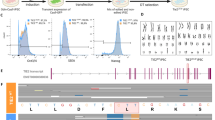
Abstract
Rationale
CD34+ cells are believed being progenitors that may be used to treat cardiovascular disease. However, the exact identity and the role of CD34+ cells in physiological and pathological conditions remain unclear.
Methods
We performed single-cell RNA sequencing analysis to provide a cell atlas of normal tissue/organ and pathological conditions. Furthermore, a genetic lineage tracing mouse model was used to investigate the role of CD34+ cells in angiogenesis and organ fibrosis.
Results
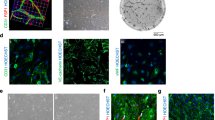
Single-cell RNA sequencing analysis revealed a heterogeneous population of CD34+ cells in both physiological and pathological conditions. Using a genetic lineage tracing mouse model, we showed that CD34+ cells not only acquired endothelial cell fate involved in angiogenesis, but also, CD34+ cells expressing Pi16 may transform into myofibroblast and thus participate in organ fibrosis.
Conclusion
A heterogeneous CD34+ cells serve as a contributor not only to endothelial regeneration but also a wound healing response that may provide therapeutic insights into fibrosis.








Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article and its supplementary information files, or are available from the corresponding author upon reasonable request.
Abbreviations
- CD:
-
Cluster of differentiation
- EPC:
-
Endothelial progenitor cell
- ScRNA-seq:
-
Single cell RNA sequencing
- MSC:
-
Mesenchymal stem cell
- CCA:
-
Canonical correlation analysis
- t-SNE:
-
T-distributed stochastic neighbor embedding
- α-SMA:
-
Alpha smooth muscle actin
- PDGFR:
-
Platelet-derived growth factor receptor
- AngII:
-
Angiotensin II
- UUO:
-
Unilateral ureteral obstruction
- BMT:
-
Bone-marrow transplantation
- EC:
-
Endothelial cell
- UMAP:
-
Uniform manifold approximation and projection
- IFN:
-
Interferon
- ECM:
-
Extracellular matrix
- CXCL:
-
C-X-C motif chemokine ligand
- FGF:
-
Fibroblast growth factor
- TGF:
-
Transforming growth factor
- IGF:
-
Insulin-like growth factor
- CTGF:
-
Connective tissue growth factor
- TAC:
-
Transverse aortic constriction
- CCL:
-
CC chemokine ligand
- VEGF:
-
Vascular endothelial growth factor
References
Asahara T, Murohara T, Sullivan A, Silver M, van der Zee R, Li T et al (1997) Isolation of putative progenitor endothelial cells for angiogenesis. Science 275:964–967
Kocher AA, Schuster MD, Szabolcs MJ, Takuma S, Burkhoff D, Wang J et al (2001) Neovascularization of ischemic myocardium by human bone-marrow-derived angioblasts prevents cardiomyocyte apoptosis, reduces remodeling and improves cardiac function. Nat Med 7:430–436
Pu X, Du L, Hu Y, Fan Y, Xu Q (2021) Stem/progenitor cells and pulmonary arterial hypertension. Arterioscler Thromb Vasc Biol 41:167–178
Werner N, Kosiol S, Schiegl T, Ahlers P, Walenta K, Link A et al (2005) Circulating endothelial progenitor cells and cardiovascular outcomes. N Engl J Med 353:999–1007
Losordo DW, Schatz RA, White CJ, Udelson JE, Veereshwarayya V, Durgin M et al (2007) Intramyocardial transplantation of autologous CD34+ stem cells for intractable angina: a phase I/IIa double-blind, randomized controlled trial. Circulation 115:3165–3172
Losordo DW, Henry TD, Davidson C, Sup Lee J, Costa MA, Bass T et al (2011) Intramyocardial, autologous CD34+ cell therapy for refractory angina. Circ Res 109:428–436
Henry TD, Schaer GL, Traverse JH, Povsic TJ, Davidson C, Lee JS et al (2016) Autologous CD34(+) cell therapy for refractory angina: 2-year outcomes from the ACT34-CMI study. Cell Transplant 25:1701–1711
Johnson GL, Henry TD, Povsic TJ, Losordo DW, Garberich RF, Stanberry LI et al (2020) CD34(+) cell therapy significantly reduces adverse cardiac events, health care expenditures, and mortality in patients with refractory angina. Stem Cells Transl Med 9:1147–1152
Henry TD, Losordo DW, Traverse JH, Schatz RA, Jolicoeur EM, Schaer GL et al (2018) Autologous CD34+ cell therapy improves exercise capacity, angina frequency and reduces mortality in no-option refractory angina: a patient-level pooled analysis of randomized double-blinded trials. Eur Heart J 39:2208–2216
Velagapudi P, Turagam M, Kolte D, Khera S, Hyder O, Gordon P et al (2019) Intramyocardial autologous CD34+ cell therapy for refractory angina: a meta-analysis of randomized controlled trials. Cardiovasc Revasc Med Including Mol Interv 20:215–219
Quyyumi AA, Waller EK, Murrow J, Esteves F, Galt J, Oshinski J et al (2011) CD34(+) cell infusion after ST elevation myocardial infarction is associated with improved perfusion and is dose dependent. Am Heart J 161:98–105
Losordo DW, Kibbe MR, Mendelsohn F, Marston W, Driver VR, Sharafuddin M et al (2012) A randomized, controlled pilot study of autologous CD34+ cell therapy for critical limb ischemia. Circ Cardiovasc Interv 5:821–830
Quyyumi AA, Vasquez A, Kereiakes DJ, Klapholz M, Schaer GL, Abdel-Latif A et al (2017) PreSERVE-AMI: a randomized, double-blind, placebo-controlled clinical trial of intracoronary administration of autologous CD34+ cells in patients with left ventricular dysfunction post STEMI. Circ Res 120:324–331
Rai B, Shukla J, Henry TD, Quesada O (2021) Angiogenic CD34 stem cell therapy in coronary microvascular repair—a systematic review. Cells 10:1137
Vrtovec B, Poglajen G, Lezaic L, Sever M, Domanovic D, Cernelc P et al (2013) Effects of intracoronary CD34+ stem cell transplantation in nonischemic dilated cardiomyopathy patients: 5-year follow-up. Circ Res 112:165–173
Hill JM, Zalos G, Halcox JP, Schenke WH, Waclawiw MA, Quyyumi AA et al (2003) Circulating endothelial progenitor cells, vascular function, and cardiovascular risk. N Engl J Med 348:593–600
Saito S, Krucoff MW, Nakamura S, Mehran R, Maehara A, Al-Khalidi HR et al (2018) Japan-United States of America Harmonized Assessment by Randomized Multicentre Study of OrbusNEich’s Combo StEnt (Japan-USA HARMONEE) study: primary results of the pivotal registration study of combined endothelial progenitor cell capture and drug-eluting stent in patients with ischaemic coronary disease and non-ST-elevation acute coronary syndrome. Eur Heart J 39:2460–2468
Meyer GP, Wollert KC, Lotz J, Pirr J, Rager U, Lippolt P et al (2009) Intracoronary bone marrow cell transfer after myocardial infarction: 5-year follow-up from the randomized-controlled BOOST trial. Eur Heart J 30:2978–2984
Wollert KC, Meyer GP, Muller-Ehmsen J, Tschope C, Bonarjee V, Larsen AI et al (2017) Intracoronary autologous bone marrow cell transfer after myocardial infarction: the BOOST-2 randomised placebo-controlled clinical trial. Eur Heart J 38:2936–2943
Beitnes JO, Hopp E, Lunde K, Solheim S, Arnesen H, Brinchmann JE et al (2009) Long-term results after intracoronary injection of autologous mononuclear bone marrow cells in acute myocardial infarction: the ASTAMI randomised, controlled study. Heart 95:1983–1989
Jiang L, Sun X, Deng J, Hu Y, Xu Q (2021) Different roles of stem/progenitor cells in vascular remodeling. Antioxid Redox Signal 35:192–203
Jiang L, Chen T, Sun S, Wang R, Deng J, Lyu L et al (2021) Nonbone marrow CD34(+) cells are crucial for endothelial repair of injured artery. Circ Res 129:e146–e165
Rockey DC, Bell PD, Hill JA (2015) Fibrosis–a common pathway to organ injury and failure. N Engl J Med 372:1138–1149
Henderson NC, Rieder F, Wynn TA (2020) Fibrosis: from mechanisms to medicines. Nature 587:555–566
McGinnis CS, Murrow LM, Gartner ZJ (2019) DoubletFinder: doublet detection in single-cell RNA sequencing data using artificial nearest neighbors. Cell Syst 8:329 e4-337 e4
Butler A, Hoffman P, Smibert P, Papalexi E, Satija R (2018) Integrating single-cell transcriptomic data across different conditions, technologies, and species. Nat Biotechnol 36:411–420
Stuart T, Butler A, Hoffman P, Hafemeister C, Papalexi E, Mauck WM 3rd et al (2019) Comprehensive integration of single-cell data. Cell 177:1888 e21-1902 e21
Yu G, Wang LG, Han Y, He QY (2012) clusterProfiler: an R package for comparing biological themes among gene clusters. OMICS 16:284–287
Qiu X, Mao Q, Tang Y, Wang L, Chawla R, Pliner HA et al (2017) Reversed graph embedding resolves complex single-cell trajectories. Nat Methods 14:979–982
Jin S, Guerrero-Juarez CF, Zhang L, Chang I, Ramos R, Kuan CH et al (2021) Inference and analysis of cell-cell communication using Cell Chat. Nat Commun 12:1088
Wang Y, Wang R, Zhang S, Song S, Jiang C, Han G et al (2019) iTALK: an R package to characterize and illustrate intercellular communication. bioRxiv. 18:359
Deng J, Ni Z, Gu W, Chen Q, Nowak WN, Chen T et al (2020) Single-cell gene profiling and lineage tracing analyses revealed novel mechanisms of endothelial repair by progenitors. Cell Mol Life Sci 77:5299–5320
LeBleu VS, Taduri G, O’Connell J, Teng Y, Cooke VG, Woda C et al (2013) Origin and function of myofibroblasts in kidney fibrosis. Nat Med 19:1047–1053
Buechler MB, Pradhan RN, Krishnamurty AT, Cox C, Calviello AK, Wang AW et al (2021) Cross-tissue organization of the fibroblast lineage. Nature 593:575–579
Tiedemann K, Boraschi-Diaz I, Rajakumar I, Kaur J, Roughley P, Reinhardt DP et al (2013) Fibrillin-1 directly regulates osteoclast formation and function by a dual mechanism. J Cell Sci 126:4187–4194
Dong XY, Yin JX, Zhang H, Liao Y (2022) High glucose stimulating ECM remodeling and an inflammatory phenotype in the IPFP via upregulation of MFAP5 expression. Biochem Biophys Res Commun 601:93–100
Ouchi N, Asaumi Y, Ohashi K, Higuchi A, Sono-Romanelli S, Oshima Y et al (2010) DIP2A functions as a FSTL1 receptor. J Biol Chem 285:7127–7134
Vasicek J, Balazi A, Tirpakova M, Svoradova A, Ondruska L, Parkanyi V et al (2021) Secretome analysis of rabbit and human mesenchymal stem and endothelial progenitor cells: a comparative study. Int J Mol Sci 22:12283
Fujisawa T, Tura-Ceide O, Hunter A, Mitchell A, Vesey A, Medine C et al (2019) Endothelial progenitor cells do not originate from the bone marrow. Circulation 140:1524–1526
Wang X, Wang R, Jiang L, Xu Q, Guo X (2021) Endothelial repair by stem and progenitor cells. J Mol Cell Cardiol 163:133–146
Acknowledgements
This work was supported by Alibaba-Zhejiang University Joint Research Center of Future Digital Healthcare and Alibaba Cloud. We are also grateful to the Core Facility Platform of Zhejiang University School of Medicine for technical assistance.
Funding
National Natural Science Foundation of China 31830039 (Q. Xu), 82030008 (Q. Xu). National Natural Science Foundation of China 82170489 and the Natural Science Foundation of Zhejiang Province (LR22H020001) (T. Chen). National Natural Science Foundation of China 81600339 and the Natural Science Foundation of Zhejiang Province LY19H020009 (X. Pu).
Author information
Authors and Affiliations
Contributions
Conceptualization: QX and HZ. Methodology: XP, PZ, XZ, and YH. Investigation: XP, PZ, XZ, HW, LD, HG, XS, and TC. Visualization: XP, PZ, and XZ. Supervision: QX, JZ, and HZ. Writing—original draft: XP, PZ, XZ, and QX. Writing—review and editing: YH, HW, LD, HG, XS, JZ, and TC. XP, TC, and QX acquired funding. All authors contributed to the article.
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Ethics approval
This study was performed in line with the principles of the Declaration of Helsinki. Human sample procedures had local ethical approval. All mice experiments were approved by the Institutional Animal Care and Use Committee of Zhejiang University School of Medicine.
Consent to participate
Not applicable.
Consent to publish
All authors have seen the manuscript and agree with publication.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pu, X., Zhu, P., Zhou, X. et al. CD34+ cell atlas of main organs implicates its impact on fibrosis. Cell. Mol. Life Sci. 79, 576 (2022). https://doi.org/10.1007/s00018-022-04606-6
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00018-022-04606-6