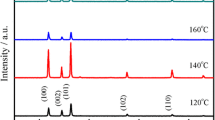
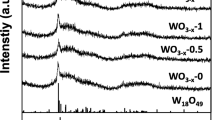
Abstract
In this work, we propose the facile controlled hydrothermal synthesis of α-MoO3 nanorods at a mild temperature. The microstructure and controlled morphology of the as-obtained product can be tailored by altering growth parameters. The average diameter of the as-obtained nanorods is about 200 nm. Electrochromic properties and capacitor performance of the as-synthesized nanorods were investigated. The results demonstrate electrochromic properties with high corresponding optical modulation range of 47% at a wavelength of 550 nm, good cyclic stability and fast response time.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Wang L, Dong ZH, Wang ZG (2013) Layered α‐Co(OH)2 nanocones as electrode materials for pseudocapacitors: understanding the effect of interlayer space on electrochemical activity. Adv Funct Mater 23:2758–2764
Xiao Y, Liu S, Li F (2012) 3D hierarchical Co3O4 twin-spheres with an urchin-like structure: Large-scale synthesis, multistep-splitting growth, and electrochemical pseudocapacitors. Adv Func Mater 22:4052–4059
Chen X, Lin H, Deng J (2014) Electrochromic fiber-shaped supercapacitors. Adv Mater 26:8126–8132
Xiao X, Ding T, Yuan L (2012) WO3-x/MoO3-x core/shell nanowires on carbon fabric as an anode for all-solid-state asymmetric supercapacitors. Adv Energy Mater 2:1328–1332
Chang J, Jin M, Yao F (2013) Asymmetric supercapacitors based on graphene/MnO2 nanospheres and graphene/MoO3 nanosheets with high energy density. Adv Funct Mater 23:5074–5083
Tang W, Liu L, Tian S (2011) Aqueous supercapacitors of high energy density based on MoO3 nanoplates as anode material. Chem Commun 47:10058–10060
Patil RS, Uplane MD, Patil PS (2008) Electrosynthesis of electrochromic molybdenum oxide thin films with rod-like features. Int J Electrochem Sci 3:259–265
Mahajan SS, Mujawar SH, Shinde PS (2019) Rapid and sensitive electrochemical monitoring of tyrosine using NiO nanoparticles modified graphite screen printed electrode. Int J Electrochem Sci 14:1556
Mahajan SS, Mujawar SH, Shinde PS (2008) Structural, optical and electrochromic properties of Nb-doped MoO3 thin films. Appl Surf Sci 254:5895–5898
Lian H, Hou Z, Shang M, Geng D, Zhang Y, Lin J (2013) Rare earth ions doped phosphors for improving efficiencies of solar cells. Energy 57:270–283
Zheng L, Xu Y, Jin D (2009) Novel metastable hexagonal MoO3 nanobelts: synthesis, photochromic, and electrochromic properties. Chem Mater 21:5681–5690
Golozar M, Chien K, Coyle TW (2012) Orthorhombic α-MoO3 coatings with lath-shaped morphology developed by SPPS: applications to super-capacitors. J Therm Spray Technol 21:469–479
Hanlon D, Backes C, Higgins TM, Hughes MO, Neill A, King P, Coleman JN (2014) Production of molybdenum trioxide nanosheets by liquid exfoliation and their application in high-performance supercapacitors. Chem Mater 26:1751–1763
Phuruangrat A, Ham DJ, Thongtem S, Lee JS (2009) Electrochemical hydrogen evolution over MoO3 nanowires produced by microwave-assisted hydrothermal reaction. Electrochem Commun 11:1740–1743
Sinaim H, Ham DJ, Lee JS, Phuruangrat A, Thongtem S, Thongtem T (2012) Free-polymer controlling morphology of α-MoO3 nanobelts by a facile hydrothermal synthesis, their electrochemistry for hydrogen evolution reactions and optical properties. J Alloy Compd 516:172–178
Taurino AM, Forleo A, Francioso L, Siciliano P, Stalder M, Nesper R (2006) Synthesis, electrical characterization, and gas sensing properties of molybdenum oxide nanorods. Appl Phys Lett 88:152111
Yu J, Ippolito SJ, Shafiei M, Wlodarski DW, Kalantar-Zadeh K (2009) Reverse biased Pt/nanostructured MoO3/SiC Schottky diode based hydrogen gas sensors. Appl Phys Lett 94:013504
Kim WS, Kim HC, Hong SH (2010) Gas sensing properties of MoO3 nanoparticles synthesized by solvothermal method. J Nanopart Res 12:1889–1896
Xing LL, Yuan S, Chen ZH, Chen YJ, Xue XY (2011) Enhanced gas sensing performance of SnO2/α-MoO3 heterostructure nanobelts. Nanotechnology 22:225502
Zhou L, Yang L, Yuan P, Zou J, Wu Y, Yu C (2010) α-MoO3 nanobelts: a high performance cathode material for lithium ion batteries. J Phys Chem C 114:21868–21872
Brezesinski T, Wang J, Tolbert SH, Dunn B (2010) Ordered mesoporous α-MoO3 with iso-oriented nanocrystalline walls for thin-film pseudocapacitors. Nat Mater 9:146–151
Sen UK, Mitra S (2012) Electrochemical activity of α-MoO3 nano-belts as lithium-ion battery cathode. RSC Adv 2:11123–11131
Meduri P, Clark E, Kim JH, Dayalan E, Sumanasekera GU, Sunkara MK (2012) MoO3–x nanowire arrays as stable and high-capacity anodes for lithium ion batteries. Nano Lett 12:1784–1788
Mohan VM, Bin H, Chen W (2010) Enhancement of electrochemical properties of MoO3 nanobelts electrode using PEG as surfactant for lithium battery. J Solid State Electrochem 14:1769–1775
Zheng L, Xu Y, Jin D, Xie Y (2009) Novel metastable hexagonal MoO3 nanobelts: synthesis, photochromic, and electrochromic properties. Chem Mater 21:5681–5690
Cai L, Rao PM, Zheng X (2011) Morphology-controlled flame synthesis of single, branched, and flower-like α-MoO3 nanobelt arrays. Nano Lett 11:872–877
Shakir I, Shahid M, Nadeem M, Kang DJ (2012) Tin oxide coating on molybdenum oxide nanowires for high performance supercapacitor devices. Electrochim Acta 72:134–137
Aravinda LS, Nagaraja KK, Bhat KU, Bhat BR (2013) Magnetron sputtered MoO3/carbon nanotube composite electrodes for electrochemical supercapacitor. J Electroanal Chem 699:28–32
Bai S, Chen S, Chen L, Zhang K, Luo R, Li D, Liu CC (2012) Ultrasonic synthesis of MoO3 nanorods and their gas sensing properties. Sens Actuators B: Chem 174:51–58
Shen Y, Xiao Y, Yan P, Yang Y, Hu F, Li Z (2014) Hydrothermal deposition and the photochromic properties of molybdenum oxide hydrate (MoO3⋅(H2O)0.69) films induced by D, L-malic acid. J Alloys Comp 588:676–680
Chen X, Lei W, Liu D, Hao J, Cui Q, Zou G (2009) Synthesis and characterization of hexagonal and truncated hexagonal shaped MoO3 nanoplates. J Phys Chem C 113:21582–21585
Ivanova T, Gesheva K, Hamelmann F (2004) Optical and electrochromic properties of CVD mixed MoO3-WO3 thin films. Vacuum 76:195–198
Lee SH, Seong MJ, Tracy CE, Mascarenhas A, Pitts JR, Deb SK (2002) Raman spectroscopic studies of electrochromic a-MoO3 thin films. Solid State Ionics 147:129–133
Qi K, Yang J, Fu J, Wang G, Zhu L, Liu G, Zheng W (2013) Morphology-controllable ZnO rings: ionic liquid-assisted hydrothermal synthesis, growth mechanism and photoluminescence properties. Cryst Eng Comm 15:6729–6735
Jia B, Gao L (2008) Growth of well-defined cubic hematite single crystals: oriented aggregation and ostwald ripening. Cryst Growth Des 8:1372–1376
Du N, Zhang H, Chen B (2007) Ligand-free self-assembly of ceria nanocrystals into nanorods by oriented attachment at low temperature. J Phys Chem C 111:12677–12680
Cao SW, Zhu YJ (2008) Surfactant-free preparation and drug release property of magnetic hollow core/shell hierarchical nanostructures. J Phys Chem C 112:12149–12156
Acknowledgements
This research is supported by Foundations of Heilongjiang Educational Committee (No. 18XN069 and No. 18XN089). We thank the Young scientists foundation from Harbin University of Commerce, China (No. 17XN018, 2019CX28) and the Young scientific research item of Harbin University of Commerce, Heilongjiang province, China (No. 2019DS084) and the National Natural Science Foundation of China (No. 52002099).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 The Author(s), under exclusive license to Springer Nature Singapore Pte Ltd.
About this paper
Cite this paper
Wang, J., Liu, Z., Cao, W. (2021). Electrochromic Properties of α-MoO3 Nanorods Fabricated by Hydrothermal Synthesis. In: Zhao, P., Ye, Z., Xu, M., Yang, L., Zhang, L., Zhu, R. (eds) Advances in Graphic Communication, Printing and Packaging Technology and Materials. Lecture Notes in Electrical Engineering, vol 754. Springer, Singapore. https://doi.org/10.1007/978-981-16-0503-1_113
Download citation
DOI: https://doi.org/10.1007/978-981-16-0503-1_113
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-16-0502-4
Online ISBN: 978-981-16-0503-1
eBook Packages: EngineeringEngineering (R0)