Abstract
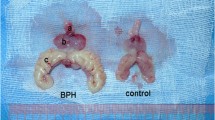
Benign prostatic hyperplasia (BPH) affects majority of the older male population. Lower urinary tract symptoms are commonly seen in patients suffering from BPH. The pathophysiology of the BPH is indefinite and ageing, hormonal imbalance, epithelial -mesenchymal interactions play role in the evolution of BPH. Pharmacologically BPH is treated with alpha 1a adrenergic antagonists acts on the dynamic component and 5-alpha reductase inhibitors act on the static component of the BPH. Different invitro, invivo, exvivo, transgenic, xenograft and spontaneous BPH screening models are developed to study the pathophysiological insights of the BPH and to develop novel drug molecules for the treatment of BPH. WPMY-1, NRP152 and NRP-154 human prostatic cell lines are used for invitro screening. Testosterone and sulpiride induces BPH in rats, hemorrhagic cystitis by cyclophosphamide, partial ligation of urethra of the bladder in rats and intraprostatic injection in mongrel dogs are used as invivo models. Isolated prostate gland, detrusor muscle and urethral contractility studies are used for exvivo screening. Transgenic BPH models are developed by transfection using probacin prolactin, murine mammary tumor virus (MMTV)-Int2 and MMTV-keratinocyte growth factor genes. Human BPH specimen are implanted in immunocompromised rats and mice are used as xenograft models. Naturally occurring BPH in old dogs is referred as spontaneous BPH model.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
Bibliography
AUA Practice Guidelines Committee. AUA guideline on management of benign prostatic hyperplasia (2003). Chapter 1: diagnosis and treatment recommendations. J Urol. 2003;170:530–47.
Bernichtein S, Pigat N, Camparo P, Latil A, Viltard M, Friedlander G, et al. Anti-inflammatory properties of Lipidosterolic extract of Serenoa repens (Permixon®) in a mouse model of prostate hyperplasia. Prostate. 2015;75:706–22.
Chuang YC, Tu CH, Huang CC, Lin HJ, Chiang PH, Yoshimura N, et al. Intraprostatic injection of botulinum toxin type-A relieves bladder outlet obstruction in human and induces prostate apoptosis in dogs. BMC Urol. 2006;6:12.
Danielpour D. Transdifferentiation of NRP-152 rat prostatic basal epithelial cells toward a luminal phenotype: regulation by glucocorticoid, insulin-like growth factor-I and transforming growth factor-beta. J Cell Sci. 1999;112:169–79.
Dawson C, Whitfield H. ABC of urology. Bladder outflow obstruction. BMJ. 1996;312:767–70.
Debiec-Rychter M, Asgari K, Jones RF, Grignon DJ, Haas GP, Wang CY. Xenografts of human benign prostatic hyperplasia tissues in the nude mouse. In Vivo. 1994;8:449–50.
Dmochowski RR. Bladder outlet obstruction: etiology and evaluation. Rev Urol. 2005;7:S3–13.
Dobrek Ł, Baranowska A, Skowron B, Thor PJ. The influence of piroxicam, a non-selective cyclooxygenase inhibitor, on autonomic nervous system activity in experimental cyclophosphamide-induced hemorrhagic cystitis and bladder outlet obstruction in rats. Acta Pol Pharm. 2014;71:497–507.
Elkahwaji J. The role of inflammatory mediators in the development of prostatic hyperplasia and prostate cancer. Res Rep Urol. 2012;5:1–10.
Hennenberg M, Stief CG, Gratzke C. Pharmacology of the lower urinary tract. Indian J Urol. 2014;30:181–8.
Izumi K, Mizokami A, Lin WJ, Lai KP, Chang C. Androgen receptor roles in the development of benign prostate hyperplasia. Am J Pathol. 2013;182:1942–9.
Jiang X, Chen Y, Zhu H, Wang B, Qu P, Chen R, et al. Sodium tanshinone IIA sulfonate ameliorates bladder fibrosis in a rat model of partial bladder outlet obstruction by inhibiting the TGF-β/smad pathway activation. PLoS One. 2015;10:1–17.
Jk P, Patel N. Epidemiology and etiology of benign prostatic hyperplasia and bladder outlet obstruction. Indian J Urol. 2014;30:170.
Kitsberg DI, Leder P. Keratinocyte growth factor induces mammary and prostatic hyperplasia and mammary adenocarcinoma in transgenic mice. Oncogene. 1996;13:2507–15.
Lesovaya EA, Kirsanov KI, Antoshina EE, Trukhanova LS, Gorkova TG, Shipaeva EV, et al. Rapatar, a nanoformulation of rapamycin, decreases chemically-induced benign prostate hyperplasia in rats. Oncotarget. 2015;6:9718–27.
Lin PH, Pan Z, Zheng L, Li N, Danielpour D, Ma JJ. Overexpression of Bax sensitizes prostate cancer cells to TGF-beta induced apoptosis. Cell Res. 2005;15:160–6.
Lopez-Barcons LA. Human benign prostatic hyperplasia heterotransplants as an experimental model. Asian J Androl. 2010;12:157–63.
Love HD, Booton SE, Boone BE, Breyer JP, Koyama T, Revelo MP, et al. Androgen regulated genes in human prostate xenografts in mice: relation to BPH and prostate cancer. PLoS One. 2009;4:1–15.
Mahapokai W, Van Sluijs FJ, Schalken JA. Models for studying benign prostatic hyperplasia. Prostate Cancer Prostatic Dis. 2000;3:28–33.
McLaren ID, Jerde TJ, Bushman W. Role of interleukins, IGF and stem cells in BPH. Differentiation. 2011;82:237–43.
Moraes JP, Pereira DS, Matos AS, Santana DG, Santos CA, Estevam CS, et al. The ethanol extract of the inner bark of Caesalpinia pyramidalis (Tul.) reduces urinary bladder damage during cyclophosphamide-induced cystitis in rats. Sci World J. 2013; https://doi.org/10.1155/2013/694010.
Muller WJ, Lee FS, Dickson C, Peters G, Pattengale P, Leder P. The int-2 gene product acts as an epithelial growth factor in transgenic mice. EMBO J. 1990;9:907–13.
Nicholson TM, Moses MA, Uchtmann KS, Keil KP, Bjorling DE, Vezina CM, et al. Estrogen receptor-α is a key mediator and therapeutic target for bladder complications of benign prostatic hyperplasia. J Urol. 2015;193:722–9.
Parsons JK, Carter HB, Partin AW, Windham BG, Metter EJ, Ferrucci L, et al. Metabolic factors associated with benign prostatic hyperplasia. J Clin Endocrinol Metab. 2006;91:2562–8.
Raja A, Hori S, Armitage JN. Hormonal manipulation of lower urinary tract symptoms secondary to benign prostatic obstruction. Indian J Urol. 2014;30:189–93.
Rick FG, Abi-Chaker A, Szalontay L, Perez R, Jaszberenyi M, Jayakumar AR, et al. Shrinkage of experimental benign prostatic hyperplasia and reduction of prostatic cell volume by a gastrin-releasing peptide antagonist. Proc Natl Acad Sci. 2013;110:2617–22.
Roehrborn CG. Male lower urinary tract symptoms (LUTS) and benign prostatic hyperplasia (BPH). Med Clin North Am. 2011;95:87–100.
Shin IS, Lee MY, Jung DY, Seo CS, Ha HK, Shin HK. Ursolic acid reduces prostate size and dihydrotestosterone level in a rat model of benign prostatic hyperplasia. Food Chem Toxicol. 2012;50:884–8.
Ventura S, Oliver V, White C, Xie J, Haynes J, Exintaris B. Novel drug targets for the pharmacotherapy of benign prostatic hyperplasia (BPH): drug targets for BPH. Br J Pharmacol. 2011;163:891–907.
Vignozzi L, Filippi S, Comeglio P, Cellai I, Morelli A, Maneschi E, et al. Tadalafil effect on metabolic syndrome-associated bladder alterations: an experimental study in a rabbit model. J Sex Med. 2014;11:1159–72.
Vyas B, Desai N, Patel P, Joshi S, Shah D. Effect of Boerhaavia diffusa in experimental prostatic hyperplasia in rats. Indian J Pharmacol. 2013;45:264.
Wang C, Du X, Yang R, Liu J, Xu D, Shi J, et al. The prevention and treatment effects of tanshinone IIA on oestrogen/androgen-induced benign prostatic hyperplasia in rats. J Steroid Biochem Mol Biol. 2015;145:28–37.
Wang X, Liao J, Yin D, Zhan F, Dai S, Xie G, et al. Establishment of a novel model for studying the effects of extracts of Chinese herb medicine on human type II 5alpha-reductase in vitro. Yakugaku Zasshi. 2010;130:1207–14.
Webber MM, Trakul N, Thraves PS, Bello-DeOcampo D, Chu WW, Storto PD, et al. A human prostatic stromal myofibroblast cell line WPMY-1: a model for stromal-epithelial interactions in prostatic neoplasia. Carcinogenesis. 1999;20:1185–92.
Zhang X, Zang N, Wei Y, Yin J, Teng R, Seftel A, et al. Testosterone regulates smooth muscle contractile pathways in the rat prostate: emphasis on PDE5 signaling. AJP Endocrinol Metab. 2012;302:243–53.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Santosh Kumar, S.C. (2022). Screening Methods for the Evaluation of Drugs for Benign Prostatic Hyperplasia. In: Lakshmanan, M., Shewade, D.G., Raj, G.M. (eds) Introduction to Basics of Pharmacology and Toxicology. Springer, Singapore. https://doi.org/10.1007/978-981-19-5343-9_45
Download citation
DOI: https://doi.org/10.1007/978-981-19-5343-9_45
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-19-5342-2
Online ISBN: 978-981-19-5343-9
eBook Packages: MedicineMedicine (R0)