Abstract
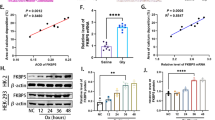
Recent evidence has suggested that recurrent urinary tract infection (UTI) can cause not only infection stones but also metabolic stones (e.g., those containing calcium oxalate monohydrate or COM). However, precise mechanisms underlying UTI-induced metabolic stones remained unknown. In this study, Escherichia coli, the most common bacterium found in recurrent UTI was used to establish the in vitro model for persistent infection of renal epithelial cells. The promoting effects of persistent E. coli infection on kidney stone formation were validated by COM crystal–cell adhesion assay, followed by immunofluorescence study for changes in surface expression of the known COM crystal receptors. Among the five receptors examined, only ezrin had significantly increased level on the surface of persistently infected cells without change in its total level. Such translocation of ezrin to apical membranes was confirmed by Western blotting of apical membrane and cytosolic fractions and confocal microscopic examination. Additionally, persistent infection increased phosphorylation (Thr567) of ezrin. However, all of these changes induced by persistent E. coli infection were significantly inhibited by small-interfering RNA (siRNA) specific for ezrin or a Rho-associated kinase (ROCK)-specific inhibitor (Y-27632). In summary, this study provides a piece of evidence demonstrating that persistent infection by E. coli, one of the non-urease-producing bacteria, may contribute to COM metabolic stone formation by translocation of ezrin to apical membranes, thereby promoting COM crystal–cell adhesion. Such ezrin translocation was mediated via Rho/ROCK signaling pathway. These findings may, at least in part, explain the pathogenic mechanisms underlying recurrent UTI-induced metabolic kidney stone disease.
Graphical abstract









Similar content being viewed by others
Availability of data and materials
All data generated or analysed during this study are included in this published article and its supplementary information files.
References
Norsworthy AN, Pearson MM (2017) From catheter to kidney stone: the uropathogenic lifestyle of Proteus mirabilis. Trends Microbiol 25(4):304–315. https://doi.org/10.1016/j.tim.2016.11.015
He Z, Jing Z, Jing-Cun Z, Chuan-Yi H, Fei G (2017) Compositional analysis of various layers of upper urinary tract stones by infrared spectroscopy. Exp Ther Med 14(4):3165–3169. https://doi.org/10.3892/etm.2017.4864
Tavichakorntrakool R, Prasongwattana V, Sungkeeree S, Saisud P, Sribenjalux P, Pimratana C, Bovornpadungkitti S, Sriboonlue P, Thongboonkerd V (2012) Extensive characterizations of bacteria isolated from catheterized urine and stone matrices in patients with nephrolithiasis. Nephrol Dial Transplant 27(11):4125–4130. https://doi.org/10.1093/ndt/gfs057
Abrahams HM, Stoller ML (2003) Infection and urinary stones. Curr Opin Urol 13(1):63–67. https://doi.org/10.1097/00042307-200301000-00011
Djojodimedjo T, Soebadi DM, Soetjipto (2013) Escherichia coli infection induces mucosal damage and expression of proteins promoting urinary stone formation. Urolithiasis 41(4):295–301. https://doi.org/10.1007/s00240-013-0577-4
Kodner CM, Thomas Gupton EK (2010) Recurrent urinary tract infections in women: diagnosis and management. Am Fam Physician 82(6):638–643
Hooton TM, Roberts PL, Stapleton AE (2021) Asymptomatic Bacteriuria and Pyuria in premenopausal women. Clin Infect Dis 72(8):1332–1338. https://doi.org/10.1093/cid/ciaa274
Chutipongtanate S, Sutthimethakorn S, Chiangjong W, Thongboonkerd V (2013) Bacteria can promote calcium oxalate crystal growth and aggregation. J Biol Inorg Chem 18(3):299–308. https://doi.org/10.1007/s00775-012-0974-0
Barr-Beare E, Saxena V, Hilt EE, Thomas-White K, Schober M, Li B, Becknell B, Hains DS, Wolfe AJ, Schwaderer AL (2015) The interaction between Enterobacteriaceae and calcium oxalate deposits. PLoS One 10(10):e0139575
Mulvey MA, Schilling JD, Hultgren SJ (2001) Establishment of a persistent Escherichia coli reservoir during the acute phase of a bladder infection. Infect Immun 69(7):4572–4579. https://doi.org/10.1128/IAI.69.7.4572-4579.2001
Somsuan K, Peerapen P, Boonmark W, Plumworasawat S, Samol R, Sakulsak N, Thongboonkerd V (2019) ARID1A knockdown triggers epithelial-mesenchymal transition and carcinogenesis features of renal cells: role in renal cell carcinoma. FASEB J 33(11):12226–12239. https://doi.org/10.1096/fj.201802720RR
Chaiyarit S, Thongboonkerd V (2012) Changes in mitochondrial proteome of renal tubular cells induced by calcium oxalate monohydrate crystal adhesion and internalization are related to mitochondrial dysfunction. J Proteome Res 11(6):3269–3280. https://doi.org/10.1021/pr300018c
Thongboonkerd V, Semangoen T, Chutipongtanate S (2006) Factors determining types and morphologies of calcium oxalate crystals: molar concentrations, buffering, pH, stirring and temperature. Clin Chim Acta 367(1–2):120–131. https://doi.org/10.1016/j.cca.2005.11.033
Thongboonkerd V, Chutipongtanate S, Semangoen T, Malasit P (2008) Urinary trefoil factor 1 is a novel potent inhibitor of calcium oxalate crystal growth and aggregation. J Urol 179(4):1615–1619. https://doi.org/10.1016/j.juro.2007.11.041
Chaiyarit S, Mungdee S, Thongboonkerd V (2010) Non-radioactive labelling of calcium oxalate crystals for investigations of crystal-cell interaction and internalization. Anal Methods 2(10):1536–1541. https://doi.org/10.1039/C0AY00321B
Peerapen P, Thongboonkerd V (2020) Differential bound proteins and adhesive capabilities of calcium oxalate monohydrate crystals with various sizes. Int J Biol Macromol 163:2210–2223. https://doi.org/10.1016/j.ijbiomac.2020.09.085
Vinaiphat A, Thongboonkerd V (2018) Characterizations of PMCA2-interacting complex and its role as a calcium oxalate crystal-binding protein. Cell Mol Life Sci 75(8):1461–1482. https://doi.org/10.1007/s00018-017-2699-2
Fong-ngern K, Peerapen P, Sinchaikul S, Chen ST, Thongboonkerd V (2011) Large-scale identification of calcium oxalate monohydrate crystal-binding proteins on apical membrane of distal renal tubular epithelial cells. J Proteome Res 10(10):4463–4477. https://doi.org/10.1021/pr2006878
Peerapen P, Thongboonkerd V (2016) Caffeine prevents kidney stone formation by translocation of apical surface annexin A1 crystal-binding protein into cytoplasm: in vitro evidence. Sci Rep 6:38536. https://doi.org/10.1038/srep38536
Fong-ngern K, Sueksakit K, Thongboonkerd V (2016) Surface heat shock protein 90 serves as a potential receptor for calcium oxalate crystal on apical membrane of renal tubular epithelial cells. J Biol Inorg Chem 21(4):463–474. https://doi.org/10.1007/s00775-016-1355-x
Fong-ngern K, Chiangjong W, Thongboonkerd V (2009) Peeling as a novel, simple, and effective method for isolation of apical membrane from intact polarized epithelial cells. Anal Biochem 395(1):25–32. https://doi.org/10.1016/j.ab.2009.08.007
Grant SS, Hung DT (2013) Persistent bacterial infections, antibiotic tolerance, and the oxidative stress response. Virulence 4(4):273–283. https://doi.org/10.4161/viru.23987
Wagenlehner F, Nicolle L, Bartoletti R, Gales AC, Grigoryan L, Huang H, Hooton T, Lopardo G, Naber K, Poojary A, Stapleton A, Talan DA, Saucedo JT, Wilcox MH, Yamamoto S, Yang SS, Lee SJ (2021) A global perspective on improving patient care in uncomplicated urinary tract infection: expert consensus and practical guidance. J Glob Antimicrob Resist 28:18–29. https://doi.org/10.1016/j.jgar.2021.11.008
Fisher RA, Gollan B, Helaine S (2017) Persistent bacterial infections and persister cells. Nat Rev Microbiol 15(8):453–464. https://doi.org/10.1038/nrmicro.2017.42
Hebert M, Potin S, Sebbagh M, Bertoglio J, Breard J, Hamelin J (2008) Rho-ROCK-dependent ezrin-radixin-moesin phosphorylation regulates Fas-mediated apoptosis in Jurkat cells. J Immunol 181(9):5963–5973. https://doi.org/10.4049/jimmunol.181.9.5963
Bretscher A, Reczek D, Berryman M (1997) Ezrin: a protein requiring conformational activation to link microfilaments to the plasma membrane in the assembly of cell surface structures. J Cell Sci 110(Pt 24):3011–3018
Chen Y, Wang D, Guo Z, Zhao J, Wu B, Deng H, Zhou T, Xiang H, Gao F, Yu X, Liao J, Ward T, Xia P, Emenari C, Ding X, Thompson W, Ma K, Zhu J, Aikhionbare F, Dou K, Cheng SY, Yao X (2011) Rho kinase phosphorylation promotes ezrin-mediated metastasis in hepatocellular carcinoma. Cancer Res 71(5):1721–1729. https://doi.org/10.1158/0008-5472.CAN-09-4683
Glover M, Moreira CG, Sperandio V, Zimmern P (2014) Recurrent urinary tract infections in healthy and nonpregnant women. Urol Sci 25(1):1–8. https://doi.org/10.1016/j.urols.2013.11.007
Delcaru C, Alexandru I, Podgoreanu P, Grosu M, Stavropoulos E, Chifiriuc MC, Lazar V (2016) Microbial biofilms in urinary tract infections and prostatitis: etiology, pathogenicity, and combating strategies. Pathogens. https://doi.org/10.3390/pathogens5040065
Koksoy AY, Gorukmez O (2021) Urinary stone and infection does not always mean a “chicken and egg dilemma”: answers. Pediatr Nephrol 36(8):2295–2297. https://doi.org/10.1007/s00467-021-04979-z
Cetin N, Gencler A, Kavaz Tufan A (2020) Risk factors for development of urinary tract infection in children with nephrolithiasis. J Paediatr Child Health 56(1):76–80. https://doi.org/10.1111/jpc.14495
Bauza JL, Pieras EC, Grases F, Tubau V, Guimera J, Sabate XA, Piza P (2018) Urinary tract infection’s etiopathogenic role in nephrolithiasis formation. Med Hypotheses 118:34–35. https://doi.org/10.1016/j.mehy.2018.06.002
Naas T, Al-Agili S, Bashir O (2001) Urinary calculi: bacteriological and chemical association. East Mediterr Health J 7(4–5):763–770
Shafi H, Shahandeh Z, Heidari B, Sedigiani F, Ramaji AA, Pasha YR, Kassaeian AA, Pasha AA, Mir MM (2013) Bacteriological study and structural composition of staghorn stones removed by the anatrophic nephrolithotomic procedure. Saudi J Kidney Dis Transpl 24(2):418–423. https://doi.org/10.4103/1319-2442.109623
Chen D, Zhang Y, Huang J, Liang X, Zeng T, Lan C, Duan X, Zhao Z, Zeng G, Tiselius HG, Lu X, Wu W (2018) The analysis of microbial spectrum and antibiotic resistance of uropathogens isolated from patients with urinary stones. Int J Clin Pract 72(6):e13205. https://doi.org/10.1111/ijcp.13205
Schwaderer AL, Wolfe AJ (2017) The association between bacteria and urinary stones. Ann Transl Med 5(2):32. https://doi.org/10.21037/atm.2016.11.73
Tavichakorntrakool R, Boonsiri P, Prasongwatana V, Lulitanond A, Wongkham C, Thongboonkerd V (2017) Differential colony size, cell length, and cellular proteome of Escherichia coli isolated from urine vs. stone nidus of kidney stone patients. Clin Chim Acta 466:112–119. https://doi.org/10.1016/j.cca.2016.12.018
Nilekani S, SivaRaman C (1983) Purification and properties of citrate lyase from Escherichia coli. Biochemistry 22(20):4657–4663. https://doi.org/10.1021/bi00289a008
Srivastava T, Winston MJ, Auron A, Alon US (2009) Urine calcium/citrate ratio in children with hypercalciuric stones. Pediatr Res 66(1):85–90. https://doi.org/10.1203/PDR.0b013e3181a2939e
Schilling JD, Lorenz RG, Hultgren SJ (2002) Effect of trimethoprim-sulfamethoxazole on recurrent bacteriuria and bacterial persistence in mice infected with uropathogenic Escherichia coli. Infect Immun 70(12):7042–7049. https://doi.org/10.1128/IAI.70.12.7042-7049.2002
Thongboonkerd V (2019) Proteomics of crystal-cell interactions: a model for kidney stone research. Cells 8(9):1076. https://doi.org/10.3390/cells8091076
Lieske JC, Leonard R, Swift H, Toback FG (1996) Adhesion of calcium oxalate monohydrate crystals to anionic sites on the surface of renal epithelial cells. Am J Physiol 270(1 Pt 2):F192–F199
Kanlaya R, Sintiprungrat K, Chaiyarit S, Thongboonkerd V (2013) Macropinocytosis is the major mechanism for endocytosis of calcium oxalate crystals into renal tubular cells. Cell Biochem Biophys 67(3):1171–1179. https://doi.org/10.1007/s12013-013-9630-8
Lieske JC, Deganello S, Toback FG (1999) Cell-crystal interactions and kidney stone formation. Nephron 81(Suppl 1):8–17
Chutipongtanate S, Fong-ngern K, Peerapen P, Thongboonkerd V (2012) High calcium enhances calcium oxalate crystal binding capacity of renal tubular cells via increased surface annexin A1 but impairs their proliferation and healing. J Proteome Res 11(7):3650–3663. https://doi.org/10.1021/pr3000738
Kanlaya R, Fong-ngern K, Thongboonkerd V (2013) Cellular adaptive response of distal renal tubular cells to high-oxalate environment highlights surface alpha-enolase as the enhancer of calcium oxalate monohydrate crystal adhesion. J Proteomics 80:55–65. https://doi.org/10.1016/j.jprot.2013.01.001
Manissorn J, Khamchun S, Vinaiphat A, Thongboonkerd V (2016) Alpha-tubulin enhanced renal tubular cell proliferation and tissue repair but reduced cell death and cell-crystal adhesion. Sci Rep 6:28808. https://doi.org/10.1038/srep28808
Pongsakul N, Vinaiphat A, Chanchaem P, Fong-ngern K, Thongboonkerd V (2016) Lamin A/C in renal tubular cells is important for tissue repair, cell proliferation, and calcium oxalate crystal adhesion, and is associated with potential crystal receptors. FASEB J 30(10):3368–3377. https://doi.org/10.1096/fj.201600426R
Fong-ngern K, Thongboonkerd V (2016) α-enolase on apical surface of renal tubular epithelial cells serves as a calcium oxalate crystal receptor. Sci Rep 6:36103. https://doi.org/10.1038/srep36103
Peerapen P, Thongboonkerd V (2019) Protective cellular mechanism of estrogen against kidney stone formation: a proteomics approach and functional validation. Proteomics 19(19):e1900095. https://doi.org/10.1002/pmic.201900095
Sueksakit K, Thongboonkerd V (2019) Protective effects of finasteride against testosterone-induced calcium oxalate crystallization and crystal-cell adhesion. J Biol Inorg Chem 24(7):973–983. https://doi.org/10.1007/s00775-019-01692-z
Sutthimethakorn S, Thongboonkerd V (2020) Effects of high-dose uric acid on cellular proteome, intracellular ATP, tissue repairing capability and calcium oxalate crystal-binding capability of renal tubular cells: Implications to hyperuricosuria-induced kidney stone disease. Chem Biol Interact 331:109270. https://doi.org/10.1016/j.cbi.2020.109270
Garcia-Ortiz A, Serrador JM (2020) ERM proteins at the crossroad of leukocyte polarization, migration and intercellular adhesion. Int J Mol Sci. https://doi.org/10.3390/ijms21041502
Gautreau A, Louvard D, Arpin M (2000) Morphogenic effects of ezrin require a phosphorylation-induced transition from oligomers to monomers at the plasma membrane. J Cell Biol 150(1):193–203. https://doi.org/10.1083/jcb.150.1.193
Amano M, Nakayama M, Kaibuchi K (2010) Rho-kinase/ROCK: a key regulator of the cytoskeleton and cell polarity. Cytoskeleton (Hoboken) 67(9):545–554. https://doi.org/10.1002/cm.20472
Kotani H, Takaishi K, Sasaki T, Takai Y (1997) Rho regulates association of both the ERM family and vinculin with the plasma membrane in MDCK cells. Oncogene 14(14):1705–1713. https://doi.org/10.1038/sj.onc.1200998
Matsui T, Maeda M, Doi Y, Yonemura S, Amano M, Kaibuchi K, Tsukita S, Tsukita S (1998) Rho-kinase phosphorylates COOH-terminal threonines of ezrin/radixin/moesin (ERM) proteins and regulates their head-to-tail association. J Cell Biol 140(3):647–657. https://doi.org/10.1083/jcb.140.3.647
Simonovic I, Arpin M, Koutsouris A, Falk-Krzesinski HJ, Hecht G (2001) Enteropathogenic Escherichia coli activates ezrin, which participates in disruption of tight junction barrier function. Infect Immun 69(9):5679–5688. https://doi.org/10.1128/IAI.69.9.5679-5688.2001
Skoudy A, Nhieu GT, Mantis N, Arpin M, Mounier J, Gounon P, Sansonetti P (1999) A functional role for ezrin during Shigella flexneri entry into epithelial cells. J Cell Sci 112(Pt 13):2059–2068. https://doi.org/10.1242/jcs.112.13.2059
Pust S, Morrison H, Wehland J, Sechi AS, Herrlich P (2005) Listeria monocytogenes exploits ERM protein functions to efficiently spread from cell to cell. EMBO J 24(6):1287–1300. https://doi.org/10.1038/sj.emboj.7600595
Berger CN, Crepin VF, Jepson MA, Arbeloa A, Frankel G (2009) The mechanisms used by enteropathogenic Escherichia coli to control filopodia dynamics. Cell Microbiol 11(2):309–322. https://doi.org/10.1111/j.1462-5822.2008.01254.x
Swanson KA, Crane DD, Caldwell HD (2007) Chlamydia trachomatis species-specific induction of ezrin tyrosine phosphorylation functions in pathogen entry. Infect Immun 75(12):5669–5677. https://doi.org/10.1128/IAI.01096-07
Di Pietro C, Zhang PX, O’Rourke TK, Murray TS, Wang L, Britto CJ, Koff JL, Krause DS, Egan ME, Bruscia EM (2017) Ezrin links CFTR to TLR4 signaling to orchestrate anti-bacterial immune response in macrophages. Sci Rep 7(1):10882. https://doi.org/10.1038/s41598-017-11012-7
Jianjun Z, Baochun Z, Limei M, Lijun L (2021) Exploring the beneficial role of ROCK inhibitors in sepsis-induced cerebral and cognitive injury in rats. Fundam Clin Pharmacol 35(5):882–891. https://doi.org/10.1111/fcp.12645
Matsuzawa T, Kuwae A, Yoshida S, Sasakawa C, Abe A (2004) Enteropathogenic Escherichia coli activates the RhoA signaling pathway via the stimulation of GEF-H1. EMBO J 23(17):3570–3582. https://doi.org/10.1038/sj.emboj.7600359
Bulgin R, Raymond B, Garnett JA, Frankel G, Crepin VF, Berger CN, Arbeloa A (2010) Bacterial guanine nucleotide exchange factors SopE-like and WxxxE effectors. Infect Immun 78(4):1417–1425. https://doi.org/10.1128/IAI.01250-09
Woolery AR, Yu X, LaBaer J, Orth K (2014) AMPylation of Rho GTPases subverts multiple host signaling processes. J Biol Chem 289(47):32977–32988. https://doi.org/10.1074/jbc.M114.601310
Acknowledgements
We are grateful to Sutthirat Srisuwan and Chonnicha Subkod for their assistance.
Funding
This study was supported by Mahidol University research grant.
Author information
Authors and Affiliations
Contributions
RK and VT designed research; RK performed experiments; RK and VT analyzed data; RK and VT wrote the manuscript; All authors reviewed and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare NO conflict of interest.
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kanlaya, R., Thongboonkerd, V. Persistent Escherichia coli infection in renal tubular cells enhances calcium oxalate crystal–cell adhesion by inducing ezrin translocation to apical membranes via Rho/ROCK pathway. Cell. Mol. Life Sci. 79, 381 (2022). https://doi.org/10.1007/s00018-022-04414-y
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00018-022-04414-y