sugar factories) have been built in Kruszwica. The town lies immediately lakeside on the northern shore. In the early 1990s, sanitary sewerage systems and related sewage treatment plants were built. Gradually, they have extended their coverage to serve Kruszwica and other towns in the lake's catchment area. Lake Gopło is also within the zone of influence of open-cast lignite mines that have been operating in this region since the 1950s. They are located about 10–18 km as the crow flies to the south-east and west of the lake. The drainage of the open-cast mines has contributed to changing the lake’s groundwater supply conditions (Przybyłek 2020). Another potential source of pollution is the air pollution that results from coal mining and coal combustion in three thermal power stations 21 km from the lake.
Sediment sampling and analytical methods
Twenty sediment cores were collected using a Uwitec Corer. The locations of the sample points (SP) were determined using a Garmin dual-beam GPS sonar. The sites were selected based on a bathymetric plan such that the SPs were evenly distributed across the entire lake area, while maintaining principles that ensure the collecting of samples of undisturbed structure (Fig. 2). From each core, from its upper 10-cm layer, four sediment samples were collected for sedimentological and geochemical analyses, providing eighty samples in total. Metal concentrations averaged from the four samples were used both to analyse the spatial variation in the selected metal concentrations in the sediments and to assess the degree of contamination and ecological risk. The content of organic matter and carbonates in the sediments was determined by loss on ignition (Heiri et al. 2001). The particle-size composition was determined by laser diffraction on a Fritsch Analysette 22 device. After mineralisation of samples in concentrated nitric acid, the concentrations of the toxic metals (copper [Cu], lead [Pb], cadmium [Cd], zinc [Zn], nickel [Ni], chromium [Cr], arsenic [As] and mercury [Hg]) were determined in a microwave oven by atomisation atomic absorption spectrometry methods in the accredited "Labotest" laboratory in Toruń; depending on the element to be determined, this was done in a graphite furnace, in a flame, by hydride technique or by cold vapour technique.
Data analysis
The grain-size analysis results were statistically processed in GRADISTAT software (Blott and Pye 2001), which calculated grain-size indicators according to Folk and Ward (1957).
The contamination of bottom surface sediments with toxic metals was assessed using the common geochemical index, which is Müller’s (1979) geoaccumulation index (Igeo):
where Cm concentration of the analysed metal (mg·kg˗1), GB geochemical background (mg·kg˗1).
The index value was then assigned to one of the seven purity classes according to Müller (1981): class 0 (Igeo < 0) uncontaminated; class 1 (0 < Igeo ≤ 1) uncontaminated to moderately contaminated; class 2 (1 < Igeo ≤ 2) moderately contaminated; class 3 (2 < Igeo ≤ 3) moderately to heavily contaminated; class 4 (3 < Igeo ≤ 4) highly contaminated; class 5 (4 < Igeo ≤ 5) heavily to extremely contaminated; class 6 (5 > Igeo) extremely contaminated. We adopted the geochemical background values that represent the geometric mean concentration of toxic metals in lake and river water sediments in Poland from Bojakowska and Sokołowska (1998).
The summative assessment of sediment contamination with all analysed metals was presented using the pollution load index (PLI) (Tomlinson et al. 1980):
where n number of elements, CF the ratio of selected elements to their background values. The PLI results were used to divide the sediments into three purity classes: PLI ≤ 0, no pollution; PLI = 1, background pollution; and PLI > 1, elevated pollution.
The potential ecological effects of the toxic metal contamination of lake sediments are described in the ecological risk index (RI) (Håkanson 1980). The RI is the sum of the toxic effects of individual metals expressed by the risk factor (ER):
where TRiis the biological toxic factor of element i, which is determined for Cu = Pb = Ni = 5, Cd = 30, Zn = 1, Cr = 2, Hg = 40, As = 10 (Håkanson 1980); and CFi – contamination factor of element i. Due to the set of metals being different in this study, the original (Håkanson 1980) classification of ecological risk indicators (Li et al. 2012) was modified. The risk factor (ERi) is classified as: low risk (ERi < 40), moderate risk (40 ≤ ERi < 80), considerable risk (80 ≤ ERi < 160), high risk (160 ≤ ERi < 320), or very high risk (ERi ≥ 320); whereas, ecological risk index is categorised as: low risk (RI < 100), moderate risk (100 ≤ RI < 200), considerable risk (200 ≤ RI < 400), or very high risk (RI ≥ 400). A set of TEL / PEL guidelines was also used (MacDonald et al. 2000).
Chemometric analyses of toxic metal concentrations in sediments were performed using Pearson's correlation analysis, Principal Component Analysis (PCA) and Cluster Analysis (CA) in the software package Statistica. Pearson correlation analysis was applied to identify the co-occurrence correlations between toxic metals and the nature of the relationship of metal concentrations to the physical properties of sediments and environmental parameters. To identify the possible sources of toxic metals in the Lake Gopło catchment and to discover the similarities and differences in toxic metal concentrations in sediments at each sampling point, PCA and CA analyses were performed. Cluster Analysis (CA) helped identify the measurement points that were most similar to one another in terms of toxic metal concentrations. The most similar points are grouped into one cluster. PCA reduced the number of variables describing a given point (in this case to eight variables—i.e. metal concentrations) and 20 sample points, to several indirect factors, the so-called principle components (PCs) to analyse the relationship between the observed variables. This provides information on the relationship between sampling sites and pollutant concentrations. Cluster analysis was performed on real data (Table S1). In the PCA analysis, data normalised by z-scale transformation were used to avoid the potential for misclassification due to large differences in data dimensionality (Liu et al. 2003). Cluster analyses were performed by Ward's method, using Euclidean distances as a measure of similarity. The PCA was calculated based on a correlation matrix of measured parameters. The number of significant principal components was selected based on the Kaiser criterion of eigenvalues higher than 1 (Kaiser 1960). The correlation between principal component and concentration of toxic metals was classified according to values > 0.75, 0.75–0.50 and 0.50–0.30 as strong, moderate and weak, respectively (Liu et al. 2003). Multivariate statistical analysis is an alternative methods of identifying pollution sources that can determine anthropogenicity or geogenicity (Yalcin et al. 2010, Huang et al. 2017). Combining PCA analysis with CA ensures that sources for a given metal distribution pattern in the sediment will be correctly identified (Upadhyay et al. 2006; Zhou et al. 2008).
Results
General description of lake sediments
The grain-size composition, organic matter content of the bottom surface sediments of Lake Gopło are all characterised by low diversity. Only the share of calcium carbonate in the analysed sediment samples ranged widely, from 20 to 74%, averaging 58%. The content was found to be lower in the sediments in the north of the lake (SP1–SP5), where it averaged 45% of sample mass. In the rest of the lake, the share of CaCO3 in the sediments averaged 63%, and the maximum (SP16) even exceeded 70%. The calcium carbonate that precipitates from the lake water due to biogeochemical and physico-chemical changes mainly derives from young glacial sediments (e.g. Mazurek 1999). The average organic matter content in the sediments is 13.8% and is relatively poorly diversified (8–20%). In the very north of the lake (SP1–SP5) and in the bay on its western side (SP13, SP15, SP17), organic matter constitutes an average of 16% of dry matter, and a maximum of 20%. The share of clastic material in the tested samples ranged from 13 to 62% (averaging 28%), and the grain size was in the range of 8–38 μm. The dominant fraction was silt, which constituted as much as 98%, complemented by clay. The highest number of grains of the corresponding clay fraction was found in sediment samples from the western, bay part of the lake (SP15, SP17). Fifty-nine percent of the silt fraction was coarse grained, 7% very coarse grained, and 34% medium grained. Despite organic matter and carbonates having been removed before grain size was measured, the tested mineral material still contained biogenic silica and metal hydroxides, which probably somewhat affected the grain-size measurement (Żarczyński et al. 2019).
Based on the share of the main components, the bottom surface sediments of the Gopło Lake are—according to the Succow’s classification (1988) with modifications by Kaiser (2001)—carbonate gyttja. Only in the north of the lake (SP1, SP5) was the mineral–organic/carbonate gyttja.
Concentrations of toxic metals in bottom surface sediments
The basic statistics related to toxic metals in bottom surface sediments of Gopło Lake, as well as regional and local values of geochemical background, sediment quality guidelines (SQGs) TEL (Threshold Effect Level) and PEL (Probable Effect Level) is summarised in Table 1. The variability in concentrations of analysed metals at individual sample points is shown in Fig. 3. The mean concentration of all analysed metals except Cd, Cr and As exceeded regional geochemical background, as did all maximum values (including for those three elements). The degree of enrichment of the studied toxic metals in Lake Gopło decreased in the order of Cu > Pb > Zn > Ni > Cd > Cr > Hg > As. The concentration of toxic metals in sediments in Lake Gopło is similar to or below that of other lakes in the region, with the exception of Cu and Zn. Similarly, in comparison to the lakes of post-glacial origin in northern Poland, the concentration of most of the analysed metals is lower. The opposite case only held for copper and mercury contents in the sediments (Table 1). Except for mercury, the mean content of other metals in the sediments was lower than the corresponding TEL values. In a few sediment samples, the concentrations of all metals exceeded TEL values, and lead, zinc, nickel and mercury also exceeded PEL values (Table 1). PEL values were exceeded by nickel in SP1, mercury in SP1 and SP5, and zinc and lead in SP5. It follows that, in the north of the lake, the concentrations of Pb, Zn, Ni and Hg may be causing adverse biological effects (Fig. 3).
Results for toxic metal concentration and chemometric analyses in bottom sediments from Lake Gopło. A Spatial differentiation of concentration of toxic metals in sediments of Lake Gopło, B results of PCA analysis, C correlations between toxic metals in sediments, D Cluster Analysis (CA), dendrogram of the sample sites from the Goło Lake, numbers from 1 to 20—sample points
The coefficients of variation (CV) of all metals (except arsenic) indicate high variability of concentrations (Table 1). The distribution pattern of toxic metals in the lake sediments is presented in Fig. 3 indicates the presence of very high concentrations of all metals at the sampling points in the north of the lake. Concentrations of As, Cr, Zn and Cu are also elevated in the south of the lake.
Pollution levels of the toxic metals of the sediments
The toxic metal contamination of bottom surface sediments in Lake Gopło is assessed as low according to the Igeo index, except at three SPs in the north of the lake (Table 2). Of these three, the most highly contaminated sediments are found in SP5, where the concentration of Cu, Zn and Pb corresponds to purity class 3 (moderately to heavily contaminated), and the Hg contamination is extremely high (class 6). There is also a moderate degree of contamination in the sediments at the second point (SP1), where Ni and Hg concentrations are purity class 3, and Cu, Zn and Pb are class 2. There were also moderate Cu contamination of sediments at the third point (SP2) (Table 2). The summative assessment of sediment contamination with all analysed metals according to the PLI index confirms the results of the geoaccumulation index. The PLI values are above 1 at SP1–3 and SP5, which indicates metal contamination of the sediments. Also noteworthy are the elevated PLI values in the south of the lake (Table 2).
Geochemical indicators that assess the potential ecological and toxicological impact of the total presence of toxic metals in bottom surface sediments have been proposed by Håkanson (1980). The potential ecological risk index (RI) values confirm the poor condition of the bottom surface sediments in the north of Lake Gopło (Table 2). Specifically, at SP5 and SP1, the RI indicator reaches values corresponding to class 4, which means that the excessive concentration of metals is a very high ecological risk. In turn, at SP2 and SP3 the index is high (class 3), and at SP6, SP16 and SP20, it is moderate. However, it should be clearly indicated that, at all the aforementioned SPs, the poor ecological state of bottom surface sediments was due in each case to high mercury contents. At most other SPs, mercury contamination corresponds to a moderate threat, but considerable at SP6 (Table 2). The ER values calculated for this metal at SP1 and SP5 correspond to class 5 (classification of ecological risk indicators, Li et al. 2012), which represents a very high ecological threat, and in the case of SP2 and SP3 a high threat.
Mercury contamination aside, the high RI index values at SP1 and SP5 are also influenced by the sediments’ average level of contamination with copper and cadmium at both locations, plus with lead at SP5 and with nickel at SP1.
Results of chemometric analysis
The Pearson correlation analysis results show a strong cross-correlation between most of the analysed metals (Fig. 3). The six pairings Cu–Pb, Cu–Zn, Cu–Hg, Pb–Zn, Pb–Hg and Zn–Hg are very strongly positively correlated (r > 0.9) at p < 0.05. Also strongly correlated (r = 0.7–0.9) are: Cu–Cd, Cu–Cr, Pb–Cd, Pb–Cr, Cd–Zn, Cd–Ni, Cd–Cr and Cr–Hg. Moderate correlations (r = 0.7–0.4) were found for the pairings Cu–Ni, Cu–As, Pb–As, Pb–Ni, Cd–Hg, Cd–As, Zn–Ni, Zn–As, Ni–Cr and As–Cr. Correlations were weak or absent for the relationships Ni–Hg, As and Hg–As. The principal component analysis identified two significant PCs for sediments. The distinguished components meet the Kaiser criterion and that explain 89.96% of the total variance in the variables (Table 1). The first component (PC1) accounts for 75.92% of the variance and is strongly positively correlated with all metals except nickel (which correlates only moderately with the component values).
The second component explains 13% of the geochemical variability of the sediments and correlates moderately with nickel concentrations, weakly positively with cadmium, and weakly negatively with mercury. The principal component analysis results agree closely with the Pearson correlation analysis results.
The results of cluster analysis confirmed the earlier described picture of the spatial differentiation of toxic metal concentrations in the bottom surface sediments of Lake Gopło. However, this analysis also distinguished an additional group consisting of four points: two in the north of the lake (SP4 and SP6) and points SP13 and SP18 (characterised by deep water conditions in the central and southern part of the lake) (Fig. 3). In these places, concentrations of Zn and Pb were higher than at other points in the central and southern parts of the lake.
Discussion
The analysis of the concentration of toxic metals in the bottom sediments of Lake Gopło showed that most metals except Cu, Pb and Hg correlate positively (not very strongly, but statistically significantly) with the organic matter content in the sediments (Table 1). Such relationships have also been described in other water bodies (Bartoli et al. 2012; Gierszewski 2018; Saleem et al. 2018; Wakida et al. 2008). Many study results have indicated that the fine-grained fraction in the sediments, especially clay, contributes greatly to an increased accumulation of metals (Rubio et al. 2000; Fukue et al. 2006; Dung et al. 2013). This is associated with, inter alia, the high sorption capacity of clay minerals (Foster and Charlesworth 1996; Kabata-Pendias and Pendias 1999; Canavan et al. 2007). However, no such relationship was found in the bottom surface sediments of Lake Gopło. Indeed, the opposite was observed: the metal concentrations were higher in samples containing a greater sandy fraction or with a dominant coarse-silt fraction (Table 1). The unusual nature of the relationship between metal concentrations and grain size of the sediments results from the sediments’ low lithological diversity and, at the same time, their toxic contamination with metals of anthropogenic origin. This blurs the relationship between grain size and metal concentration in the sediment (Abracham 1998; Gierszewski 2018). This may explain, too, why the textural composition does not correlate with the spatial variability of metal concentrations in the bottom surface sediments (Lin et al. 2016). Such a situation occurs in the northern part of Lake Gopło, where the bottom sediments are more mineral in nature. These sediment properties were influenced by the discharges of wastewaters from the Kruszwica sugar refinery, which until 1993 were discharged into the lake. Wastewater from sugar refineries usually contains large amounts of total suspended matter (Kocaturk and Erguder 2015). Although the sorption properties of sediments in this part of the lake are slightly worse, it is here that the toxic metal concentrations were highest. Their source, until the municipal wastewater treatment plant in Kruszwica was established in 1993, was point discharges of industrial and municipal wastewater that flowed into the lake directly or through the hydrographic network. After these sources were eliminated, after 1993, pollutants were supplied only from non-point sources. One factor contributing to the accumulation of pollutants in this part of the lake is its impeded water exchange with the main lake basin.
By contrast, the metal concentration levels were not affected by other substances included in the analysis that participate in toxic metal adsorption, i.e. phosphorus and calcium carbonate compounds (Sigg et al. 1997; Kabata-Pendias and Pendias 1999; Thakur et al. 2006; Reddy et al. 2014). The negative correlation between the content of metals and the share of calcium carbonate in the sediments is primarily the result of there being a large supply of metals to the north of the lake, where carbonate sediments happen to occur least.
Toxic metals that accumulate in bottom surface sediments are variously interrelated. The nature and strength of these relationships can indirectly inform on their origins and migration routes (Håkanson and Jansson 1983; Sutherland 2000; Yang et al. 2009). High values of positively correlated metals generally indicate a common source of origin and similar behaviour during transport (Lu et al. 2010; Chen et al. 2012; Wang et al. 2016). The nature of the relationships between pairs of metals in the bottom sediments of Lake Gopło, considering the low toxic metal contamination of the sediments at most sampling points, indicates their geogenic origin and possible delivery from non-point sources (Wang et al. 2015; Guan et al. 2016). In the case of the lake under study, these are mainly non-point pollutants related to more than a century of intensive agriculture in the catchment. The very strong correlations between Pb, Zn, Cu, and Hg indicate that their concentrations in the sediments were also influenced by supply from a specific anthropogenic source.
Conclusions drawn from the analysis of dependencies between the analysed metals confirm and complement the PCA analysis results. This shows that all elements, especially Cu, Zn, Pb, Hg and Cd, achieved high loads in the first component (PC1). In the second principle component (PC2), Ni had a higher content than in the first principle component (Table S1). This means that the nickel in certain parts of the lake was also supplied from point sources. The distribution of sample points on a coordinate system defined by the two principal components clearly distinguishes SP1–SP3 and SP5 from the others (Fig. 3). These points are located in the north of the lake, which is surrounded by municipal and industrial buildings of Kruszwica. This part of the lake consists of two pools separated by narrow straits and a shallow bay of approx. 2 m deep, which impedes water exchange with the rest of the lake. Bottom surface sediments from SP1 and SP2 contain significantly more nickel, cadmium and chromium than the other points. The nickel is being supplied to the very north of the lake by Kujawskie Zakłady Tłuszczowe (Kuyavian manufacture of oils and fats). The concentration of this metal increased significantly in the 1960s (Juśkiewicz et al. 2015), when a margarine factory was established in a company expansion (Komosiński 2007). The production of margarine from liquid vegetable fats uses nickel as a catalyst in the hydrogenation reaction (Tarrago-Trani et al. 2006). In addition to the nickel levels, the mineral–organic sediments here were found to have much higher concentrations of Cu, Pb, Zn, Hg, at SP3 and SP5 (and additionally of Cr and As at SP5) than in the central and southern parts of the lake. The remaining SPs form one cluster of points with a very similar geochemistry of toxic metals (Fig. 3). It can be identified with the supply of metals from non-point agricultural sources, but also with the deposition of atmospheric pollutants (combustion products, traffic pollution) and above all with a geogenic source. Many agricultural chemicals contain toxic metals. For example, Cu, Zn and As are present in fungicides, pesticides and herbicides (He et al. 2005). Meanwhile, mineral fertilisers may contain contaminants in the form of Cd and Pb or additions of Cu and Zn. Furthermore, many toxic metals (Cu, Zn, Pb, Cd, Ni, Cr) also derive from organic fertilisers (farm manures, biosolids, composts). They feature much more richly in these products than in most agricultural soils (He et al. 2005). Long-range atmospheric transport followed by wet and dry deposition is responsible for the accumulation of many metals (As, Cd, Cu, Zn and Pb) in soil and sub-aqueous sediments (Sarkar et al. 2015).
The ecological effects of the toxic metal contamination of the bottom surface sediments are varied. Some metals (e.g. Zn, Cu, Fe, Mn, Mg) are essential to biological systems. However, they can also be toxic when critical concentrations are exceeded (Magnitskiy 2011; Chen et al. 2018). Cadmium, lead, arsenic, chromium and mercury have no practical biological significance when contained in sediments at natural levels, but when present in high concentrations they are toxic to organisms (Alloway 2013; Zheng et al. 2013). For all the sediments, the elevated (and in some parts of the lake, high) concentrations of mercury have the greatest potential impact on the functioning of Lake Gopło’s ecosystem.
Besides its supply in the municipal and industrial wastewater that affects the north of the lake, this element also probably derives from atmospheric supply as airborne ash that is a by-product of lignite combustion (Lorenz and Grudziński 2007). Research has shown that the lignite extracted from the mines to the south of the lake has the highest mercury content in Poland. Its concentration in the fuel supplied to the Adamów power plant was 442 ppb (parts per billion) (Bojakowska and Sokołowska 2001). Another source of mercury is agricultural pesticides (Sojka et al. 2019).
The results showed that more than a century of intensive agricultural activity in the catchment area of Lake Gopło has not significantly contaminated the bottom surface sediments with toxic metals. The way the soil has been fertilised has had some part to play in this. In Poland, including in the Kuyavia region, artificial fertilisers have been used (Jankowiak et al. 2003; Dach and Starmans 2005). Such fertilisers contain significantly lower levels of toxic metals than do organic fertilisers (especially liquid manure) (Kabata-Pendias and Pendias 1999). However, it cannot be ruled out that the use of crop protection products, especially herbicides (Piwowar 2021), combined with the deposition of pollutants from fossil fuel combustion processes, has contributed to the increased accumulation of mercury in the bottom surface sediments.
Conclusion
Based on the analysis of the differentiation of toxic metal concentrations in the surface layer of bottom sediments of Lake Gopło and chemical analyses, the factors determining the levels, origins and probable sources of supply of metals to the lake were determined. It was shown that the average concentrations of the analysed metals (except Cd, Cr and As) exceeded regional geochemical background levels. The content of toxic metals in the sediments of Lake Gopło was similar to or even below that of many other lakes in central and northern Poland (except for Cu, Zn and Hg). The low variation in the lithological features of the bottom surface sediments means that the size of the clay fraction and the levels of carbonates and organic matter content do not affect the metal content in the sediments in Lake Gopło, nor, consequently, their spatial diversity. The significantly greater concentration of toxic metals in the north of the lake is due to their supply in municipal and industrial pollution from Kruszwica, and to the fact that the restricted water exchange between the north of the lake and the rest of the lake limits the transfer of pollutants to other parts of the lake. As shown by the PCA analysis, the bottom surface sediments in the north of the lake have significantly higher contents of Ni, Cd, and Cr. The strong correlation between most of the analysed metals and the results of the factor analysis indicate a similarity of source and method of supply to the lake. Except in the north of the lake, the metals are from agricultural area sources, atmospheric pollution (mainly fossil fuel combustion products) and natural sources. The weaker correlation of Ni with other metals indicates that this metal has a different source. The PCA analysis results indicate that the source is in the northern part of the lake. There, the sources of Ni, but also of Cd, are industrial pollutants from food industry plants (fat plants, sugar plants).
Compared to the rest of the lake, the sediments in the northern part are heavily polluted according to Igeo and PLI values. These values indicate moderate and moderate-to-toxic contamination of sediments with Cu, Pb and Zn, Ni and Hg. The most polluted sediments were those in the lake bay that protrudes into the town area. The values of contamination indices were highest there for most of the analysed metals, and the contamination of the sediments with mercury, which is ecologically dangerous, was extremely high. Compared to mercury, the copper, cadmium, lead and nickel contamination of the sediments in the north of the lake has a lesser average impact on the index of potential ecological threat.
The relatively low toxic metal contamination of the bottom surface sediments in the southern and central parts of the lake contrasts with the contaminated sediments in the north of the lake, which receives industrial and municipal pollution from the town of Kruszwica. However, the morphology of the lake basin and the restricted water exchange between the north of the lake and the rest of the lake prevent these pollutants from being transmitted southwards. Despite the previous intensive use of the lake and adjacent areas, the condition of the bottom surface sediments of Lake Gopło is good. Now that the site is managed in an ecologically sustainable manner, it has a full chance of ensuring the long-term survival of some of Europe's most valuable and endangered species and habitats listed in both the Birds and Habitats Directives.
Availability of data and materials
The datasets supporting the conclusions of this article are included within the article and its additional files.
References
Abraham J (1998) Spatial distribution of major and trace elements in shallow reservoir sediments: an example from Lake Waco, Texas. Environ Geol 36:349–363
Alloway BJ (ed) (2013) Heavy metals in soils. Trace metals and metalloids in soils and their bioavailability. Springer, Berlin
Atkinson C, Jolley D, Simpson S (2007) Effect of overlying water pH, dissolved oxygen, salinity and sediment disturbances on metal release and sequestration from metal contaminated marine sediments. Chemosphere 69:1428–1437. https://doi.org/10.1016/j.chemosphere.2007.04.068
Atlas geochemiczny Polski, 2012. http://www.mapgeochem.pgi.gov.pl.
Bartoli G, Papa S, Sagnella E, Fioretto A (2012) Heavy metal content in sediments along the Calore river: relationships with physical–chemical characteristics. J Environ Manage 95:9–14. https://doi.org/10.1016/j.jenvman.2011.02.013
Blott SJ, Pye K (2001) Gradistat: a grain size distribution and statistics package for the analysis of unconsolidated sediments. Earth Surf Proc Land 26:1237–1248. https://doi.org/10.1002/esp.261
Bojakowska I, Sokołowska G (1996) Metale ciężkie w osadach jezior Pojezierza Kaszubskiego. Przegląd Geologiczny 44(9):920–923
Bojakowska I, Sokołowska G (1997) Akumulacja pierwiastków śladowych w osadach jeziornych w zależności od strefy ich sedymentacji. Przegląd Geologiczny 45(5):505–508
Bojakowska I, Sokołowska G (1998) Geochemiczne klasy czystości osadów wodnych. Przegląd Geologiczny 46(1):49–54
Bojakowska I, Sokołowska G (2001) Polycyclic aromatic hydrocarbons in brown coals from Poland. Geol Quart 45(1):93–98
Brekhovskikh V, Volkova Z, Kocharyan A (2001) Heavy metals in the Ivan’kovo reservoir bottom sediments. Water Resour 28:278–287. https://doi.org/10.1023/A:1010448723589
Cai Y, Zhang W, Zhou M, Jiang H, Xu D, An S, Leng X (2015) Comprehensive assessment of heavy metal contamination insurface sediments from the inflow rivers of Taihu basin. Clean - Soil, Air, Water 43:1582–1591. https://doi.org/10.1002/clen.201300886
Canavan RW, Van Cappellen P, Zwolsman JJG, van den Berg GA, Slomp CP (2007) Geochemistry of trace metals in a fresh water sediment: field results and diagenetic modeling. Sci Total Environ 381:263–279. https://doi.org/10.1016/j.scitotenv.2007.04.001
Cantwell MG, Burgess RM, King JW (2008) Resuspension of contaminated field and formulated reference sediments Part I: evaluation of metal release under controlled laboratory conditions. Chemosphere 73:1824–1831. https://doi.org/10.1016/j.chemosphere.2008.08.007
Chabukdhara M, Nema AK (2012) Assessment of heavy metal contamination in Hindon River sediments: a chemometric and geochemical approach. Chemosphere 87:945–953
Chang X, Yingxia L (2020) Using a multimedia aquivalence model to evaluate the environmental fate of Fe, Mn and trace metals in an industrial City. China Water 12:1580. https://doi.org/10.3390/w12061580
Chen B, Liang X, Xu W, Huang X, Li X (2012) The changes in trace metal contamination over the last decade in surface sediments of the Pearl River Estuary. South China. Sci Total Environ 439:141–149
Chen L, Zhou S, Shi Y, Wang C, Li B, Li Y, Wu S (2018) Heavy metals in food crops, soil, and water in the Lihe River watershed of the Taihu Region and their potential health risks when ingested. Sci Total Environ 615:141–149. https://doi.org/10.1016/j.scitotenv.2017.09.230
Cieśla W, Dąbkowska-Naskręt H, Borowska K, Malczyk P, Długosz J, Jaworska H, Kędzia W, Zalewski W (1994) Pierwiastki śladowe w glebach wybranych obszarów Pomorza i Kujaw. Zeszyty Problemowe Postępów Nauk Rolniczych 414:63–70
Cobelo-Garcia A, Prego R (2004) Influence of point sources on trace metal contamination and distribution in a semi-enclosed industrial embayment: the Ferrol Ria (NW Spain) Estuarine. Coast Shelf Sci 60:695–703
Copat C, Bella F, Castaing M, Fallico R, Sciacca S, Ferrante M (2012) Heavy metals concentrations in fish from sicily (Mediterranean Sea) and evaluation of possible health risks to consumers. Bull Environ Contam Toxicol 88:78–83. https://doi.org/10.1007/s00128-011-0433-6
Dach J, Starmans D (2005) Heavy metals balance in Polish and Dutch agronomy: Actual state and previsions for the future. Agr Ecosyst Environ 107(4):309–316. https://doi.org/10.1016/j.agee.2005.02.017pendias
De Vleeschouwer F, Fagel N, Cheburkin A, Pazdur A, Sikorski J, Mattielli N, Renson V, Fialkiewicz B, Piotrowska N, Le Roux G (2009) Anthropogenic impacts in North Poland over the last 1300years—a record of Pb, Zn, Cu, Ni and S in an ombrotrophic peat bog. Sci Total Environ 407(21):5674–5684. https://doi.org/10.1016/j.scitotenv.2009.07.020
Dickinson WW, Dunbar GB, McLeod H (1996) Heavy metal history from cores in Wellington Harbour, New Zealand. Environ Geol 27:59–69
Dung TTT, Cappuyns V, Swennen R, Phung NK (2013) From geochemical background determination to pollution assessment of heavy metals in sediments and soils. Rev Environ Sci Bio/Technol 12:335–353. https://doi.org/10.1007/s11157-013-9315-1
EEA (European Environment Agency), 2012. natura2000.eea.europa.eu.
Eggleton J, Thomas KV (2004) A review of factors affecting the release and bioavailability of contaminants during sediment disturbance events. Environ Int 30:973–980. https://doi.org/10.1016/j.envint.2004.03.001
EU (European Union), 2013. Council Directive 92/43/EEC, http://data.europa.eu/eli/dir/1992/43/oj.
EU (European Union), 2019. Council Directive 2009/147/EC, http://data.europa.eu/eli/dir/2009/147/oj.
Folk RL, Ward WC (1957) Brazos River bar: a study in the significance of grain size parameters. J Sediment Petrol 27:3–26
Förstner U (1982) Accumulative phases for heavy metals in limnic sediments. Hydrobiologia 91–92:269–284. https://doi.org/10.1007/BF02391944
Foster IDL, Charlesworth SM (1996) Heavy metals in the hydrological cycle: trends and explanation. Hydrol Process 10(2):227–261
Fukue M, Yanai M, Sato Y, Fujikawa T, Furukawa Y, Tani S (2006) Background values for evaluation of heavy metal contamination in sediments. J Hazard Mater 136(1):111–119. https://doi.org/10.1016/j.jhazmat.2005.11.020
Garrett RT (2000) Natural sources of metals to the environment. Hum Ecol Risk Assessm 6:945–962. https://doi.org/10.1080/10807030091124383
Gierszewski P (2018) Hydromorfologiczne uwarunkowania funkcjonowania geoekosystemu Zbiornika Włocławskiego. Prace Geograficzne. Instytut Geografii i Przestrzennego Zagospodarowania PAN, Warszawa
GIOŚ (Chief Inspectorate of Environmental Protection), 2012. clc.gios.gov.pl
Goszczyński J, Jutrowska E, (1996) Stan czystości jeziora Gopło. Biblioteka Monitoringu Środowiska. Bydgoszcz
Graf R (2003) Mapa Hydrograficzna Polski w skali 1:50 000, arkusz n-34–122-c Sompolno, komentarz, Geomat Poznań
Guan Q, Cai A, Wang F, Wang L, Wu T, Pan B, Song N, Li F, Lu M (2016) Heavy metals in the riverbed surface sediment of the Yellow River, China. Environ Sci Pollut Res Int 23:24768–24780. https://doi.org/10.1007/s11356-016-7712-z
Gunawardena J, Egodawatta P, Ayoko GA, Goonetilleke A (2013) Atmospheric deposition as a source of heavy metals in urban stormwater. Atmos Environ 68:235–242
GUS (Central Statistical Office), 2011. Środki produkcji w rolnictwie w roku gospodarczym 2010/2011, Warszawa
Häder D-P, Banaszak AT, Villafañe VE, Narvarte MA, González RAE, Helbling W (2020) Anthropogenic pollution of aquatic ecosystems: emerging problems with global implications. Sci Total Environ 713:136586. https://doi.org/10.1016/j.scitotenv.2020.136586
Håkanson L (1980) An ecological risk index for aquatic pollution control a sedimentological approach. Water Res 14(8):975–1001. https://doi.org/10.1016/0043-1354(80)90143-8
Håkanson L, Jansson M (1983) Principles of lake sedimentology. Springer-Verlag, Berlin
He Z, Yang XE, Stoffella PJ (2005) Trace eelements in agroecosystems and impacts on the environment. J Trace Elem Med Biol 19(2–3):125–140. https://doi.org/10.1016/j.jtemb.2005.02.010
Heiri O, Lotter AF, Lemcke G (2001) Loss on ignition as a method for estimating organic and carbonate content in sediments: reproducibility and comparability of results. J Paleolimnol 25:101–110. https://doi.org/10.1023/A:1008119611481
Helios-Rybicka E (1986) The role of clay materials in binding heavy metals by bottom sediments in the upper sector of the River Vistula
Helios-Rybicka E (1997) Parametry określające rozkład metali ciężkich w systemie woda–osad. Geochemiczne zmiany w środowisku gruntowo-wodnym pod wpływem związków toksycznych. Materiały IV Konferencji Naukowej, Akademia Górniczo Hutnicza, Kraków, 31–38.
Huang F, Xu Y, Tan Z, Wu Z, Xu H, Shen L, Xu X, Han Q, Guo H, Hu Z (2018) Assessment of pollutions and identification of sources of heavy metals in sediments from west coast of Shenzhen, China. Environ Sci Pollut Res Int 25:3647–3656. https://doi.org/10.1007/s11356-017-0362-y
IERiGŻ (Institute of Agricultural and Food Economics), 2015.Rolnictwo i Gospodarka Żywnościowa w Polsce, Warszawa.
Jain CK, Sharma MK (2001) Distribution of trace metals in the Hindon river system, India. J Hydrol 253:81–90. https://doi.org/10.1016/S0022-1694(01)00484-X
Jankowiak J, Szpakowska B, Bienkowski J (2003) Ecological aspects of transformation in Poland’s agriculture based on the Wielkopolska region. A J Hum Environ 32(6):418–423. https://doi.org/10.1579/0044-7447-32.6.418
Jiang X, Wang W, Wang S, Zhang B, Hu J (2012) Initial identification of heavy metals contamination in Taihu Lake, a eutrophic lake in China. J Environ Sci 24(9):1539–1548. https://doi.org/10.1016/S1001-0742(11)60986-8
Juśkiewicz W (2014) Attempt modelling water velocity and the spread of contamination in the Gopło Lake. Landform Anal 25:21–27. https://doi.org/10.12657/landfana.025.003
Juśkiewicz W, Marszelewski W, Tylmann W (2015) Differentiation of the concentration of heavy metals and persistent organic pollutants in lake sediments depending on the catchment management (Lake Gopło case study). Bull Geogr Phys Geogr Ser 8:71–80. https://doi.org/10.1515/bgeo-2015-0006
Kabata-Pendias A, Pendias H (1999) Biogeochemia pierwiastków śladowych. Wyd. II. PWN, Warszawa.
Kaiser HF (1960) The application of electronic computers to factor analysis. Educ Psychol Measur 20:141–151. https://doi.org/10.1177/001316446002000116
Kaiser K (2001) Diespätpleistozäne bis frühholozäne Beckenentwicklung in Mecklenburg—Vorpommern. Greifswalder Geographische Arbeiten 24:208
Kang T-W, Yang HJ, Han JH, Han Y-U, Kim M-S, Kim J, Hwang SH, Na EH, Chang YS (2022) Identifying pollution sources of sediment in Lake Jangseong, Republic of Korea, through an extensive survey: internal disturbances of past aquaculture sedimentation. Environ Pollut 306:119403
Kaniecki A (2011) Przemiany stosunków wodnych w dolinie Górnej Noteci do połowy XIX wieku związane z antropopresją. Badania fizjograficzne, R II. Ser A Geografia Fizyczna A62:41–58. https://doi.org/10.2478/v10116-011-0004-x
Kocaturk I, Erguder TH (2015) Investigation of the use of aerobic granules forthe treatment of sugar beet processing wastewater. Environ Technol 36(20):2577–2587
Komosiński S (2007) Mikroekonomiczny obraz przemysłu Polski Ludowej w latach 1950–1980: na przykładzie regionu kujawsko-pomorskiego, Wydawnictwo Poznańskie.
Kostka A, Leśniak A (2021) Natural and anthropogenic origin of metals in lacustrine sediments; assessment and consequences—a case study of wigry lake (Poland). Minerals 11(2):158. https://doi.org/10.3390/min11020158
Kuriata-Potasznik A, Szymczyk S, Skwierawski A, Glińska-Lewczuk K, Cymes I (2016) Heavy metal contamination in the surface layer of bottom sediments in a flow-through lake: a case study of lake Symsar in northern Poland. Water 8:358. https://doi.org/10.3390/w8080358
Larrose A, Coynel A, Schafer J, Blanc G, Masse L et al (2010) Assessing the current state of the Gironde Estuary by mapping priority contaminant distribution and risk potential in surface sediment. Appl Geochem 25:1912–1923
Li RZ, Pan CR, Xu JJ, Ding GZ, Zou Y (2012) Application of potential ecological risk assessment model based on Monte Carlo simulation. Res Environ Sci 25:1336–1343
Lin Q, Liu E, Zhang E, Li K, Shen J (2016) Spatial distribution, contamination and ecological risk assessment of heavy metals in surface sediments of Erhai Lake, a large eutrophic plateau lake in southwest China. CATENA 145:193–203. https://doi.org/10.1016/j.catena.2016.06.003
Lindström M (2001) Urban land use influences on heavy metals fluxes and surface sediment concentrations of small lakes. Water Air Soil Pollut 126(3–4):363–383. https://doi.org/10.1023/A:1005291611702
Liu CW, Lin KH, Kuo YM (2003) Application of factor analysis in the assessment of groundwater quality in a Blackfoot disease area in Taiwan. Sci Total Environ 313:77–89
Lorenz U, Grudziński Z (2007) Zawartość rtęci jako potencjalny czynnik ograniczający wartość użytkową węgla kamiennego i brunatnego. Górnictwo i GeoinżYnieria 31(3):335–349
Loska K, Wiechuła D (2003) Application of principal component analysis for the estimation of source of heavy metal contamination in surface sediments from the rybnik reservoir. Chemosphere 51(8):723–733. https://doi.org/10.1016/S0045-6535(03)00187-5
Lu X, Wang L, Li LY, Lei K, Huang L, Kang D (2010) Multivariate statistical analysis of heavy metals in street dust of Baoji, NW China. J Hazard Mater 173:744–749
MacDonald DD, Ingerosoll CG, Berger TA (2000) Development and evaluation of consensus-based sediment quality guidelines for freshwater ecosystems. Arch Environ Contamin Toxicol 39:20–31. https://doi.org/10.1007/s002440010075
Magnitskiy S (2011) Nickel: the last of the essential micronutrients. Agron Colomb 29:49–56
Mazurek M (1999) Mineralogy of the opalinus clay. In: Thury M, Bossart P (eds), Mont terri rock laboratory. Results of the hydrogeological, geochemical and geotechnical experiments performed in 1996 and 1997. Geological Reports No. 23. Federal Office of Topography (swisstopo), Wabem, Switzerland. http://www.mont-terri.ch.
Molewski P (1999) Rynna Gopła–problem jej genezy i roli w odpływie wód roztopowych podczas zlodowacenia vistuliańskiego Studia Societatis Scientiarum Torunensis. Geogr Geol 10:6
Müller G (1979) Schwermetalle in den Sedimenten des Rheins Veränderungenseit 1971. U Umschau in Wissenschaft Und Technik 79:778–783
Müller G (1981) The heavy metal pollution of the sediments of neckars and its tributary: a stocktaking. Chemiker Zeitung 105:157–164
Nriagu JO (1996) A history of global metal pollution. Science 272(5259):223–224. https://doi.org/10.1126/science.272.5259.223
PIG (Polish Geological Institute), 2012, Geochemical Atlas of Poland, mapgeochem.pgi.gov.pl.
Piwowar A (2021) The use of pesticides in Polish agriculture after integrated pest management (IPM) implementation. Environ Sci Pollut Res 28:26628–26642. https://doi.org/10.1007/s11356-020-12283-w
Przybyłek J (2020) Środowiskowe uwarunkowania skutków odkrywkowej eksploatacji węgla brunatnego na Pojezierzu Gnieźnieńskim. Przegląd Geol 68(8):645–654. https://doi.org/10.7306/2020.24
Reddy KR, Xie T, Dastgheibi S (2014) Removal of heavy metals from urban stormwater runoff using different filter materials. J Environ Chem Eng 2(1):282–292. https://doi.org/10.1016/j.jece.2013.12.020
Rubio B, Nombela MA, Vilas F (2000) Geochemistry of major and trace elements in sediments of the Ria de Vigo (NW Spain) an assessment of metal pollution. Mar Pollut Bull 40(11):968–980
Sahoo PK, Dall’Agnol R, Negreiros SG (2019) High resolution hydrogeochemical survey and estimation of baseline concentrations of trace elements in surface water of the Itacaiúnas River Basin, southeastern Amazonia. J Geochem Explor 205:106321
Saleem M, Iqbal J, Akhter G, Shah MH (2018) Fractionation, bioavailability, contamination and environmental risk of heavy metals in the sediments from a freshwater reservoir, Pakistan. J Geochem Explor 184:199–208. https://doi.org/10.1016/j.gexplo.2017.11.002
Salomons W, Stigliani W (1995) Biogeodynamics of pollutants in soils and sediments. Springer Verlag, Berlin
Sarkar ST, Ahmed K, Swami CD, Judd A, Bari V, Dutkiewicz A, Husain L (2015) History of atmospheric deposition of trace elements in lake sediments, ~1880 to 2007. J Geophys Res Atmos 120:5658–5669. https://doi.org/10.1002/2015JD023202
Siegel F (2002) Heavy metals mobility/immobility in environmental media. 45–59. https://doi.org/10.1007/978-3-662-04739-2_3
Sigg L, Sturm M, Kistler D (1997) Vertical transport of heavy metals by settling particles in Lake Zurich. Limnol Oceanogr 32(1):112–130
Singh UK, Kumar B (2017) Pathways of heavy metals contamination and associated human health risk in Ajay River basin, India. Chemosphere 174:183–199. https://doi.org/10.1016/j.chemosphere.2017.01.103
Skwierawski A, Sidoruk M (2014) Heavy metal concentrations in the sediment profiles of the antropogenically transformed Płociduga reservoir. Ecol Chem Eng S 21:79–88
Smal H, Ligęza S, Wójcikowska-Kapusta A, Baran S, Urban D, Obroślak R, Pawłowski A (2015) Spatial distribution and risk assessment of heavy metals in bottom sediments of two small dam reservoirs (south-east Poland). Arch Environ Protect 41:67–80
Smith SL, MacDonald DD, Keenleyside KA, Ingersol CG, Field LJ (1996) A preliminary evaluation of sediment quality assessment values for freshwater ecosystems. J Great Lakes Res 22(3):624–638
Sojka M, Siepak M, Gnojska E (2013) Assessment of heavy metal concentration in bottom sediments of Stare Miasto pre-dam reservoir on the Powa river. Ann Set the Environ Protect 15:1916–1928
Sojka M, Siepak M, Jaskuła J, Wicher-Dysarz J (2018) Heavy metal transport in a river-reservoir system: a case study from central poland. Pol J Environ Stud 27:1725–1734
Sojka M, Jaskuła J, Wróżyński R (2019) Analysis of heavy metals contamination in bottom sediments of lakes located in the Gniezno Lakeland. Acta Sci Polonorum Formatio Circumiectus 18(4):137–149. https://doi.org/10.15576/ASP.FC/2019.18.4.137
Succow, M., 1988. Landschaftsakologische Moorkunde, Jena, 340.
Sutherland RA (2000) Bed sediment-associated trace metals in an urban stream, Oahu. Hawaii Environ Geol 39(6):611–627
Tao Y, Yan Z, Yuan Z (2015) Distribution and bioavailability of heavy metals in different particle-size fractions of sediments in Taihu Lake, China. Chem Spec Bioavailab 24(4):205–215. https://doi.org/10.3184/095422912X13488240379124
Tarrago-Trani MT, Phillips KM, Lemar LE, Joanne M, Holden JM (2006) New and existing oils and fats used in products with reduced trans-fatty acid content. J Am Diet Assoc 106(6):867–880. https://doi.org/10.1016/j.jada.2006.03.010
Thakur SK, Tomar NK, Pandeya SB (2006) Influence of phosphate on cadmium sorption by calcium carbonate. Geoderma 130(3–4):240–249. https://doi.org/10.1016/j.geoderma.2005.01.026
Tomlinson DL, Wilson JG, Harris CR, Jeffrey DW (1980) Problems in the assessment of heavy-metal levels in estuaries and the formation of a pollution index. Helgolander Meeresunters 33:566–575
Upadhyay AK, Gupta KK, Sircar JK, Deb MK, Mundhara GL (2006) Heavy metals in freshly deposited sediments of the river Subernarekha, India: an example of lithogenic and anthropogenic effects. Environ Geol 50:397–403. https://doi.org/10.1007/s00254-006-0218-0
Varol M (2011) Assessment of heavy metal contamination in sediments of the Tigris river (Turkey) using pollution indices and multivariate statistical techniques. J Hazard Mater 195:355–364. https://doi.org/10.1016/j.jhazmat.2011.08.051
Violante A, Cozzolino V, Perelomov L, Caporale AG, Pigna M (2010) Mobility and bioavailability of heavy metals and metalloids in soil environments. J Soil Sci Plant Nutr 10(3):268–292. https://doi.org/10.4067/S0718-95162010000100005
Wakida FT, Lara-Ruiz D, Temores-Peña J, Rodriguez-Ventura JG, Diaz C, Garcia-Flores E (2008) Heavy metals in sediments of the Tecate River, Mexico. Environ Geol 54:637–642
Wang LF, Yang LY, Kong LH, Li S, Zhu JR, Wang YQ (2014) Spatial distribution, source identification and pollution assessment of metal content in the surface sediments of Nansi Lake, China. J Geochem Explor 140:87–95. https://doi.org/10.1016/j.gexplo.2014.02.008
Wang Y, Yang L, Kong L, Liu E, Wang L, Zhu J (2015) Spatial distribution, ecological risk assessment and source identification for heavy metals in surface sediments from Dongping Lake, Shandong, East China. CATENA 125:200–205. https://doi.org/10.1016/j.catena.2014.10.023
Wang Y, Jiao J, Zhang K, Zhou Y (2016) Enrichment and mechanisms of heavy metal mobility in a coastal quaternary groundwater system of the Pearl River Delta, China. Sci Total Environ 545–546:493–502. https://doi.org/10.1016/j.scitotenv.2015.12.019
Wang X, Zhang L, Zhao Z, Cai Y (2018) Heavy metal pollution in reservoir in the hilly area of southern China: distribution, source apportionment and health risk assessment. Sci Total Environ 634:158–169. https://doi.org/10.1016/j.scitotenv.2018.03.340
Wilson DC (2018) Potential urban runoff impacts and contaminant distributions in shoreline and reservoir environments of Lake Havasu, southwestern United States. Sci Total Environ 621:95–107. https://doi.org/10.1016/j.scitotenv.2017.11.223
Wiśniewski R (1992) The eutrophication of Gopło lake: hydrological instability as a cause of zoobenthos changes. Acta Univ Nicolai Copernici Nauki Matematyczno-Przyrodnicze Prace Limnologiczne 17:63–72
Wu B, Wang G, Wu J, Fu Q, Liu C (2014) Sources of heavy metals in surface sediments and an ecological risk assessment from two adjacent plateau reservoirs. PLoS ONE 9(7):e102101. https://doi.org/10.1371/journal.pone.010210
Xia F, Zhang C, Qu L, Song Q, Ji X, Mei K, Dahlgren RA, Zhang M (2020) A comprehensive analysis and source apportionment of metals in riverine sediments of a rural-urban watershed. J Hazard Mater 381:121230. https://doi.org/10.1016/j.jhazmat.2019.121230
Yalcin MG, Tumuklu A, Sonmez M, Erdag DS (2010) Application of multivariate statistical approach to identify heavy metal sources in bottom soil of the Seyhan River (Adana), Turkey. Environ Monit Assess 164:311–322
Yang Z, Wang Y, Shen Z, Niu J, Tang Z (2009) Distribution and speciation of heavy metals in sediments from the mainstream, tributaries, and lakes of the Yangtze River catchment of Wuhan, China. J Hazard Mater 166:1186–1194. https://doi.org/10.1016/j.jhazmat.2008.12.034
Żarczyński M, Szmańda J, Tylmann W (2019) Grain-size distribution and structural characteristics of varved sediments from Lake Zabińskie (Northeastern Poland). Quaternary 2(8):1–15. https://doi.org/10.3390/quat2010008
Zemełka G, Kryłów M, van Szalińska OE (2019) The potential impact of land use changes on heavy metal contamination in the drinking water reservoir catchment (Dobczyce Reservoir, south Poland). Arch Environ Protect 45(2):3–11. https://doi.org/10.24425/aep.2019.127975
Zeng Y, Bi C, Jia J, Deng L, Chen Z (2020) Impact of intensive land use on heavy metal concentrations and ecological risks in an urbanized river network of Shanghai. Ecol Ind 116:106501. https://doi.org/10.1016/j.ecolind.2020.106501
Zhang C, Yu Z, Zeng G, Jiang M, Yang Z, Cui F, Zhu M, Shen L, Hu L (2014) Effects of sediment geochemical properties on heavy metal bioavailability. Environ Int 73:270–281. https://doi.org/10.1016/j.envint.2014.08.010
Zhang H, Huo S, Yeager KM, Xi B, Zhang J, He Z, Ma C, Wu F (2018) Accumulation of arsenic, mercury and heavy metals in lacustrine sediment in relation to eutrophication: Impacts of sources and climate change. Ecol Ind 93:771–780. https://doi.org/10.1016/j.ecolind.2018.05.059
Zhao X, Gao B, Xu D, Gao L, Yin S (2017) Heavy metal pollution in sediments of the largest reservoir (Three Gorges Reservoir) in China: a review. Environ Sci Pollut Res 24:20844–20858. https://doi.org/10.1007/s11356-017-9874-8
Zheng S, Wang P, Wang C, Hou J, Qian J (2013) Distribution of metals in water and suspended particulate matter during the resuspension processes in Taihu Lake sediment, China. Quatern Int 286:94–102. https://doi.org/10.1016/j.quaint.2012.09.003
Zhou J, Ma D, Pan J, Nie W, Wu K (2008) Application of multivariate statistical approach to identify heavy metal sources in sediment and waters: a case study in Yangzhong, China. Environ Geol 54:373–380. https://doi.org/10.1007/s00254-007-0824-5
Zhu L, Liu J, Xu S, Xie Z (2017) Deposition behavior, risk assessment and source identification of heavy metals in reservoir sediments of Northeast China. Ecotoxicol Environ Saf 142:454–463
Acknowledgements
The authors greatly appreciate the reviewers who dedicated their considerable time and expertise to the article’s rigorous peer-review process.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Juśkiewicz, W., Gierszewski, P. Toxic metal pollution of aquatic ecosystems of European Union nature protection areas in a region of intensive agriculture (Lake Gopło, Poland). Aquat Sci 84, 52 (2022). https://doi.org/10.1007/s00027-022-00884-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00027-022-00884-1