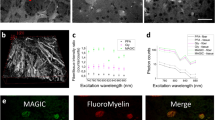
Abstract
Central myelin is an envelope composed of dozens of superimposed layers of the plasma membrane of oligodendrocytes around the neural axons. This sheath functions as an insulator that increases the efficiency and speed of the electrical impulse. Several traditional staining methods have been available to observe myelin in tissue sections for many years. However, most of what we know about the structure and organization of myelin comes from transmission electron microscopy images. Nowadays, specific molecular components of myelin can be identified and visualized by fluorescence microscopy. Even though, their lipidic nature is much more difficult to observe by this kind of microscopy technique. In the last years, superresolution and other imaging techniques were developed and applied to the study of myelin. Thanks to these and other advances in imaging that are described in this chapter, new structural details as well as many of the fine steps involved in myelination are now visually accessible.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Boullerne AI (2016) The history of myelin. Exp Neurol 283:431–445
Poitelon Y, Kopec AM, Belin S (2020) Myelin fat facts: an overview of lipids and fatty acid metabolism. Cell 9:812
Delgado AC et al (2021) Release of stem cells from quiescence reveals gliogenic domains in the adult mouse brain. Science 372:1205–1209
Mizrak D et al (2019) Single-cell analysis of regional differences in adult V-SVZ neural stem cell lineages. Cell Rep 26:394–406.e5
Bergles DE, Richardson WD (2016) Oligodendrocyte development and plasticity. Cold Spring Harb Perspect Biol 8:a020453
Nishiyama A, Shimizu T, Sherafat A, Richardson WD (2021) Life-long oligodendrocyte development and plasticity. Semin Cell Dev Biol 116:25–37
Hildebrand C, Remahl S, Persson H, Bjartmar C (1993) Myelinated nerve fibres in the CNS. Prog Neurobiol 40:319–384
Studer D, Humbel BM, Chiquet M (2008) Electron microscopy of high pressure frozen samples: bridging the gap between cellular ultrastructure and atomic resolution. Histochem Cell Biol 130:877–889
Möbius W et al (2010) Electron microscopy of the mouse central nervous system. In: Methods in cell biology, vol 96. Elsevier, Burlington, MA, pp 475–512
Möbius W, Nave K-A, Werner HB (2016) Electron microscopy of myelin: structure preservation by high-pressure freezing. Brain Res 1641:92–100
Lučić V, Leis A, Baumeister W (2008) Cryo-electron tomography of cells: connecting structure and function. Histochem Cell Biol 130:185–196
Kizilyaprak C, Daraspe J, Humbel BM (2014) Focused ion beam scanning electron microscopy in biology: focused ion beam scanning electron microscopy. J Microsc 254:109–114
Steyer AM et al (2020) Pathology of myelinated axons in the PLP-deficient mouse model of spastic paraplegia type 2 revealed by volume imaging using focused ion beam-scanning electron microscopy. J Struct Biol 210:107492
Weigert C (1885) Eine vergesserung der Haematoxylin Blutlaugen-salzmethod fur das Centranlnervensystem. Fortschr Deutsch Med 3:236–239
Clark SL, Ward JW (1934) A variation of the Pal Weigert method for staining myelin sheaths. Stain Technol 9:53–55
Weil A (1928) A rapid method for staining myelin sheaths. Arch Neurol Psychiatry 20:392
Berube GR, Powers MM, Clark G (1965) Iron hematoxylin chelates: II. Histochemistry of myelin sheath stains. Stain Technol 40:235–238
Klüver H, Barrera E (1953) A method for the combined staining of cells and fibers in the nervous system. J Neuropathol Exp Neurol 12:400–403
Hori SH (1963) A simplified acid Hematein test for phospholipids. Stain Technol 38:221–225
Meier C (1976) Some observations on early myelination in the human spinal cord. Light and electron microscope study. Brain Res 104:21–32
Meywald T, Scherthan H, Nagl W (2004) Increased specificity of colloidal silver staining by means of chemical attenuation. Hereditas 124:63–70
Jain N (1998) A histologically visible representation of the fingers and palm in primate area 3b and its immutability following long-term deafferentations. Cereb Cortex 8:227–236
Pistorio AL, Hendry SH, Wang X (2006) A modified technique for high-resolution staining of myelin. J Neurosci Methods 153:135–146
Valério-Gomes B, Guimarães DM, Szczupak D, Lent R (2018) The absolute number of oligodendrocytes in the adult mouse brain. Front Neuroanat 12:90
Nishiyama A, Lin X-H, Giese N, Heldin C-H, Stallcup WB (1996) Co-localization of NG2 proteoglycan and PDGF ?-receptor on O2A progenitor cells in the developing rat brain. J Neurosci Res 43:299–314
Scherer SS et al (1994) Differential regulation of the 2′,3′-cyclic nucleotide 3′-phosphodiesterase gene during oligodendrocyte development. Neuron 12:1363–1375
Bhat RV et al (1996) Expression of the APC tumor suppressor protein in oligodendroglia. Glia 17:169–174
Stelzer EHK et al (2021) Light sheet fluorescence microscopy. Nat Rev Methods Primers 1:73
Werner C, Sauer M, Geis C (2021) Super-resolving microscopy in neuroscience. Chem Rev 121:11971–12015
Rusch H et al (2022) Finding the best clearing approach—towards 3D wide-scale multimodal imaging of aged human brain tissue. NeuroImage 247:118832
Ryan DP et al (2017) Automatic and adaptive heterogeneous refractive index compensation for light-sheet microscopy. Nat Commun 8:612
Gao R et al (2019) Cortical column and whole-brain imaging with molecular contrast and nanoscale resolution. Science 363:eaau8302
Duan R et al (2021) Novel insight into the potential pathogenicity of mitochondrial dysfunction resulting from PLP1 duplication mutations in patients with Pelizaeus–Merzbacher disease. Neuroscience 476:60–71
Abe Y et al (2019) Correlative study using structural MRI and super-resolution microscopy to detect structural alterations induced by long-term optogenetic stimulation of striatal medium spiny neurons. Neurochem Int 125:163–174
Hainsworth AH et al (2018) Super-resolution imaging of subcortical white matter using stochastic optical reconstruction microscopy (STORM) and super-resolution optical fluctuation imaging (SOFI). Neuropathol Appl Neurobiol 44:417–426
Steshenko O et al (2016) Reorganization of lipid diffusion by myelin basic protein as revealed by STED nanoscopy. Biophys J 110:2441–2450
Micu I et al (2007) Real-time measurement of free Ca2+ changes in CNS myelin by two-photon microscopy. Nat Med 13:874–879
Fu Y, Huff TB, Wang H-W, Cheng J-X, Wang H (2008) Ex vivo and in vivo imaging of myelin fibers in mouse brain by coherent anti-Stokes Raman scattering microscopy. Opt Express 16:19396
Zhai Y-H et al (2011) Multimodal coherent anti-Stokes Raman spectroscopic imaging with a fiber optical parametric oscillator. Appl Phys Lett 98:191106
Meyer T et al (2013) Expanding multimodal microscopy by high spectral resolution coherent anti-stokes Raman scattering imaging for clinical disease diagnostics. Anal Chem 85:6703–6715
Micu I, Brideau C, Lu L, Stys PK (2017) Effects of laser polarization on responses of the fluorescent Ca2+ indicator X-Rhod-1 in neurons and myelin. Neurophoton 4:025002
Wu W, Li X, Qu JY, He S (2021) In vivo imaging of biological tissues with combined two-photon fluorescence and stimulated Raman scattering microscopy. J Vis Exp 178:63411. https://doi.org/10.3791/63411
Bélanger E et al (2012) Live animal myelin histomorphometry of the spinal cord with video-rate multimodal nonlinear microendoscopy. J Biomed Opt 17:021107
Okada SLM, Stivers NS, Stys PK, Stirling DP (2014) An ex vivo laser-induced spinal cord injury model to assess mechanisms of axonal degeneration in real-time. J Vis Exp 93:52173. https://doi.org/10.3791/52173
Stivers NS et al (2017) The toll-like receptor 2 agonist Pam3CSK4 is neuroprotective after spinal cord injury. Exp Neurol 294:1–11
Chu S-W et al (2002) Nonlinear bio-photonic crystal effects revealed with multimodal nonlinear microscopy. J Microsc 208:190–200
Huang JY, Chen Z, Lewis A (1989) Second-harmonic generation in purple membrane-poly(vinyl alcohol) films: probing the dipolar characteristics of the bacteriorhodopsin chromophore in bR570 and M412. J Phys Chem 93:3314–3320
Jin L et al (2006) Characterization and application of a new optical probe for membrane lipid domains. Biophys J 90:2563–2575
Moen EK, Ibey BL, Beier HT (2014) Detecting subtle plasma membrane perturbation in living cells using second harmonic generation imaging. Biophys J 106:L37–L40
Nuriya M et al (2016) Multimodal two-photon imaging using a second harmonic generation-specific dye. Nat Commun 7:11557
Garduno-Robles A et al (2020) MRI features in a rat model of H-ABC tubulinopathy. Front Neurosci 14:555
Rehberg M, Krombach F, Pohl U, Dietzel S (2011) Label-free 3D visualization of cellular and tissue structures in intact muscle with second and third harmonic generation microscopy. PLoS One 6:e28237
Farrar MJ, Wise FW, Fetcho JR, Schaffer CB (2011) In vivo imaging of myelin in the vertebrate central nervous system using third harmonic generation microscopy. Biophys J 100:1362–1371
Witte S et al (2011) Label-free live brain imaging and targeted patching with third-harmonic generation microscopy. Proc Natl Acad Sci 108:5970–5975
Anzalone A et al (2018) Feasibility study on mouse live imaging after spinal cord injury and poly(lactide-co-glycolide) bridge implantation. J Biomed Opt 23:1
Redlich MJ, Lim H (2019) A method to measure myeloarchitecture of the murine cerebral cortex in vivo and ex vivo by intrinsic third-harmonic generation. Front Neuroanat 13:65
Redlich MJ et al (2021) High-pulse-energy multiphoton imaging of neurons and oligodendrocytes in deep murine brain with a fiber laser. Sci Rep 11:7950
Chorghay Z et al (2021) Activity-dependent alteration of early myelin ensheathment in a developing sensory circuit. J Comp Neurol 530:25253. https://doi.org/10.1002/cne.25253
Pezacki JP et al (2011) Chemical contrast for imaging living systems: molecular vibrations drive CARS microscopy. Nat Chem Biol 7:137–145
Wang H, Fu Y, Zickmund P, Shi R, Cheng J-X (2005) Coherent anti-Stokes Raman scattering imaging of axonal myelin in live spinal tissues. Biophys J 89:581–591
Gasecka A, Daradich A, Dehez H, Piché M, Côté D (2013) Resolution and contrast enhancement in coherent anti-Stokes Raman-scattering microscopy. Opt Lett 38:4510
Lucas A, Poleg S, Klug A, McCullagh EA (2021) Myelination deficits in the auditory brainstem of a mouse model of fragile X syndrome. Front Neurosci 15:772943
Tamosaityte S et al (2016) Inflammation-related alterations of lipids after spinal cord injury revealed by Raman spectroscopy. J Biomed Opt 21:061008
Costantini I et al (2021) Autofluorescence enhancement for label-free imaging of myelinated fibers in mammalian brains. Sci Rep 11:8038
Fowler SD, Greenspan P (1985) Application of Nile red, a fluorescent hydrophobic probe, for the detection of neutral lipid deposits in tissue sections: comparison with oil red O. J Histochem Cytochem 33:833–836
Prioli S, Reinholdt P, Hornum M, Kongsted J (2019) Rational design of Nile red analogs for sensing in membranes. J Phys Chem B 123:10424–10432
Teo W et al (2021) Nile red fluorescence spectroscopy reports early physicochemical changes in myelin with high sensitivity. Proc Natl Acad Sci USA 118:e2016897118
Sakdinawat A, Attwood D (2010) Nanoscale X-ray imaging. Nat Photon 4:840–848
Dučić T et al (2011) Structure and composition of myelinated axons: a multimodal synchrotron spectro-microscopy study. J Struct Biol 173:202–212
Töpperwien M et al (2017) Three-dimensional mouse brain cytoarchitecture revealed by laboratory-based X-ray phase-contrast tomography. Sci Rep 7:42847
Carboni E et al (2017) Imaging of neuronal tissues by X-ray diffraction and X-ray fluorescence microscopy: evaluation of contrast and biomarkers for neurodegenerative diseases. Biomed Opt Express 8:4331
Jensen TH et al (2011) Brain tumor imaging using small-angle X-ray scattering tomography. Phys Med Biol 56:1717–1726
Lwin T-T, Yoneyama A, Maruyama H, Takeda T (2021) Visualization ability of phase-contrast synchrotron-based X-ray imaging using an X-ray interferometer in soft tissue tumors. Technol Cancer Res Treat 20. https://doi.org/10.1177/15330338211010121
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Piazza, V., Hernandez, V.H. (2022). Myelin Imaging. In: Mazumder, N., Gangadharan, G., Kistenev, Y.V. (eds) Advances in Brain Imaging Techniques. Springer, Singapore. https://doi.org/10.1007/978-981-19-1352-5_5
Download citation
DOI: https://doi.org/10.1007/978-981-19-1352-5_5
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-19-1351-8
Online ISBN: 978-981-19-1352-5
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)