Abstract
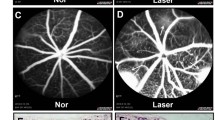
Proliferative vitreoretinal diseases such as diabetic retinopathy, proliferative vitreoretinopathy (PVR), and age-related macular degeneration are a leading cause of decreased vision and blindness in developed countries. In these diseases, retinal fibro(vascular) membrane (FVM) formation above and beneath the retina plays an important role. Gene expression profiling of human FVMs revealed significant upregulation of periostin. Subsequent analyses demonstrated increased periostin expression in the vitreous of patients with both proliferative diabetic retinopathy and PVR. Immunohistochemical analysis showed co-localization of periostin with α-SMA and M2 macrophage markers in FVMs. In vitro, periostin blockade inhibited migration and adhesion induced by PVR vitreous and transforming growth factor-β2 (TGF-β2). In vivo, a novel single-stranded RNAi agent targeting periostin showed the inhibitory effect on experimental retinal and choroidal FVM formation without affecting the viability of retinal cells. These results indicated that periostin is a pivotal molecule for FVM formation and a promising therapeutic target for these proliferative vitreoretinal diseases.




Similar content being viewed by others
References
Yoshida S (2014) Identification of molecular targets for intraocular proliferative diseases using genomicapproaches. J Jpn Ophthalmol Soc 118:241–282
Hiscott P, Wong D, Grierson I (2000) Challenges in ophthalmic pathology: the vitreoretinal membrane biopsy. Eye 14(Pt 4):549–559
Nakama T, Yoshida S, Ishikawa K, Kobayashi Y, Zhou Y, Nakao S, Sassa Y, Oshima Y, Takao K, Shimahara A, Yoshikawa K, Hamasaki T, Ohgi T, Hayashi H, Matsuda A, Kudo A, Nozaki M, Ogura Y, Kuroda M, Ishibashi T (2015) Inhibition of choroidal fibrovascular membrane formation by new class of rna interference therapeutic agent targeting periostin. Gene Ther 22:127–137
Kirchhof B (2004) Strategies to influence PVR development. Graefes Arch Clin Exp Ophthalmol 242:699–703
Kobayashi Y, Yoshida S, Zhou Y, Nakama T, Ishikawa K, Arima M, Nakao S, Sassa Y, Takeda A, Hisatomi T, Ikeda Y, Matsuda A, Sonoda KH, Ishibashi T (2016) Tenascin-c promotes angiogenesis in fibrovascular membranes in eyes with proliferative diabetic retinopathy. Mol Vis 22:436–445
Yoshida S, Ogura A, Ishikawa K, Yoshida A, Kohno R, Yamaji Y, Ikeo K, Gojobori T, Kono T, Ishibashi T (2010) Gene expression profile of fibrovascular membranes from patients with proliferative diabetic retinopathy. Br J Ophthalmol 94:795–801
Ishikawa K, Yoshida S, Kobayashi Y, Zhou Y, Nakama T, Nakao S, Sassa Y, Oshima Y, Niiro H, Akashi K, Kono T, Ishibashi T (2015) Microarray analysis of gene expression in fibrovascular membranes excised from patients with proliferative diabetic retinopathy. Investig Ophthalmol Vis Sci 56:932–946
Asato R, Yoshida S, Ogura A, Nakama T, Ishikawa K, Nakao S, Sassa Y, Enaida H, Oshima Y, Ikeo K, Gojobori T, Kono T, Ishibashi T (2013) Comparison of gene expression profile of epiretinal membranes obtained from eyes with proliferative vitreoretinopathy to that of secondary epiretinal membranes. PLoS One 8:e54191
Rios H, Koushik SV, Wang H, Wang J, Zhou HM, Lindsley A, Rogers R, Chen Z, Maeda M, Kruzynska-Frejtag A, Feng JQ, Conway SJ (2005) Periostin null mice exhibit dwarfism, incisor enamel defects, and an early-onset periodontal disease-like phenotype. Mol Cell Biol 25:11131–11144
Snider P, Hinton RB, Moreno-Rodriguez RA, Wang J, Rogers R, Lindsley A, Li F, Ingram DA, Menick D, Field L, Firulli AB, Molkentin JD, Markwald R, Conway SJ (2008) Periostin is required for maturation and extracellular matrix stabilization of noncardiomyocyte lineages of the heart. Circ Res 102:752–760
Malanchi I, Santamaria-Martinez A, Susanto E, Peng H, Lehr HA, Delaloye JF, Huelsken J (2011) Interactions between cancer stem cells and their niche govern metastatic colonization. Nature 481:85–89
Shimazaki M, Nakamura K, Kii I, Kashima T, Amizuka N, Li M, Saito M, Fukuda K, Nishiyama T, Kitajima S, Saga Y, Fukayama M, Sata M, Kudo A (2008) Periostin is essential for cardiac healing after acute myocardial infarction. J Exp Med 205:295–303
Conway SJ, Molkentin JD (2008) Periostin as a heterofunctional regulator of cardiac development and disease. Curr Genomics 9:548–555
Ontsuka K, Kotobuki Y, Shiraishi H, Serada S, Ohta S, Tanemura A, Yang L, Fujimoto M, Arima K, Suzuki S, Murota H, Toda S, Kudo A, Conway SJ, Narisawa Y, Katayama I, Izuhara K, Naka T (2012) Periostin, a matricellular protein, accelerates cutaneous wound repair by activating dermal fibroblasts. Exp Dermatol 21:331–336
Masuoka M, Shiraishi H, Ohta S, Suzuki S, Arima K, Aoki S, Toda S, Inagaki N, Kurihara Y, Hayashida S, Takeuchi S, Koike K, Ono J, Noshiro H, Furue M, Conway SJ, Narisawa Y, Izuhara K (2012) Periostin promotes chronic allergic inflammation in response to Th2 cytokines. J Clin Investig 122:2590–2600
Sivaprasad S, Gupta B, Crosby-Nwaobi R, Evans J (2012) Prevalence of diabetic retinopathy in various ethnic groups: a worldwide perspective. Surv Ophthalmol 57:347–370
Yoshida A, Yoshida S, Ishibashi T, Inomata H (1999) Intraocular neovascularization. Histol Histopathol 14:1287–1294
Yoshida S, Kubo Y, Kobayashi Y, Zhou Y, Nakama T, Yamaguchi M, Tachibana T, Ishikawa K, Arita R, Nakao S, Sassa Y, Oshima Y, Kono T, Ishibashi T (2015) Increased vitreous concentrations of MCP-1 and IL-6 after vitrectomy in patients with proliferative diabetic retinopathy: possible association with postoperative macular oedema. Br J Ophthalmol 99:960–966
Yoshida S, Nakama T, Ishikawa K, Arima M, Tachibana T, Nakao S, Sassa Y, Yasuda M, Enaida H, Oshima Y, Kono T, Ishibashi T (2012) Antiangiogenic shift in vitreous after vitrectomy in patients with proliferative diabetic retinopathy. Investig Ophthalmol Vis Sci 53:6997–7003
Tachibana T, Yoshida S, Kubo Y, Koayashi Y, Nakama T, Ishikawa K, Nakao S, Izuhara K, Kono T, Ishibashi T (2016) Reduced vitreal concentration of periostin after vitrectomy in patients with proliferative diabetic retinopathy. Acta Ophthalmol 94:e81–e82
Yoshida S, Ishikawa K, Matsumoto T, Yoshida A, Ishibashi T, Kono T (2010) Reduced concentrations of angiogenesis-related factors in vitreous after vitrectomy in patients with proliferative diabetic retinopathy. Graefes Arch Clin Exp Ophthalmol 248:799–804
Simo R, Carrasco E, Garcia-Ramirez M, Hernandez C (2006) Angiogenic and antiangiogenic factors in proliferative diabetic retinopathy. Curr Diabetes Rev 2:71–98
Nakama T, Yoshida S, Ishikawa K, Kobayashi Y, Abe T, Kiyonari H, Shioi G, Katsuragi N, Ishibashi T, Morishita R, Taniyama Y (2016) Different roles played by periostin splice variants in retinal neovascularization. Exp Eye Res 153:133–140
Yoshida S, Ishikawa K, Asato R, Arima M, Sassa Y, Yoshida A, Yoshikawa H, Narukawa K, Obika S, Ono J, Ohta S, Izuhara K, Kono T, Ishibashi T (2011) Increased expression of periostin in vitreous and fibrovascular membranes obtained from patients with proliferative diabetic retinopathy. Investig Ophthalmol Vis Sci 52:5670–5678
Nakama T, Yoshida S, Ishikawa K, Kubo Y, Kobayashi Y, Zhou Y, Nakao S, Hisatomi T, Ikeda Y, Takao K, Yoshikawa K, Matsuda A, Ono J, Ohta S, Izuhara K, Kudo A, Sonoda KH, Ishibashi T (2017) Therapeutic effect of novel single-stranded RNAi agent targeting periostin in eyes with retinal neovascularization. Mol Ther Nucleic Acids 6:279–289
Ishikawa K, Yoshida S, Nakao S, Sassa Y, Asato R, Kohno R, Arima M, Kita T, Yoshida A, Ohuchida K, Ishibashi T (2012) Bone marrow-derived monocyte lineage cells recruited by MIP-1beta promote physiological revascularization in mouse model of oxygen-induced retinopathy. Lab Investig 92:91–101
Snead DR, James S, Snead MP (2008) Pathological changes in the vitreoretinal junction 1: epiretinal membrane formation. Eye 22:1310–1317
Sunderkotter C, Beil W, Roth J, Sorg C (1991) Cellular events associated with inflammatory angiogenesis in the mouse cornea. Am J Pathol 138:931–939
Yoshida S, Yoshida A, Ishibashi T, Elner SG, Elner VM (2003) Role of MCP-1 and MIP-1alpha in retinal neovascularization during postischemic inflammation in a mouse model of retinal neovascularization. J Leukoc Biol 73:137–144
Ishikawa K, Yoshida S, Kadota K, Nakamura T, Niiro H, Arakawa S, Yoshida A, Akashi K, Ishibashi T (2010) Gene expression profile of hyperoxic and hypoxic retinas in a mouse model of oxygen-induced retinopathy. Investig Ophthalmol Vis Sci 51:4307–4319
Martinez FO, Helming L, Gordon S (2009) Alternative activation of macrophages: an immunologic functional perspective. Annu Rev Immunol 27:451–483
Sica A, Mantovani A (2012) Macrophage plasticity and polarization: in vivo veritas. J Clin Investig 122:787–795
Kobayashi Y, Yoshida S, Nakama T, Zhou Y, Ishikawa K, Arita R, Nakao S, Miyazaki M, Sassa Y, Oshima Y, Izuhara K, Kono T, Ishibashi T (2015) Overexpression of CD163 in vitreous and fibrovascular membranes of patients with proliferative diabetic retinopathy: possible involvement of periostin. Br J Ophthalmol 99:451–456
Mantovani A, Biswas SK, Galdiero MR, Sica A, Locati M (2013) Macrophage plasticity and polarization in tissue repair and remodelling. J Pathol 229:176–185
Yoshida S, Kobayashi Y, Nakama T, Zhou Y, Ishikawa K, Arita R, Nakao S, Miyazaki M, Sassa Y, Oshima Y, Izuhara K, Kono T, Ishibashi T (2015) Increased expression of M-CSF and IL-13 in vitreous of patients with proliferative diabetic retinopathy: implications for M2 macrophage-involving fibrovascular membrane formation. Br J Ophthalmol 99:629–634
Liu W, Xu GZ, Jiang CH, Da CD (2009) Expression of macrophage colony-stimulating factor (M-CSF) and its receptor in streptozotocin-induced diabetic rats. Curr Eye Res 34:123–133
Zhou Y, Yoshida S, Nakao S, Yoshimura T, Kobayashi Y, Nakama T, Kubo Y, Miyawaki K, Yamaguchi M, Ishikawa K, Oshima Y, Akashi K, Ishibashi T (2015) M2 macrophages enhance pathological neovascularization in the mouse model of oxygen-induced retinopathy. Investig Ophthalmol Vis Sci 56:4767–4777
Zurawski SM, Vega F Jr, Huyghe B, Zurawski G (1993) Receptors for interleukin-13 and interleukin-4 are complex and share a novel component that functions in signal transduction. EMBO J 12:2663–2670
O’Reilly S (2013) Role of interleukin-13 in fibrosis, particularly systemic sclerosis. Biofactors 39:593–596
Leiderman YI, Miller JW (2009) Proliferative vitreoretinopathy: pathobiology and therapeutic targets. Semin Ophthalmol 24:62–69
Hiscott PS, Grierson I, McLeod D (1984) Retinal pigment epithelial cells in epiretinal membranes: an immunohistochemical study. Br J Ophthalmol 68:708–715
Ishikawa K, Yoshida S, Nakao S, Nakama T, Kita T, Asato R, Sassa Y, Arita R, Miyazaki M, Enaida H, Oshima Y, Murakami N, Niiro H, Ono J, Matsuda A, Goto Y, Akashi K, Izuhara K, Kudo A, Kono T, Hafezi-Moghadam A, Ishibashi T (2014) Periostin promotes the generation of fibrous membranes in proliferative vitreoretinopathy. FASEB J 28:131–142
Kita T, Hata Y, Arita R, Kawahara S, Miura M, Nakao S, Mochizuki Y, Enaida H, Goto Y, Shimokawa H, Hafezi-Moghadam A, Ishibashi T (2008) Role of TGF-beta in proliferative vitreoretinal diseases and rock as a therapeutic target. Proc Natl Acad Sci USA 105:17504–17509
Banerjee S, Savant V, Scott RA, Curnow SJ, Wallace GR, Murray PI (2007) Multiplex bead analysis of vitreous humor of patients with vitreoretinal disorders. Investig Ophthalmol Vis Sci 48:2203–2207
Harada C, Mitamura Y, Harada T (2006) The role of cytokines and trophic factors in epiretinal membranes: involvement of signal transduction in glial cells. Prog Retin Eye Res 25:149–164
He S, Chen Y, Khankan R, Barron E, Burton R, Zhu D, Ryan SJ, Oliver N, Hinton DR (2008) Connective tissue growth factor as a mediator of intraocular fibrosis. Investig Ophthalmol Vis Sci 49:4078–4088
Elner SG, Elner VM, Jaffe GJ, Stuart A, Kunkel SL, Strieter RM (1995) Cytokines in proliferative diabetic retinopathy and proliferative vitreoretinopathy. Curr Eye Res 14:1045–1053
Pennock S, Rheaume MA, Mukai S, Kazlauskas A (2011) A novel strategy to develop therapeutic approaches to prevent proliferative vitreoretinopathy. Am J Pathol 179:2931–2940
Li G, Oparil S, Sanders JM, Zhang L, Dai M, Chen LB, Conway SJ, McNamara CA, Sarembock IJ (2006) Phosphatidylinositol-3-kinase signaling mediates vascular smooth muscle cell expression of periostin in vivo and in vitro. Atherosclerosis 188:292–300
Dangaria SJ, Ito Y, Walker C, Druzinsky R, Luan X, Diekwisch TG (2009) Extracellular matrix-mediated differentiation of periodontal progenitor cells. Differentiation 78:79–90
de Jong PT (2006) Age-related macular degeneration. N Engl J Med 355:1474–1485
Schlingemann RO (2004) Role of growth factors and the wound healing response in age-related macular degeneration. Graefes Arch Clin Exp Ophthalmol 242:91–101
Bloch SB, Lund-Andersen H, Sander B, Larsen M (2013) Subfoveal fibrosis in eyes with neovascular age-related macular degeneration treated with intravitreal ranibizumab. Am J Ophthalmol 156(116–124):e111
Daniel E, Toth CA, Grunwald JE, Jaffe GJ, Martin DF, Fine SL, Huang J, Ying GS, Hagstrom SA, Winter K, Maguire MG, Comparison of Age-related Macular Degeneration Treatments Trials Research G (2014) Risk of scar in the comparison of age-related macular degeneration treatments trials. Ophthalmology 121:656–666
Kobayashi Y, Yoshida S, Zhou Y, Nakama T, Ishikawa K, Kubo Y, Arima M, Nakao S, Hisatomi T, Ikeda Y, Matsuda A, Sonoda KH, Ishibashi T (2016) Tenascin-c secreted by transdifferentiated retinal pigment epithelial cells promotes choroidal neovascularization via integrin alphav. Lab Investig 96:1178–1188
Elbashir SM, Harborth J, Lendeckel W, Yalcin A, Weber K, Tuschl T (2001) Duplexes of 21-nucleotide rnas mediate rna interference in cultured mammalian cells. Nature 411:494–498
Pecot CV, Calin GA, Coleman RL, Lopez-Berestein G, Sood AK (2011) Rna interference in the clinic: challenges and future directions. Nat Rev Cancer 11:59–67
Kleinman ME, Yamada K, Takeda A, Chandrasekaran V, Nozaki M, Baffi JZ, Albuquerque RJ, Yamasaki S, Itaya M, Pan Y, Appukuttan B, Gibbs D, Yang Z, Kariko K, Ambati BK, Wilgus TA, DiPietro LA, Sakurai E, Zhang K, Smith JR, Taylor EW, Ambati J (2008) Sequence- and target-independent angiogenesis suppression by sirna via tlr3. Nature 452:591–597
Yang Z, Stratton C, Francis PJ, Kleinman ME, Tan PL, Gibbs D, Tong Z, Chen H, Constantine R, Yang X, Chen Y, Zeng J, Davey L, Ma X, Hau VS, Wang C, Harmon J, Buehler J, Pearson E, Patel S, Kaminoh Y, Watkins S, Luo L, Zabriskie NA, Bernstein PS, Cho W, Schwager A, Hinton DR, Klein ML, Hamon SC, Simmons E, Yu B, Campochiaro B, Sunness JS, Campochiaro P, Jorde L, Parmigiani G, Zack DJ, Katsanis N, Ambati J, Zhang K (2008) Toll-like receptor 3 and geographic atrophy in age-related macular degeneration. N Engl J Med 359:1456–1463
Cho WG, Albuquerque RJ, Kleinman ME, Tarallo V, Greco A, Nozaki M, Green MG, Baffi JZ, Ambati BK, De Falco M, Alexander JS, Brunetti A, De Falco S, Ambati J (2009) Small interfering RNA-induced TLR3 activation inhibits blood and lymphatic vessel growth. Proc Natl Acad Sci USA 106:7137–7142
Hamasaki T, Suzuki H, Shirohzu H, Matsumoto T, D’Alessandro-Gabazza CN, Gil-Bernabe P, Boveda-Ruiz D, Naito M, Kobayashi T, Toda M, Mizutani T, Taguchi O, Morser J, Eguchi Y, Kuroda M, Ochiya T, Hayashi H, Gabazza EC, Ohgi T (2012) Efficacy of a novel class of rna interference therapeutic agents. PLoS One 7:e42655
Fujita Y, Takeshita F, Mizutani T, Ohgi T, Kuwano K, Ochiya T (2013) A novel platform to enable inhaled naked RNAi medicine for lung cancer. Sci Rep 3:3325
Takanashi M, Sudo K, Ueda S, Ohno S, Yamada Y, Osakabe Y, Goto H, Matsunaga Y, Ishikawa A, Usui Y, Kuroda M (2015) Novel types of small RNA exhibit sequence- and target-dependent angiogenesis suppression without activation of toll-like receptor 3 in an age-related macular degeneration (AMD) mouse model. Mol Ther Nucleic Acids 4:e258
Saint-Geniez M, Kurihara T, Sekiyama E, Maldonado AE, D’Amore PA (2009) An essential role for RPE-derived soluble VEGF in the maintenance of the choriocapillaris. Proc Natl Acad Sci USA 106:18751–18756
Olsson AK, Dimberg A, Kreuger J, Claesson-Welsh L (2006) Vegf receptor signalling—in control of vascular function. Nat Rev Mol Cell Biol 7:359–371
Acknowledgements
We thank Drs. Kinuko Sasada, Yuki Kubo and Yoshiyuki Kobayashi for their fruitful discussions. We also thank Ms. Masayo Eto for her excellent technical assistance. This work was supported in part by JSPS KAKENHI Grant numbers 26293374, 26670757, 15H04995 and 16K15734.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yoshida, S., Nakama, T., Ishikawa, K. et al. Periostin in vitreoretinal diseases. Cell. Mol. Life Sci. 74, 4329–4337 (2017). https://doi.org/10.1007/s00018-017-2651-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00018-017-2651-5
Keywords
- Vitreoretinal disease
- Genome-wide gene expression profiling
- Proliferative diabetic retinopathy
- Proliferative vitreoretinopathy
- Age-related macular degeneration
- Fibrovascular membranes
- Epiretinal membranes
- Neovascularization
- Fibrosis
- Retina
- Choroid
- Mouse model of oxygen-induced retinal neovascularization
- Mouse model of laser-induced choroidal neovascuarization
- Single-stranded RNA interference