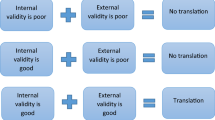
Abstract
Animal research provides a major contribution to the discovery of new compounds and its mechanism of action. It also deals with the pharmacokinetics profile and determination of safe dose of a compound which is to be tested in humans. There is a necessity to choose an appropriate animal model for preclinical research in order to carry out a clinical trial. Research can be performed on already existing validated animal model or by validating a newer model. Validation criteria of an animal model changes from one to another based on the purpose of the model (fit-for-purpose). Face validity, predictive validity and construct validity ensures the closeness of the animal model to humans. In addition to these validity, few more criteria have been added to assess and optimise the animal model, i.e. epidemiology, symptomatology, natural history, end points, genetics, and biochemical parameters, pharmacological and histological features. There is no single animal model which can satisfy all types of validity for any disease. Even though shortcomings are inevitable, these models pave way for the safer research study in humans. One can choose an animal model closer to an ideal one. Thus validation plays a crucial role in translation of animal research to humans.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
Bibliography
Andersen ML, Winter LMF. Animal models in biological and biomedical research – experimental and ethical concerns. An Acad Bras Cienc. 2017;91(Suppl 1):1–13.
Bailey J, Balls M. Recent efforts to elucidate the scientific validity of animal-based drug tests by the pharmaceutical industry, pro-testing lobby groups, and animal welfare organisations. BMC Med Ethics. 2019;20(16):1–7.
Denayer T, Stohr T, Van Roy M. Animal models in translational medicine: validation and prediction. New Horiz Transl Med. 2014;2:5–11.
Ericsson AC, Crim MJ, Franklin CL. A brief history of animal modeling. Mo Med. 2013;110(3):201–5.
Ferreira GS, Veening-Griffioen DH, Boon WPC, Moors EHM, Gispen-de Wied CC, Schellekens H, et al. A standardised framework to identify optimal animal models for efficacy assessment in drug development. PLoS One. 2019;14(6):1–17.
Ferreira GS, Veening-Griffioen DH, Boon WPC, Moors EHM, Meer PJK. Levelling the translational gap for animal to human efficacy data. Animals. 2020;10(1199):1–13.
Hau J. Animal models. In: Hau J, Hoosier GLV, editors. Handbook of laboratory animal science. 2nd ed. Boca Raton: CRC Press; 2003. p. 1–11.
Hau J. Animal models for human diseases. In: Conn PM, editor. Sourcebook of models for biomedical research. Totowa, NJ: Humana Press Inc; 2008. p. 3–8.
McGonigle P, Ruggeri B. Animal models of human disease: challenges in enabling translation. Biochem Pharmacol. 2013;87(1):1–39.
Pound P, Ebrahim S, Sandercock P, Bracken MB, Roberts I. Where is the evidence that animal research benefits humans? BMJ. 2014;328:514–7.
Robinson NB, Krieger K, Khan F, Huffman W, Chang M, Naik A, Yongle R, Hameed I, Krieger K, Girardi LN, Gaudino M. The current state of animal models in Research: a review. Int J Surg. 2019;10:1–21.
Sam-Dodd F. Strategies to optimize the validity of disease models in the drug discovery process. Drug Discov Today. 2006;11(7):355–63.
Sjoberg EA. Logical fallacies in animal model research. Sjoberg Behav Brain Funct. 2017;13(3):1–13.
Varga OE, Hansen AK, Sandoe P, Olsson IAS. Validating animal models for preclinical research: a scientific and ethical discussion. ATLA. 2010;38:245–8.
Worp HB, Howells DW, Emily Sena ES, Porritt MJ, Rewell S, Collins VO, et al. Can animal models of disease reliably inform human studies? PLoS Med. 2010;7(3):e1000245.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Subramanian, U. (2022). Validation of Animal Models. In: Lakshmanan, M., Shewade, D.G., Raj, G.M. (eds) Introduction to Basics of Pharmacology and Toxicology. Springer, Singapore. https://doi.org/10.1007/978-981-19-5343-9_12
Download citation
DOI: https://doi.org/10.1007/978-981-19-5343-9_12
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-19-5342-2
Online ISBN: 978-981-19-5343-9
eBook Packages: MedicineMedicine (R0)