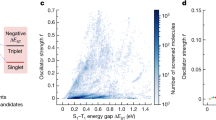
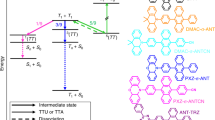
Abstract
We describe the relationship between the rate constant of internal conversion and vibronic coupling constant (VCC) based on the crude-adiabatic approximation. Vibronic coupling density (VCD) is introduced to clarify the origin of vibronic couplings from the local picture. The control of vibronic couplings from pseudo-degenerate excited electronic states gives the suppression of internal conversions. We discuss the fluorescence via higher triplets (FvHT) mechanism observed in the organic light-emitting diodes (OLEDs) of 1,4-bis(10-phenylanthracene-9-yl)benzene (BD1) used as a fluorescent dopant and the aggregation-induced enhanced emission (AIEE) of 1,2-bis(pyridylphenyl)ethene (CNPPE).
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Sato T, Tokunaga K, Tanaka K (2008) Vibronic coupling in naphthalene anion: vibronic coupling density analysis for totally symmetric vibrational modes. J Phys Chem A 112:758–767
Sato T, Uejima M, Iwahara N, Haruta N, Shizu K, Tanaka K (2013) Vibronic coupling density and related concepts. J Phys Conf Ser 428:012010
Kasha M (1950) Characterization of electronic transitions in complex molecules. Disc Faraday Soc 9:14–19
Sato T, Hayashi R, Haruta N, Pu YJ (2017) Fluorescence via reverse intersystem crossing from higher triplet states in a bisanthracene derivative. Sci Rep 7:4820
Sato T (2015) Fluorescence via reverse intersystem crossing from higher triplet states. J Comput Chem Jpn 14:189–192
Pu YJ, Satake R, Koyama Y, Otomo T, Hayashi R, Haruta N, Katagiri H, Otsuki D, Kim D, Sato T (2019) Absence of delayed fluorescence and triplet–triplet annihilation in organic light emitting diodes with spatially orthogonal bianthracenes. J Mater Chem C 7:2541–2547
Lin SH (1966) Rate of interconversion of electronic and vibrational energy. J Chem Phys 44:3759–3767
Azumi T, Matsuzaki K (1977) What does the term “vibronic coupling” mean? Photochem Photobiol 25:315–326
Uejima M, Sato T, Yokoyama D, Tanaka K, Park JW (2014) Quantum yield in blue-emitting anthracene derivatives: vibronic coupling density and transition dipole moment density. Phys Chem Chem Phys 16:14244–14256
Niu Y, Peng Q, Shuai Z (2008) Promoting-mode free formalism for excited state radiationless decay process with Duschinsky rotation effect. Sci China, Ser B: Chem 51:1153–1158
Niu Y, Peng Q, Deng C, Gao X, Shuai Z (2010) Theory of excited state decays and optical spectra: application to polyatomic molecules. J Phys Chem A 114:7817–7831
Hutchisson E (1930) Band Spectra intensities for symmetrical diatomic molecules. Phys Rev 36:410
Uejima M, Sato T, Tanaka K, Kaji H (2014) Enhancement of fluorescence in anthracene by chlorination: vibronic coupling and transition dipole moment density analysis. Chem Phys 430:47–55
Uejima M, Sato T, Detani M, Wakamiya A, Suzuki F, Suzuki H, Fukushima T, Tanaka K, Murata Y, Adachi C, Kaji H (2014) A designed fluorescent anthracene derivative: theory, calculation, synthesis, and characterization. Chem Phys Lett 602:80–83
Kameoka Y, Uebe M, Ito A, Sato T, Tanaka K (2014) Fluorescent triphenylamine derivative: theoretical design based on reduced vibronic coupling. Chem Phys Lett 615:44–49
Uebe M, Ito A, Kameoka Y, Sato T, Tanaka K (2015) Fluorescence enhancement of nonfluorescent triphenylamine: a recipe to utilize carborane cluster substituents. Chem Phys Lett 633:190–194
Sato T, Shizu K, Kuga T, Tanaka K, Kaji H (2008) Electron–vibration interactions in carriertransport material: vibronic coupling density analysis in TPD. Chem Phys Lett 458:152–156
Shizu K, Sato T, Tanaka K, Kaji H (2010) Electron–vibration interactions in triphenylamine cation: why are triphenylamine-based molecules good hole-transport materials? Chem Phys Lett 486:130–136
Shizu K, Sato T, Tanaka K, Kaji H (2010) A boron-containing molecule as an efficient electrontransporting material with low-power consumption. Appl Phys Lett 97:142111
Shizu K, Sato T, Ito A, Tanaka K, Kaji H (2011) Theoretical design of a hole-transporting molecule: hexaaza [1 6] parabiphenylophane. J Mater Chem 21:6375–6382
Sato T, Iwahara N, Haruta N, Tanaka K (2012) C60 bearing ethylene moieties. Chem Phys Lett 531:257–260
Haruta N, Sato T, Iwahara N, Tanaka K (2013) Vibronic couplings in cycloadditions to fullerenes. J Phys Conf Ser 428:012003
Haruta N, Sato T, Tanaka K (2014) Regioselectivity in multiple cycloadditions to fullerene c60: vibronic coupling density analysis. Tetrahedron 70:3510–3513
Kojima Y, Ota W, Teramura K, Hosokawa S, Tanaka T, Sato T (2019) Model building of metal oxide surfaces and vibronic coupling density as a reactivity index: regioselectivity of CO2 adsorption on Ag-loaded Ga2O3. Chem Phys Lett 715:239–243
Endo A, Ogasawara M, Takahashi A, Yokoyama D, Kato Y, Adachi C (2009) Thermally activated delayed fluorescence from Sn4+–porphyrin complexes and their application to organic light emitting diodes-a novel mechanism for electroluminescence. Adv Mater 21:4802–4806
Adachi C (2014) Third-generation organic electroluminescence materials. Jpn J Appl Phys 53:060101
Hu JY, Pu YJ, Satoh F, Kawata S, Katagiri H, Sasabe H, Kido J (2014) Bisanthracene-based donor–acceptor-type light-emitting dopants: Highly efficient deep-blue emission in organic light-emitting devices. Adv Funct Mater 24:2064–2071
Hong Y, Lam JWY, Tang BZ (2009) Aggregation-induced emission: phenomenon, mechanism and applications. Chem Commun 29:4332–4353
Hong Y, Lam JWY, Tang BZ (2011) Aggregation-induced emission. Chem Soc Rev 40:5361–5388
Nishio S, Higashiguchi K, Matsuda K (2014) The effect of cyano substitution on the fluorescence behavior of 1,2-bis(pyridylphenyl)ethene. Asian J Org Chem 3:686–690
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Petersson GA, Nakatsuji H, Li X, Caricato M, Marenich AV, Bloino J, Janesko BG, Gomperts R, Mennucci B, Hratchian HP, Ortiz JV, Izmaylov AF, Sonnenberg JL, Williams-Young D, Ding F, Lipparini F, Egidi F, Goings J, Peng B, Petrone A, Henderson T, Ranasinghe D, Zakrzewski VG, Gao J, Rega N, Zheng G, Liang W, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Throssell K, Montgomery Jr JA, Peralta JE, Ogliaro F, Bearpark MJ, Heyd JJ, Brothers EN, Kudin KN, Staroverov VN, Keith T, Kobayashi R, Normand J, Raghavachari K, Rendell A, Burant JC, Iyengar SS, Tomasi J, Cossi M, Millam JM, Klene M, Adamo C, Cammi R, Ochterski JW, Martin RL, Morokuma K, Farkas O, Foresman JB, Fox DJ (2016) Gaussian 16, Revision B. 01. Gaussian Inc., Wallingford, CT
Shizu K, Sato T, Tanaka K (2013) Inverse relationship of reorganization energy to the number of π electrons from perspective of vibronic coupling density. J Comput Chem Jpn 12:215–221
Ota W, Takahashi K, Higashiguchi K, Matsuda K, Sato T (2020) Origin of aggregation-induced enhanced emission: role of pseudo-degenerate electronic states of excimers formed in aggregation phases. J Mater Chem C. https://doi.org/10.1039/C9TC07067B
Acknowledgements
This study was supported by JSPS KAKENHI Grant Number JP17H05259 in Scientific Research on Innovative Areas “Photosynergetics”, JSPS KAKENHI Grant Number JP18K05261 in Scientific Research (C), and Element Strategy Initiative of MEXT Grant Number JPMXP0112101003. The computations were partly performed at Supercomputer System, Institute for Chemical Research, Kyoto University, Academic Center for Computing and Media Studies (ACCMS), Kyoto University, and Research Center for Computational Science, Okazaki.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Ota, W., Sato, T. (2020). Suppression of Internal Conversions from Pseudo-Degenerate Excited Electronic States. In: Miyasaka, H., Matsuda, K., Abe, J., Kawai, T. (eds) Photosynergetic Responses in Molecules and Molecular Aggregates. Springer, Singapore. https://doi.org/10.1007/978-981-15-5451-3_5
Download citation
DOI: https://doi.org/10.1007/978-981-15-5451-3_5
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-15-5450-6
Online ISBN: 978-981-15-5451-3
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)