Abstract
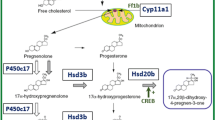
Steroid hormones are associated with the regulation of various processes in fish, like embryonic development, sex differentiation, metabolism, immune responses, circadian rhythms, stress response, and reproduction. Steroids in fish are generally classified into C21, C19, and C18 steroids based on their structure. These steroids like estrogens and androgens are used in fish farming to increase fish production based on sexual dimorphism. Progesterone (P4), 17,20β-dihydroxy-4-pregnen-3-one (17,20βP or MIH or DHP), 17,20β,21-trihydroxy-4-pregnen-3-one (20βS) and 11-deoxycortisol (S) are some of the major C21 steroids that are essential for gonadal maturation and production of other endogenous steroids. The C19 steroids, i.e. testosterone (T), 17α-Methyltestosterone (MT), and 11-Ketotestosterone (11-KT) classified as androgens help in fish spermatogenesis and C18 steroids, called as Estranes, are known as female hormones. Except for the role of steroids in fish reproduction they have a major role in immunity, puberty, and stress. Corticosteroids, a major C21 steroid, are associated with stress response in fish. Steroids like, 17β-estradiol (E2), 11KT, medroxyprogesterone, 17α,20β-dihydroxy-4-pregnen-3-one (DHP), are associated with fish adaptive and innate immunity response. Similarly, 11KT is a major steroid for fish puberty. At present, further insights are required in the field of synthetic steroids in fish and their impacts over various roles in fish physiology and future economic importance.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Borg, B. (1994). Androgens in teleost fishes. Comparative Biochemistry and Physiology, 109C, 219–245.
Busby, E. R., Roch, G. J., & Sherwood, N. M. (2010). Endocrinology of zebrafish: A small fish with a large gene pool. Fish Physiology, 29, 173–247.
Cavaco, J. E. B., Vilrokx, C., Trudeau, V. L., Schulz, R. W., & Goos, H. J. (1998). Sex steroids and the initiation of puberty in male African catfish (Clarias gariepinus). American Journal of Physiology-Regulatory, Integrative and Comparative Physiology, 275(6), R1793–R1802.
Chaves-Pozo, E., García-Ayala, A., & Cabas, I. (2018). Effects of sex steroids on fish leukocytes. Biology (Basel), 7: 9.
Guzmán, J. M., Luckenbach, J. A., Yamamoto, Y., & Swanson, P. (2014). Expression profiles of Fsh-regulated ovarian genes during oogenesis in coho salmon. PLoS One, 9(12), e114176.
Hammes, S. R., & Levin, E. R. (2007). Extranuclear steroid receptors: Nature and actions. Endocrine Reviews, 28(7), 726–741.
Kime, D. E. (1993). Classical’ and ‘non-classical’ reproductive steroids in fish. Reviews in Fish Biology and Fisheries, 3, 160–180.
Mananós, E., Duncan, N., & Mylonas, C. C. (2008). Reproduction and control of ovulation spermiation and spawning in cultured fish. In E. Cabrita, V. Robles, & P. Herráez (Eds.), Methods in reproductive aquaculture: Marine and freshwater species (pp. 3–80). Boca Raton: CRC Press.
Miura, T., Miura, C., Konda, Y., & Yamauchi, K. (2002). Spermatogenesis-preventing substance in Japanese eel. Development, 129, 2689–2697.
Miura, T., Miura, C., Yamauchi, K., & Nagahama, Y. (1995). Human recombinant activin induces proliferation of spermatogonia in vitro in the Japanese eel (Anguilla japonica). Fisheries Science, 61, 434–437.
Nagahama, Y., & Yamashita, M. (2008). Regulation of oocyte maturation in fish. Development, Growth & Differentiation., 50(Suppl. 1), S195–S219. https://doi.org/10.1111/j.1440-169X.2008.01019.x.
Ogino, Y., Miyagawa, S., & Iguchi, T. (2016). Subchapter 94C—17,20β,21-Trihydroxy-4-pregnen-3-one. In Y. Takei, H. Ando, & K. Tsutsui (Eds.), (2015), Handbook of hormones: Comparative endocrinology for basic and clinical research (pp. 511–512). Academic Press.
Okuzawa, K. (2002). Puberty in teleosts. Fish Physiology and Biochemistry, 26, 31–41.
Rather, M. A., Bhat, I. A., Sharma, N., Sharma, R., Chaudhari, A., & Sundaray, J. K. (2016). Molecular characterization, tissue distribution of Follicle-Stimulating Hormone (FSH) beta subunit and effect of kisspeptin-10 on reproductive hormonal profile of Catla catla (Hamilton, 1822). Aquaculture Research, 47(7), 2089–2100.
Ruiz-Jarabo, I., Barragán-Méndez, C., Jerez-Cepa, I., Fernández-Castro, M., Sobrino, I., Mancera, J. M., et al. (2019). Plasma 1α-hydroxycorticosterone as biomarker for acute stress in catsharks (Scyliorhinus canicula). Frontiers in Physiology, 10, 1217.
Schulz, R. W., de França, L. R., Lareyre, J. J., LeGac, F., Garcia, H. C., Nobrega, R. H., et al. (2010). Spermatogenesis in fish. General and Comparative Endocrinology, 165(3), 390–411.
Sundararaj, B. I., Anand, T. C., & Donaldson, E. M. (1972). Effects of partially purified salmon pituitary gonadotropin on ovarian maintenenace, ovulation, and vitellogenesis in the hypophysectomized catfish, Heteropneustes fossilis (Bloch). General and Comparative Endocrinology, 18, 102–114.
Taranger, G. L., Carrillo, M., Rüdiger, W., Fontaine, S. P., Zanuy, S., Felip, A., et al. (2010). Control of puberty in farmed fish. General and Comparative Endocrinology, 165(3), 483–515.
Tokarz, J., Möller, G., de Angelis, M. H., & Adamski, J. (2015). Steroids in teleost fishes: A functional point of view. Steroids, 103, 123–144.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Tripathy, P.S., Parhi, J., Mandal, S.C. (2021). Steroids and Its Receptors in Fish Reproduction. In: Sundaray, J.K., Rather, M.A., Kumar, S., Agarwal, D. (eds) Recent updates in molecular Endocrinology and Reproductive Physiology of Fish. Springer, Singapore. https://doi.org/10.1007/978-981-15-8369-8_4
Download citation
DOI: https://doi.org/10.1007/978-981-15-8369-8_4
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-15-8368-1
Online ISBN: 978-981-15-8369-8
eBook Packages: Earth and Environmental ScienceEarth and Environmental Science (R0)