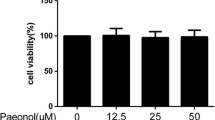
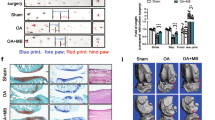
Abstract
Objective
Malvidin is one of the most widespread anthocyanidins which exhibits significant antioxidant and anti-inflammatory activity. The aim of this paper is to investigate the effects of Malvidin on osteoarthritis (OA).
Materials and methods
We created an animal model of OA using Wistar rats administered by monosodium iodoacetate (MIA). Effects of Malvidin on hyperalgesia were evaluated by paw pressure tests and compression threshold test. Articular chondrocytes were isolated from the OA rats to detect the apoptotic chondrocytes using senescence-associated β-galactosidase (SA-β-gal) staining kit. The expression levels of pro-inflammatory cytokines and matrix metalloproteinase (MMPs) were assessed by western blot and qPCR. Luciferase assay was used to determine the impact of Malvidin on nuclear factor-kappa B (NF-κB) pathway.
Results
Malvidin treatment exhibited significant pain-relieving effects in OA rats and decreased the expression level of apoptotic marker SA-β-gal in chondrocytes. We found that the upregulated expressions of interleukin (IL)-1β, IL-6, tumor necrosis factor-α (TNF-α), and MMPs induced by MIA in cartilage tissues were significantly reversed by Malvidin. Furthermore, Malvidin inhibited NF-κB pathway via an NF-κB inhibitor (IκBα)-independent manner through suppressing p65 nuclear transportation in vitro.
Conclusions
Our findings suggest that Malvidin significantly attenuates the OA-induced pain and inflammation by inhibiting NF-κB signaling pathway and suppressing pro-inflammatory cytokine expression and chondrocyte apoptosis.





Similar content being viewed by others
References
Bijlsma JW, Berenbaum F, Lafeber FP. Osteoarthritis: an update with relevance for clinical practice. Lancet. 2011;377(9783):2115–26.
Poole AR. Osteoarthritis as a whole joint disease. HSS J. 2012;8(1):4–6.
Vos T, Flaxman AD, Naghavi M, Lozano R, Michaud C, Ezzati M, et al. Years lived with disability (YLDs) for 1160 sequelae of 289 diseases and injuries 1990–2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet. 2012;380(9859):2163–96.
Wang K, Xu J, Hunter DJ, Ding C. Investigational drugs for the treatment of osteoarthritis. Expert Opin Investig Drugs. 2015;24(12):1539–56.
Hunter DJ, Felson DT. Osteoarthritis. BMJ. 2006;332(7542):639–42.
Hu H, Yang B, Li Y, Zhang S, Li Z. Blocking of the P2X7 receptor inhibits the activation of the MMP-13 and NF-kappaB pathways in the cartilage tissue of rats with osteoarthritis. Int J Mol Med. 2016;38(6):1922–32.
Bian Q, Wang YJ, Liu SF, Li YP. Osteoarthritis: genetic factors, animal models, mechanisms, and therapies. Front Biosci (Elite Ed). 2012;4:474–1000.
Loeser RF, Goldring SR, Scanzello CR, Goldring MB. Osteoarthritis: a disease of the joint as an organ. Arthritis Rheum. 2012;64(6):1697–707.
Rigoglou S, Papavassiliou AG. The NF-kappaB signalling pathway in osteoarthritis. Int J Biochem Cell Biol. 2013;45(11):2580–4.
Oeckinghaus A, Ghosh S. The NF-kappaB family of transcription factors and its regulation. Cold Spring Harb Perspect Biol. 2009;1(4):a000034.
Niederberger E, Geisslinger G. The IKK-NF-kappaB pathway: a source for novel molecular drug targets in pain therapy? FASEB J. 2008;22(10):3432–42.
Lianxu C, Hongti J, Changlong Y. NF-kappaBp65-specific siRNA inhibits expression of genes of COX-2, NOS-2 and MMP-9 in rat IL-1beta-induced and TNF-alpha-induced chondrocytes. Osteoarthr Cartil. 2006;14(4):367–76.
Tetlow LC, Adlam DJ, Woolley DE. Matrix metalloproteinase and proinflammatory cytokine production by chondrocytes of human osteoarthritic cartilage: associations with degenerative changes. Arthr Rheum. 2001;44(3):585–94.
Bognar E, Sarszegi Z, Szabo A, Debreceni B, Kalman N, Tucsek Z, et al. Antioxidant and anti-inflammatory effects in RAW264.7 macrophages of Malvidin, a major red wine polyphenol. PLoS ONE. 2013;8(6):e65355.
Leighton GE, Rodriguez RE, Hill RG, Hughes J. Kappa-opioid agonists produce antinociception after i.v. and i.c.v. but not intrathecal administration in the rat. Br J Pharmacol. 1988;93(3):553–60.
Barton NJ, Strickland IT, Bond SM, Brash HM, Bate ST, Wilson AW, et al. Pressure application measurement (PAM): a novel behavioural technique for measuring hypersensitivity in a rat model of joint pain. J Neurosci Methods. 2007;163(1):67–75.
Aqil F, Gupta A, Munagala R, Jeyabalan J, Kausar H, Sharma RJ, et al. Antioxidant and antiproliferative activities of anthocyanin/ellagitannin-enriched extracts from Syzygium cumini L. (Jamun, the Indian Blackberry). Nutr Cancer. 2012;64(3):428–38.
Lee SG, Kim B, Yang Y, Pham TX, Park YK, Manatou J, et al. Berry anthocyanins suppress the expression and secretion of proinflammatory mediators in macrophages by inhibiting nuclear translocation of NF-kappaB independent of NRF2-mediated mechanism. J Nutr Biochem. 2014;25(4):404–11.
Nizamutdinova IT, Kim YM, Chung JI, Shin SC, Jeong YK, Seo HG, et al. Anthocyanins from black soybean seed coats preferentially inhibit TNF-alpha-mediated induction of VCAM-1 over ICAM-1 through the regulation of GATAs and IRF-1. J Agric Food Chem. 2009;57(16):7324–30.
Garcia-Alonso M, Rimbach G, Rivas-Gonzalo JC, De Pascual-Teresa S. Antioxidant and cellular activities of anthocyanins and their corresponding vitisins A—studies in platelets, monocytes, and human endothelial cells. J Agric Food Chem. 2004;52(11):3378–84.
Kanterman J, Sade-Feldman M, Baniyash M. New insights into chronic inflammation-induced immunosuppression. Semin Cancer Biol. 2012;22(4):307–18.
Lee C, Han D, Kim B, Baek N, Baik BK. Antioxidant and anti-hypertensive activity of anthocyanin-rich extracts from hulless pigmented barley cultivars. Int J Food Sci Technol. 2013;48(5):984–91.
Huang WY, Wang J, Liu YM, Zheng QS, Li CY. Inhibitory effect of Malvidin on TNF-alpha-induced inflammatory response in endothelial cells. Eur J Pharmacol. 2014;723:67–72.
Kumar A, Takada Y, Boriek AM, Aggarwal BB. Nuclear factor-kappaB: its role in health and disease. J Mol Med (Berl). 2004;82(7):434–48.
Miagkov AV, Kovalenko DV, Brown CE, Didsbury JR, Cogswell JP, Stimpson SA, et al. NF-kappaB activation provides the potential link between inflammation and hyperplasia in the arthritic joint. Proc Natl Acad Sci USA. 1998;95(23):13859–64.
Gerlag DM, Ransone L, Tak PP, Han Z, Palanki M, Barbosa MS, et al. The effect of a T cell-specific NF-kappa B inhibitor on in vitro cytokine production and collagen-induced arthritis. J Immunol. 2000;165(3):1652–8.
Blanco FJ, Guitian R, Moreno J, de Toro FJ, Galdo F. Effect of antiinflammatory drugs on COX-1 and COX-2 activity in human articular chondrocytes. J Rheumatol. 1999;26(6):1366–73.
Csaki C, Mobasheri A, Shakibaei M. Synergistic chondroprotective effects of curcumin and resveratrol in human articular chondrocytes: inhibition of IL-1beta-induced NF-kappaB-mediated inflammation and apoptosis. Arthr Res Ther. 2009;11(6):R165.
Fernandes JC, Martel-Pelletier J, Pelletier JP. The role of cytokines in osteoarthritis pathophysiology. Biorheology. 2002;39(1–2):237–46.
Shakibaei M, John T, Schulze-Tanzil G, Lehmann I, Mobasheri A. Suppression of NF-kappaB activation by curcumin leads to inhibition of expression of cyclo-oxygenase-2 and matrix metalloproteinase-9 in human articular chondrocytes: implications for the treatment of osteoarthritis. Biochem Pharmacol. 2007;73(9):1434–45.
Velinov N, Poptodorov G, Gabrovski N, Gabrovski S. The role of matrix metalloproteinases in the tumor growth and metastasis. Khirurgiia (Sofiia). 2010;1:44–9.
Ozler K, Aktas E, Atay C, Yilmaz B, Arikan M, Gungor S. Serum and knee synovial fluid matrix metalloproteinase-13 and tumor necrosis factor-alpha levels in patients with late stage osteoarthritis. Acta Orthop Traumatol Turc. 2016;50(6):670–3.
Frank S, Peters MA, Wehmeyer C, Strietholt S, Koers-Wunrau C, Bertrand J, et al. Regulation of matrix metalloproteinase-3 and matrix metalloproteinase-13 by SUMO-2/3 through the transcription factor NF-kappaB. Ann Rheum Dis. 2013;72(11):1874–81.
Perkins ND. Integrating cell-signalling pathways with NF-kappaB and IKK function. Nat Rev Mol Cell Biol. 2007;8(1):49–62.
Gilmore TD. The Rel/NF-kappaB signal transduction pathway: introduction. Oncogene. 1999;18(49):6842–4.
Gilmore TD. Introduction to NF-kappaB: players, pathways, perspectives. Oncogene. 2006;25(51):6680–4.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Research involving human participants and/or animals
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Informed consent
Not applicable.
Funding
None.
Additional information
Responsible Editor: John Di Battista.
Rights and permissions
About this article
Cite this article
Dai, T., Shi, K., Chen, G. et al. Malvidin attenuates pain and inflammation in rats with osteoarthritis by suppressing NF-κB signaling pathway. Inflamm. Res. 66, 1075–1084 (2017). https://doi.org/10.1007/s00011-017-1087-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00011-017-1087-6