Abstract
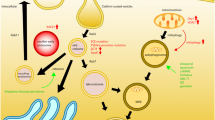
The accumulation of abnormal protein aggregates contributes to the pathological progression of diverse neurodegenerative diseases. An increasing body of evidence indicates that defects in the protein clearance system play a crucial role in this process. Cargoes delivered via endosomes, phagosomes, and autophagosomes converge on lysosomes for degradation, which process is collectively called “the endosome-autophagosome-lysosome pathway” or EALP. As such, dysfunction of lysosomes may result in the accumulation of all these upstream vesicles/cargoes and may play a key role in diverse neurodegenerative conditions.
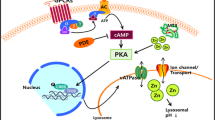
Over the years, we found that Zn-metallothionein-3 (MT3), the brain-enriched form of metallothionein, regulates lysosomal functions in cortical astrocytes. Zn-MT3 appears to interact with β-actin and activate c-Abl kinase. As a result, Zn-MT3 plays a role in maintaining lysosomal acidity, a prerequisite for vesicle fusion as well as cargo degradation. The reported downregulation of MT3 in Alzheimer’s disease (AD), hence, may contribute to lysosomal dysfunction in AD. Of interest, raising intracellular free zinc levels also caused lysosomal acidification and normalization of degradation, even in the context of arrested autophagy. Pending further research on the mechanisms of these effects, we propose that measures increasing Zn-MT3 and/or intracellular/lysosomal free zinc may be useful in normalizing lysosomal functions in neurodegenerative conditions.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Alsaqati M, Thomas RS, Kidd EJ (2018) Proteins involved in endocytosis are upregulated by ageing in the Normal human brain: implications for the development of Alzheimer’s disease. J Gerontol A Biol Sci Med Sci 73:289–298
Aras MA, Hara H, Hartnett KA, Kandler K, Aizenman E (2009) Protein kinase C regulation of neuronal zinc signaling mediates survival during preconditioning. J Neurochem 110:106–117
Baker M, Mackenzie IR, Pickering-Brown SM, Gass J, Rademakers R, Lindholm C, Snowden J, Adamson J, Sadovnick AD, Rollinson S, Cannon A, Dwosh E, Neary D, Melquist S, Richardson A, Dickson D, Berger Z, Eriksen J, Robinson T, Zehr C, Dickey CA, Crook R, McGowan E, Mann D, Boeve B, Feldman H, Hutton M (2006) Mutations in progranulin cause tau-negative frontotemporal dementia linked to chromosome 17. Nature 442:916–919
Boland B, Kumar A, Lee S, Platt FM, Wegiel J, Yu WH, Nixon RA (2008) Autophagy induction and autophagosome clearance in neurons: relationship to autophagic pathology in Alzheimer’s disease. J Neurosci 28:6926–6937
Bourdenx M, Daniel J, Genin E, Soria FN, Blanchard-Desce M, Bezard E, Dehay B (2016) Nanoparticles restore lysosomal acidification defects: implications for Parkinson and other lysosomal-related diseases. Autophagy 12:472–483
Boutajangout A, Authelet M, Blanchard V, Touchet N, Tremp G, Pradier L, Brion JP (2004) Characterisation of cytoskeletal abnormalities in mice transgenic for wild-type human tau and familial Alzheimer’s disease mutants of APP and presenilin-1. Neurobiol Dis 15:47–60
Chen S, Zhang X, Song L, Le W (2012) Autophagy dysregulation in amyotrophic lateral sclerosis. Brain Pathol 22:110–116
Cipolat Mis MS, Brajkovic S, Frattini E, Di Fonzo A, Corti S (2016) Autophagy in motor neuron disease: key pathogenetic mechanisms and therapeutic targets. Mol Cell Neurosci 72:84–90
Coffey EE, Beckel JM, Laties AM, Mitchell CH (2014) Lysosomal alkalization and dysfunction in human fibroblasts with the Alzheimer’s disease-linked presenilin 1 A246E mutation can be reversed with cAMP. Neuroscience 263:111–124
Croce KR, Yamamoto A (2019) A role for autophagy in Huntington’s disease. Neurobiol Dis 122:16–22
Cruts M, Gijselinck I, van der Zee J, Engelborghs S, Wils H, Pirici D, Rademakers R, Vandenberghe R, Dermaut B, Martin JJ, can Duijn C, Peeters K, Sciot R, Santens P, De Pooter T, Mattheijssens M, Van den Broeck M, Cuijt I, Vennekens K, De Deyn PP, Kumar-Singh S, Van Broeckhoven C (2006) Null mutations in progranulin cause ubiquitin-positive frontotemporal dementia linked to chromosome 17q21. Nature 442:920–924
De Craene JO, Bertazzi DL, Bar S, Friant S (2017) Phosphoinositides, major actors in membrane trafficking and lipid signaling pathways. Int J Mol Sci 18:634
De Duve C, Wattiaux R (1996) Functions of lysosomes. Annu Rev Physiol 28:435–492
Dominguez-Prieto M, Velasco A, Tabernero A, Medina JM (2018) Endocytosis and Transcytosis of amyloid-beta peptides by astrocytes: a possible mechanism for amyloid-beta clearance in Alzheimer’s disease. J Alzheimers Dis 65:1109–1124
Evers BM, Rodriguez-Navas C, Tesla RJ, Prange-Kiel J, Wasser CR, Yoo KS, McDonald J, Cenik B, Ravenscroft TA, Plattner F, Rademakers R, Yu G, White CL 3rd, Herz J (2017) Lipidomic and Transcriptomic basis of Lysosomal dysfunction in Progranulin deficiency. Cell Rep 20:2565–2574
Guo X, Tang P, Chen L, Liu P, Hou C, Zhang X, Liu Y, Chong L, Li X, Li R (2017) Amyloid beta-induced redistribution of transcriptional factor EB and Lysosomal dysfunction in primary microglial cells. Front Aging Neurosci 9:228
Hamano T, Gendron TF, Causevic E, Yen SH, Lin WL, Isidoro C, Deture M, Ko LW (2008) Autophagic-lysosomal perturbation enhances tau aggregation in transfectants with induced wild-type tau expression. Eur J Neurosci 27:1119–1130
Han H, Wei W, Duan W, Guo Y, Li Y, Wang J, Bi Y, Li C (2015) Autophagy-linked FYVE protein (Alfy) promotes autophagic removal of misfolded proteins involved in amyotrophic lateral sclerosis (ALS). In Vitro Cell Dev Biol Anim 51:249–263
He K, Aizenman E (2010) ERK signaling leads to mitochondrial dysfunction in extracellular zinc-induced neurotoxicity. J Neurochem 114:452–461
Heming TA, Bidani A (1995) Effects of myristate phorbol ester on V-ATPase activity and Na(+)-H+ exchange in alveolar macrophages. J Leukoc Biol 57:600–608
Huse JT, Pijak DS, Leslie GJ, Lee VM, Doms RW (2000) Maturation and endosomal targeting of beta-site amyloid precursor protein-cleaving enzyme. Alzheimer Dis Beta-secretase J Biol Chem 275:33729–33737
Jaeger PA, Pickford F, Sun CH, Lucin KM, Masliah E, Wyss-Coray T (2010) Regulation of amyloid precursor protein processing by the Beclin 1 complex. PLoS One 5:e11102
Jian JL, Hettinghouse A, Liu CJ (2017) Progranulin acts as a shared chaperone and regulates multiple lysosomal enzymes. Genes & Diseases 4:125–126
Jin S (2006) Autophagy, mitochondrial quality control, and oncogenesis. Autophagy 2:80–84
Kambe T, Tsuji T, Hashimoto A, Itsumura N (2015) The physiological, biochemical, and molecular roles of zinc transporters in zinc homeostasis and metabolism. Physiol Rev 95:749–784
Kiedrowski L (2012) Cytosolic acidification and intracellular zinc release in hippocampal neurons. J Neurochem 121:438–450
Komatsu M, Waguri S, Chiba T, Murata S, Iwata J, Tanida I, Ueno T, Koike M, Uchiyama Y, Kominami E, Tanaka K (2006) Loss of autophagy in the central nervous system causes neurodegeneration in mice. Nature 441:880–884
Konagaya M (2015) SMON: toxicity of clioquinol and the status quo. Brain Nerve 67:49–62
Kumar DK, Choi SH, Washicosky KJ, Eimer WA, Tucker S, Ghofrani J, Lefkowitz A, McColl G, Goldstein LE, Tanzi RE, Moir RD (2016) Amyloid-beta peptide protects against microbial infection in mouse and worm models of Alzheimer’s disease. Sci Transl Med 8:340ra72
Lee JH, Yu WH, Kumar A, Lee S, Mohan PS, Peterhoff CM, Wolfe DM, Martinez-Vicente M, Massey AC, Sovak G, Uchiyama Y, Westaway D, Cuervo AM, Nixon RA (2010a) Lysosomal proteolysis and autophagy require presenilin 1 and are disrupted by Alzheimer-related PS1 mutations. Cell 141:1146–1158
Lee SJ, Park MH, Kim HJ, Koh JY (2010b) Metallothionein-3 regulates lysosomal function in cultured astrocytes under both normal and oxidative conditions. Glia 58:1186–1196
Lee S, Sato Y, Nixon RA (2011) Lysosomal proteolysis inhibition selectively disrupts axonal transport of degradative organelles and causes an Alzheimer’s-like axonal dystrophy. J Neurosci 31:7817–7830
Lee SJ, Seo BR, Koh JY (2015) Metallothionein-3 modulates the amyloid beta endocytosis of astrocytes through its effects on actin polymerization. Mol Brain 8:84
Lee S, Rivera OC, Kelleher SL (2017) Zinc transporter 2 interacts with vacuolar ATPase and is required for polarization, vesicle acidification, and secretion in mammary epithelial cells. J Biol Chem 292:21598–21613
Leibiger C, Deisel J, Aufschnaiter A, Ambros S, Tereshchenko M, Verheijen BM, Büttner S, Braun RJ (2018) Endolysosomal pathway activity protects cells from neurotoxic TDP-43. Microb Cell 5:212–214
Lin CL, Tseng HC, Chen RF, Chen WP, Su MJ, Fang KM, Wu ML (2011) Intracellular zinc release-activated ERK-dependent GSK-3beta-p53 and Noxa-Mcl-1 signaling are both involved in cardiac ischemic-reperfusion injury. Cell Death Differ 18:1651–1663
Liuzzi JP, Guo L, Yoo C, Stewart TS (2014) Zinc and autophagy. Biometals 27:1087–1096
Manucat-Tan NB, Saadipour K, Wang YJ, Bobrovskaya L, Zhou XF (2019) Cellular trafficking of amyloid precursor protein in Amyloidogenesis physiological and pathological significance. Mol Neurobiol 56(2):812–830
Moors TE, Hoozemans JJ, Ingrassia A, Beccari T, Parnetti L, Chartier-Harlin MC, van de Berg WD (2017) Therapeutic potential of autophagy-enhancing agents in Parkinson’s disease. Mol Neurodegener 12:11
Muller UC, Deller T, Korte M (2017) Not just amyloid: physiological functions of the amyloid precursor protein family. Nat Rev Neurosci 18:281–298
Nascimbeni AC, Codogno P, Morel E (2017) Phosphatidylinositol-3-phosphate in the regulation of autophagy membrane dynamics. FEBS J 284:1267–1278
Nixon RA (2013) The role of autophagy in neurodegenerative disease. Nat Med 19:983–997
Noh KM, Kim YH, Koh JY (1999) Mediation by membrane protein kinase C of zinc-induced oxidative neuronal injury in mouse cortical cultures. J Neurochem 72:1609–1616
Park MH, Lee SJ, Byun HR, Kim Y, Oh YJ, Koh JY, Hwang JJ (2011) Clioquinol induces autophagy in cultured astrocytes and neurons by acting as a zinc ionophore. Neurobiol Dis 42:242–251
Perrett RM, Alexopoulou Z, Tofaris GK (2015) The endosomal pathway in Parkinson’s disease. Mol Cell Neurosci 66:21–28
Pickford F, Masliah E, Britschgi M, Lucin K, Narasimhan R, Jaeger PA, Small S, Spencer B, Rockenstein E, Levine B, Wyss-Coray T (2008) The autophagy-related protein beclin 1 shows reduced expression in early Alzheimer disease and regulates amyloid beta accumulation in mice. J Clin Invest 118:2190–2199
Polito VA, Li H, Martini-Stoica H, Wang B, Yang L, Xu Y, Swartzlander DB, Palmieri M, di Ronza A, Lee VM, Sardiello M, Ballabio A, Zheng H (2014) Selective clearance of aberrant tau proteins and rescue of neurotoxicity by transcription factor EB. EMBO Mol Med 6:1142–1160
Rahman MA, Rhim H (2017) Therapeutic implication of autophagy in neurodegenerative diseases. BMB Rep 50:345–354
Roberson ED, Scearce-Levie K, Palop JJ, Yan F, Cheng IH, Wu T, Gerstein H, Yu GQ, Mucke L (2007) Reducing endogenous tau ameliorates amyloid beta-induced deficits in an Alzheimer’s disease mouse model. Science 316:750–754
Rusmini P, Cortese K, Crippa V, Cristofani R, Cicardi ME, Ferrari V, Vezzoli G, Tedesco B, Meroni M, Messi E, Piccolella M, Galbiati M, Garrè M, Morelli E, Vaccari T, Poletti A (2019) Trehalose induces autophagy via lysosomal-mediated TFEB activation in models of motoneuron degeneration. Autophagy 15(4):631–651
Samura E, Shoji M, Kawarabayashi T, Sasaki A, Matsubara E, Murakami T, Wuhua X, Tamura S, Ikeda M, Ishiguro K, Saido TC, Westaway D, St George Hyslop P, Harigaya Y, Abe K (2006) Enhanced accumulation of tau in doubly transgenic mice expressing mutant betaAPP and presenilin-1. Brain Res 1094:192–199
Sanchez-Varo R, Trujillo-Estrada L, Sanchez-Mejias E, Torres M, Baglietto-Vargas D, Moreno-Gonzalez I, De Castro V, Jimenez S, Ruano D, Vizuete M, Davila JC, Garcia-Verdugo JM, Jimenez AJ, Vitorica J, Gutierrez A (2012) Abnormal accumulation of autophagic vesicles correlates with axonal and synaptic pathology in young Alzheimer’s mice hippocampus. Acta Neuropathol 123:53–70
Sardiello M, Palmieri M, di Ronza A, Medina DL, Valenza M, Gennarino VA, Di Malta C, Donaudy F, Embrione V, Polishchuk RS, Banfi S, Parenti G, Cattaneo E, Ballabio A (2009) A gene network regulating lysosomal biogenesis and function. Science 325:473–477
Seo BR, Lee SJ, Cho KS, Yoon YH, Koh JY (2015) The zinc ionophore clioquinol reverses autophagy arrest in chloroquine-treated ARPE-19 cells and in APP/mutant presenilin-1-transfected Chinese hamster ovary cells. Neurobiol Aging 36:3228–3238
Settembre C, Fraldi A, Medina DL, Ballabio A (2013) Signals from the lysosome: a control centre for cellular clearance and energy metabolism. Nat Rev Mol Cell Biol 14:283–296
Shen D, Wang X, Xu H (2011) Pairing phosphoinositides with calcium ions in endolysosomal dynamics: phosphoinositides control the direction and specificity of membrane trafficking by regulating the activity of calcium channels in the endolysosomes. BioEssays 33:448–457
Stoka V, Turk V, Turk B (2016) Lysosomal cathepsins and their regulation in aging and neurodegeneration. Ageing Res Rev 32:22–37
Tanaka Y, Suzuki G, Matsuwaki T, Hosokawa M, Serrano G, Beach TG, Yamanouchi K, Hasegawa M, Nishihara M (2017) Progranulin regulates lysosomal function and biogenesis through acidification of lysosomes. Hum Mol Genet 26:969–988
Tarasoff-Conway JM, Carare RO, Osorio RS, Glodzik L, Butler T, Fieremans E, Axel L, Rusinek H, Nicholson C, Zlokovic BV, Frangione B, Blennow K, Ménard J, Zetterberg H, Wisniewski T, de Leon MJ (2015) Clearance systems in the brain-implications for Alzheimer disease. Nat Rev Neurol 11:457–470
Tesco G, Koh YH, Kang EL, Cameron AN, Das S, Sena-Esteves M, Hiltunen M, Yang SH, Zhong Z, Shen Y, Simpkin JW, Tanzi RE (2007) Depletion of GGA3 stabilizes BACE and enhances beta-secretase activity. Neuron 54:721–737
Trudeau KM, Colby AH, Zeng J, Las G, Feng JH, Grinstaff MW, Shirihai OS (2016) Lysosome acidification by photoactivated nanoparticles restores autophagy under lipotoxicity. J Cell Biol 214:25–34
Tsunemi T, Krainc D (2014) Zn(2)(+) dyshomeostasis caused by loss of ATP13A2/PARK9 leads to lysosomal dysfunction and alpha-synuclein accumulation. Hum Mol Genet 23:2791–2801
Venkatachalam K, Wong CO, Zhu MX (2015) The role of TRPMLs in endolysosomal trafficking and function. Cell Calcium 58:48–56
Wang Y, Liu FT, Wang YX, Guan RY, Chen C, Li DK, Bu LL, Song J, Yang YJ, Dong Y, Chen Y, Wang J (2018) Autophagic modulation by Trehalose reduces accumulation of TDP-43 in a cell model of amyotrophic lateral sclerosis via TFEB activation. Neurotox Res 34:109–120
Xia Q, Wang H, Hao Z, Fu C, Hu Q, Gao F, Ren H, Chen D, Han J, Ying Z, Wang G (2016) TDP-43 loss of function increases TFEB activity and blocks autophagosome-lysosome fusion. EMBO J 35:121–142
Xiao Q, Yan P, Ma X, Liu H, Perez R, Zhu A, Gonzales E, Tripoli DL, Czerniewski L, Ballabio A, Cirrito JR, Diwan A, Lee JM (2015) Neuronal-targeted TFEB accelerates Lysosomal degradation of APP, reducing Aβ generation and amyloid plaque pathogenesis. J Neurosci 35:12137–12151
Yamada M, Tsuji S, Takahashi H (2002) Involvement of lysosomes in the pathogenesis of CAG repeat diseases. Ann Neurol 52:498–503
Acknowledgments
This research was supported by grants from the National Research Foundation of Korea (NRF) funded by the Ministry of Science, ICT (NRF-2016R1E1A1A01941212 and NRF-2017M3C7A1028949 for Jae-Young Koh) and the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (HI14C1913).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Kim, H.N., Seo, BR., Lee, SJ., Koh, JY. (2019). Possible Therapeutic Roles of Metallothionein-3 and Zinc in Endosome-Autophagosome-Lysosome Pathway (EALP) Dysfunction in Astrocytes. In: Fukada, T., Kambe, T. (eds) Zinc Signaling. Springer, Singapore. https://doi.org/10.1007/978-981-15-0557-7_10
Download citation
DOI: https://doi.org/10.1007/978-981-15-0557-7_10
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-15-0556-0
Online ISBN: 978-981-15-0557-7
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)