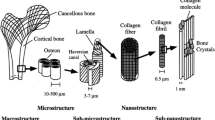
Abstract
A highly specialized connective tissue, bone, possesses inherent regenerative capacity. However, substantial degeneration and loss of bone due to bone tumour resections or traumatic injuries delay its healing, thereby suggesting alternate treatment options. Currently available treatments may offer repair to some extent; however, they are associated with certain disadvantages. Autografts fail under circumstances such as large bone loss and are associated with limited availability and donor site morbidity. Alternatives such as allografts are further associated with risk of immune rejection. Even if all of this pass, the limited availability of donors is unable to cover the associated clinical demands. In the search for bone repair and regeneration protocols, emergence of tissue engineering has greatly contributed to repair and regeneration of bone and bone-like complex tissues. Therefore, this chapter will uncover recent trends in bone tissue engineering with a focus on scaffolds, cells, growth factors and dynamic environments.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
Abbreviations
- ECM:
-
Extracellular matrix
- 3-D:
-
Three dimension
- HAp:
-
Hydroxyapatite
- Ti:
-
Titanium
- PLA:
-
Polylactic acid
- PGA:
-
Polyglycolic acid
- PLGA:
-
Poly(lactic-co-glycolic acid)
- PCL:
-
Polycaprolactone
- PLCL:
-
Poly(lactide-co-ε-caprolactone
- hADSCs:
-
Human adipose-derived stem cells
- BMSCs:
-
Bone-marrow-derived MSCs
- BSP:
-
Bone sialoprotein
- TCP:
-
Tricalcium phosphate
- ALP:
-
Alkaline phosphatase
- CPC:
-
Calcium phosphate cement
- hUCMSCs:
-
Human umblical cord-derived mesenchymal stem cells
- PVA:
-
Polyvinyl alcohol
- PEO:
-
Polyethylene oxide
- PAA:
-
Polyacrylic acid
- pDNA-NELL1:
-
Nel-like Type I molecular-1 DNA
- hAFSCs:
-
Human amniotic fluid-derived stem cells
- OX2:
-
Osterix
- RUNX2:
-
Runt-related transcription factor 2
- PEG:
-
Polyethylene glycol
- RGD:
-
Arg-Gly-Asp
- hESCd-MSC:
-
Human embryonic stem cell-derived mesenchymal stem cells
- PEGDA:
-
Polyethylene glycol diacrylate
- MSCs:
-
Mesenchymal stem cells
- HOB:
-
Human osteoblast cells
- ELR:
-
Elastin-like recombinamer
- hESCs:
-
Human embryonic stem cells
- iPSCs:
-
Induced pluripotent stem cells
- BMP-2:
-
Bone morphogenetic protein 2
- BMP-7:
-
Bone morphogenetic protein 7
- TPS:
-
Tubular perfusion system
- IL-1:
-
Interleukin-1
- IL-6:
-
Interleukin-6
- TNF-α:
-
Tumour necrosis factor alpha
- FGF-2:
-
Fibroblast growth factor 2
- M-CSF:
-
Macrophage colony-stimulating factor
- PDGF:
-
Platelet-derived growth factor
- BMPs:
-
Bone morphogenetic proteins
- VEGF:
-
Vascular endothelial growth factor
- TGF-β:
-
Transforming growth factor beta
- IGFs:
-
Insulin-like growth factors
- bFBF:
-
Basic fibroblast growth factor
- LbL:
-
Layer by layer
- MMP:
-
Matrix metalloproteinase
- PD-MCG:
-
Polydopamine-coated multichannel biphasic calcium phosphate granule system
- BCP:
-
Biphasic calcium phosphate scaffolds
- CFD:
-
Computational fluid dynamics
- RPM:
-
Rotations per minute
- RWV:
-
Rotating wall vessel
- EMF:
-
Electromagnetic field
- PEMF:
-
Pulsed electromagnetic field
- GMP:
-
Good manufacturing practice
- Micro-CT:
-
Microcomputed tomography
- CAD:
-
Computer-aided design
References
Florencio-silva R, Rodrigues G, Sasso-cerri E (2015) Biology of bone tissue : structure, function, and factors that influence bone cells. Biomed Res Int. https://doi.org/10.1155/2015/421746
Weiner S, Wagner HD (1998) The material bone: structure-mechanical function relations. Annu Rev Mater Sci 28:271–298
Gordon JAR, Tye CE, Sampaio AV (2007) Bone sialoprotein expression enhances osteoblast differentiation and matrix mineralization in vitro. J Bone 41:462–473
Kozhemyakina E, Lassar AB, Zelzer E (2015) A pathway to bone: signaling molecules and transcription factors involved in chondrocyte development and maturation. Development 142:817–831
Allen MR, Burr DB (2014) Bone modeling and remodeling. In: Burr DB, Allen MRBT-B ABB (eds) Academic Press, San Diego, pp 75–90
Gruber R, Koch H, Doll BA (2006) Fracture healing in the elderly patient. Exp Gerontol 41:1080–1093
Gandhi A, Liporace F, Azad V (2006) Diabetic fracture healing. Foot Ankle Clin 11:805–824
Wukich DK, Kline AJ (2008) The management of ankle fractures in patients with diabetes. J Bone Jt Surg–Am 90:1570–1578
Lu C, Hansen E, Sapozhnikova A (2010) Effect of age on vascularization during fracture repair. J Orthop Res 26:1384–1389
Mobini S, Ayoub A (2016) Bone tissue engineering in the maxillofacial region: the state-of-the- art practice and future prospects. Regen Reconstr Restor 1:8–14
Amini AR, Laurencin CT, Nukavarapu SP (2012) Bone tissue engineering: recent advances and challenges. Crit Rev Biomed Eng 40:363–408
Delloye C, Cornu O, Druez V, Barbier O (2007) Bone allografts: what they can offer and what they cannot. J Bone Jt Surg 89–B:5:574–580
Yeatts AB, Fisher JP (2011) Bone tissue engineering bioreactors: dynamic culture and the influence of shear stress. Bone 48:171–181
Della Porta G, Nguyen BB, Campardelli R (2014) Synergistic effect of sustained release of growth factors and dynamic culture on osteoblastic differentiation of mesenchymal stem cells. J Biomed Mater Res Part A 103:2161–2171
Nguyen BB, Ko H, Moriarty RA (2016) dynamic bioreactor culture of high volume engineered bone tissue. Tissue Eng Part A 22:263–271
Brien FJO (2011) Biomaterials & scaffolds for tissue engineering. Mater Today 14:88–95
Hutmacher DW (2000) Scaffolds in tissue engineering bone and cartilage. Biomaterials 21:2529–2543
Williams DF (2008) On the mechanisms of biocompatibility. Biomaterials 29:2941–2953
Prasadh S, Chung R, Wong W (2018) Unraveling the mechanical strength of biomaterials used as a bone scaffold in oral and maxillofacial defects. Oral Sci Int 15:48–55
Zhao H, Liang W (2017) A novel comby scaffold with improved mechanical strength for bone tissue engineering. Mater Lett 194:220–223
Chen Y, Frith JE, Dehghan- A (2017) Mechanical properties and biocompatibility of porous titanium scaffolds for bone tissue engineering. J Mech Behav Biomed Mater 75:169–174
Li G, Wang L, Pan W (2016) In vitro and in vivo study of additive manufactured porous Ti6Al4V scaffolds for repairing bone defects. Sci Rep 6:1–11
Zhang X, Zeng D, Li N (2016) Functionalized mesoporous bioactive glass scaffolds for enhanced bone tissue regeneration. Sci Rep 6:19361
Nisal A, Sayyad R, Dhavale P (2018) Silk fibroin micro-particle scaffolds with superior compression modulus and slow bioresorption for effective bone regeneration. Sci Rep 1–10
Persson M, Lehenkari PP, Berglin L (2018) Osteogenic differentiation of human mesenchymal stem cells in a 3D woven scaffold. Sci Rep 8:1–12
Schieker M, Seitz H, Drosse I (2006) Biomaterials as scaffold for bone tissue engineering. Eur J Trauma 32:114–124
Alaribe FN, Manoto SL, Motaung SCKM (2016) Scaffolds from biomaterials: advantages and limitations in bone and tissue engineering. Biologia (Bratisl) 71:353–366
Puppi D, Chiellini F, Piras AM, Chiellini E (2010) Progress in polymer science polymeric materials for bone and cartilage repair. Prog Polym Sci 35:403–440
Wang Z, Lin M, Xie Q (2016) Electrospun silk fibroin/poly(lactide-co-ε-caprolactone) nanofibrous scaffolds for bone regeneration. Int J Nanomed 11:1483–1500
Zhang Y, Reddy VJ, Wong SY (2010) Enhanced biomineralization in osteoblasts on a novel electrospun biocomposite nanofibrous substrate. Tissue Eng Part A 16:1949–1960
Jing Z, Wu Y, Su W (2017) carbon nanotube reinforced collagen/hydroxyapatite scaffolds improve bone tissue formation in vitro and in vivo. Ann Biomed Eng 45:2075–2087
Chevalier J, Gremillard L (2009) Ceramics for medical applications: a picture for the next 20 years. J Eur Ceram Soc 29:1245–1255
Thein-han W, Xu HHK (2011) Collagen-calcium phosphate cement scaffolds seeded with umbilical cord stem cells for bone tissue engineering. Tissue Eng Part A 17:2943–2954
Bose S, Roy M, Bandyopadhyay A (2012) Recent advances in bone tissue engineering scaffolds. Trends Biotechnol 30:546–554
Asaoka T, Ohtake S, Furukawa KS (2013) Development of bioactive porous a-TCP/HAp beads for bone tissue engineering. J Biomed Mater Res, Part A 101:3295–3300
Wu C, Chang J (2007) Degradation, bioactivity, and cytocompatibility of diopside, akermanite, and bredigite ceramics. J Biomed Mater Res Part B Appl Biomater 83B:153–160
Fu Q, Saiz E, Rahaman MN, Tomsia AP (2011) Bioactive glass scaffolds for bone tissue engineering: state of the art and future perspectives. Mater Sci Eng, C 31:1245–1256
Xia L, Yin Z, Mao L (2016) Akermanite bioceramics promote osteogenesis, angiogenesis and suppress osteoclastogenesis for osteoporotic bone regeneration. Sci Rep 6:1–17
Jones JR (2013) Review of bioactive glass: from Hench to hybrids. Acta Biomater 9:4457–4486
Philippart A, Boccaccini AR, Fleck C (2015) Toughening and functionalization of bioactive ceramic and glass bone scaffolds by biopolymer coatings and infiltration: a review of the last 5 years. Expert Rev Med Devices 12:93–111
Staiger MP, Pietak AM, Huadmai J, Dias G (2006) Magnesium and its alloys as orthopedic biomaterials: a review. Biomaterials 27:1728–1734
Alvarez K, Nakajima H (2009) Metallic scaffolds for bone regeneration. Materials (Basel) 2:790–832
Hussein MA, Mohammed AS, Al-aqeeli N, Arabia S (2015) Wear characteristics of metallic biomaterials: a review. Materials (Basel) 8:2749–2768
Ghassemi T, Shahroodi A, Ebrahimzadeh MH, Mousavian A (2018) Current concepts in scaffolding for bone tissue engineering. Arch Bone Jt Surg 6:90–99
Bhui AS, Singh G, Sidhu SS, Bains PS (2018) Experimental investigation of optimal ED machining parameters for Ti-6Al-4V biomaterial. FU Ser Mech Eng 16(3):337–345
Tamaddon M, Samizadeh S, Wang L (2017) Intrinsic osteoinductivity of porous titanium scaffold for bone tissue engineering. Int J Biomater 2017:5093063
Bobe K, Willbold E, Morgenthal I (2013) In vitro and in vivo evaluation of biodegradable, open-porous scaffolds made of sintered magnesium W4 short fibres. Acta Biomater 9:8611–8623
Wu C, Zhou Y, Xu M (2013) Copper-containing mesoporous bioactive glass scaffolds with multifunctional properties of angiogenesis capacity, osteostimulation and antibacterial activity. Biomaterials 34:422–433
Declercq HA, Desmet T, Dubruel P, Cornelissen MJ (2014) The role of scaffold architecture and composition on the bone formation by adipose-derived stem cells. Tissue Eng Part A 20:434–444
Kim YE, Kim Y (2013) Effect of biopolymers on the characteristics and cytocompatibility of biocomposite nanofibrous scaffolds. Polym J 45:845–853
Roohani-Esfahani SI, Newman P, Zreiqat H (2016) Design and fabrication of 3D printed scaffolds with a mechanical strength comparable to cortical bone to repair large bone defects. Sci Repor 1–8
Leite ÁJ, Oliveira NM, Song W, Mano JF (2018) Bioactive hydrogel marbles. Sci Rep 8:1–11
Sato Y, Yamamoto K, Horiguchi S (2018) Nanogel tectonic porous 3D scaffold for direct reprogramming fibroblasts into osteoblasts and bone regeneration. Sci Rep 8:15824
Nicodemus GD, Bryant SJ (2008) Cell encapsulation in biodegradable hydrogels for tissue engineering applications. Tissue Eng Part B 14:149–165
Short AR, Koralla D, Deshmukh A (2015) Hydrogels that allow and facilitate bone repair, remodeling, and regeneration. J Mater Chem B 3:7818–7830
Tibbitt MW, Anseth KS (2009) Hydrogels as extracellular matrix mimics for 3D cell culture. Biotechnol Bioeng 103:655–663
Yang X, Sun T, Dou S (2009) Block copolymer of polyphosphoester and poly (l-Lactic Acid) modified surface for enhancing osteoblast adhesion, proliferation, and function. Biomacromol 10:2213–2220
Thambi T, Li Y, Lee DS (2017) Injectable hydrogels for sustained release of therapeutic agents. J Control Release 267:57–66
Liu M, Zeng X, Ma C (2017) Injectable hydrogels for cartilage and bone tissue engineering. Bone Res 5:17014
Han Y, Zeng Q, Li H, Chang J (2013) The calcium silicate/ alginate composite: preparation and evaluation of its behavior as bioactive injectable hydrogels. Acta Biomater 9:9107–9117
Ding C, Zhao L, Liu F (2010) Dually responsive injectable hydrogel prepared by in situ cross-linking of glycol chitosan and benzaldehyde-capped PEO-PPO-PEO. Biomacromol 11:1043–1051
Dessı M, Borzacchiello A, Mohamed THA (2013) Novel biomimetic thermosensitive b -tricalcium phosphate/ chitosan-based hydrogels for bone tissue engineering. J Biomed Mater Res - Part A 101:2984–2993
Vo TN, Shah SR, Lu S (2016) Injectable dual-gelling cell-laden composite hydrogels for bone tissue engineering. Biomaterials 83:1–11
Fu S, Ni P, Wang B (2012) Injectable and thermo-sensitive PEG-PCL-PEG copolymer/collagen/n-HA hydrogel composite for guided bone regeneration. Biomaterials 33:4801–4809
Jiao Y, Gyawali D, Stark JM (2012) A rheological study of biodegradable injectable PEGMC/HA composite scaffolds. Soft Matter 8:1499–1507
Niranjan R, Koushik C, Saravanan S (2013) A novel injectable temperature-sensitive zinc doped chitosan/beta-glycerophosphate hydrogel for bone tissue engineering. Int J Biol Macromol 54:24–29
Douglas TEL, Piwowarczyk W, Pamula E (2014) Injectable self-gelling composites for bone tissue engineering based on gellan gum hydrogel enriched with different bioglasses. Biomed Mater 9:045014
Dhivya S, Saravanan S, Sastry TP, Selvamurugan N (2015) Nanohydroxyapatite—reinforced chitosan composite hydrogel for bone tissue repair in vitro and in vivo. J Nanobiotechnology 13:40
Lewandowska-ła J, Fiejdasz S, Rodzik Ł (2015) Bioactive hydrogel-nanosilica hybrid materials: a potential injectable scaffold for bone tissue engineering. Biomed Mater 10:015020
Huang Y, Zhang X, Wu A, Xu H (2016) Injectable nano-hydroxyapatite (n-HA)/glycol chitosan(G-CS)/hyaluronic acid (HyA) composite hydrogel for bone tissue engineering. RSC Adv 6:33529–33536
Tan R, She Z, Wang M (2012) Thermo-sensitive alginate-based injectable hydrogel for tissue engineering. Carbohydr Polym 87:1515–1521
Celikkin N, Mastrogiacomo S, Jaroszewicz J, Walboomers XF (2018) Gelatin methacrylate scaffold for bone tissue engineering: the influence of polymer concentration. J Biomed Mater Res Part A 106A:201–209
Ko W, Lee JS, Hwang Y (2018) Injectable hydrogel composite containing modified gold nanoparticles: implication in bone tissue regeneration. Int J Nanomedicine 13:7019–7031
Arya N, Sardana V, Saxena M (2012) Recapitulating tumour microenvironment in chitosan-gelatin three-dimensional scaffolds: an improved in vitro tumour model. J R Soc Interface 9:3288–3302
Bencherif SA, Braschler TM, Renaud P (2013) Advances in the design of macroporous polymer scaffolds for potential applications in dentistry. J Periodontal Implant Sci 43:251–261
Elamparithi D, Moorthy V (2017) On various porous scaffold fabrication methods. Mapana J Sci 16:47–52
Tamburaci S, Tihminlioglu F (2018) Materials science & engineering C Biosilica incorporated 3D porous scaffolds for bone tissue engineering applications. Mater Sci Eng, C 91:274–291
Demirtas TT, Irmak G, Gümüşderelioglu M (2017) Bioprintable form of chitosan hydrogel for bone tissue engineering. Biofabrication 9:035003
Moncal KK, Heo DN, Godzik KP (2018) 3D printing of poly (e-caprolactone)/poly (D, L-lactide-co-glycolide)/hydroxyapatite composite constructs for bone tissue engineering. J Mater Res 33:1972–1986
Nandi SK, Fielding G, Banerjee D (2018) 3D-printed b-TCP bone tissue engineering scaffolds: effects of chemistry on in vivo biological properties in a rabbit tibia model. J Mater Res 33:1939–1947
Zhang J, Chen Y, Xu J (2018) Tissue engineering using 3D printed nano-bioactive glass loaded with NELL1 gene for repairing alveolar bone defects. Regen Biomater 5:213–220
Ma H, Feng C, Chang J, Wu C (2018) 3D-printed bioceramic scaffolds : from bone tissue engineering to tumor therapy. Acta Biomater 1–23
Jammalamadaka U, Tappa K (2018) Recent advances in biomaterials for 3D printing and tissue engineering. J Funct Biomater 9:14
Karageorgiou V, Kaplan D (2005) Porosity of 3D biomaterial scaffolds and osteogenesis. Biomaterials 26:5474–5491
Guda T, Walker JA, Singleton B (2014) Hydroxyapatite scaffold pore architecture effects in large bone defects in vivo. J Biomater Appl 28:1016–1027
Gupte MJ, Swanson WB, Hu J (2018) Pore size directs bone marrow stromal cell fate and tissue regeneration in nanofibrous macroporous scaffolds by mediating vascularization. Acta Biomater 82:1–11
Xu M, Zhai D, Chang J, Wu C (2014) In vitro assessment of three-dimensionally plotted nagelschmidtite bioceramic scaffolds with varied macropore morphologies. Acta Biomater 10:463–476
Smith LA, Liu X, Ma PX (2008) Tissue engineering with nano-fibrous scaffolds. Soft Matter 4:2144–2149
Liu H, Ding X, Zhou G (2013) Electrospinning of nanofibers for tissue engineering applications. J Nanomater 2013:495708
Lyu S, Huang C, Yang H, Zhang X (2013) Electrospun fibers as a scaffolding platform for bone tissue repair. J Orthop Res 31:1382–1389
Guo Z, Xu J, Ding S (2015) In vitro evaluation of random and aligned polycaprolactone/gelatin fibers via eletrospinning for bone tissue engineering. J Biomater Sci Polym Ed 26:989–1001
Wang Y, Cai X, Wang Y (2016) Enhanced osteogenesis of BMP2-Transfected human periodontal ligament stem cells by aligned electrospun scaffolds for bone tissue engineering. J Biomater Tissue Eng 6:563–573
Chen H, Qian Y, Xia Y (2016) Enhanced osteogenesis of ADSCs by the synergistic effect of aligned fibers containing collagen I. ACS Appl Mater Interfaces 8:29289–29297
Stevens MM (2008) Biomaterials for bone tissue engineering. Mater Today 11:18–25
Turnbull G, Clarke J, Picard F (2018) 3D bioactive composite scaffolds for bone tissue engineering. Bioact Mater 3:278–314
De Witte T, Fratila-apachitei LE, Zadpoor AA, Peppas NA (2018) Bone tissue engineering via growth factor delivery: from scaffolds to complex matrices. Regen Biomater 5:197–211
Benoit DSW, Schwartz MP, Durney AR, Anseth KS (2008) Small functional groups for controlled differentiation of hydrogel-encapsulated human mesenchymal stem cells. Nat Mater 7:816–823
Arora A, Katti DS (2016) Understanding the influence of phosphorylation and polysialylation of gelatin on mineralization and osteogenic differentiation. Mater Sci Eng, C 65:9–18
Jaidev LR, Chatterjee K (2019) Surface functionalization of 3D printed polymer scaffolds to augment stem cell response. Mater Des 161:44–54
Yang F, Williams CG, Wang D-A (2005) The effect of incorporating RGD adhesive peptide in polyethylene glycol diacrylate hydrogel on osteogenesis of bone marrow stromal cells. Biomaterials 26:5991–5998
Chen W, Zhou H, Weir MD (2012) Human embryonic stem cell-derived mesenchymal stem cell seeding on calcium phosphate cement-chitosan-RGD scaffold for bone repair. Tissue Eng Part A 21201:1–37
Beck G, Crichton HJ, Baer E (2014) Surface modifying oligomers used to functionalize polymeric surfaces: consideration of blood contact applications. J Appl Polym Sci 131:40328
Gümüsderelioglu M, Aday S (2011) Heparin-functionalized chitosan scaffolds for bone tissue engineering. Carbohydr Res 346:606–613
Kim HD, Lee EA, An Y (2017) Chondroitin sulfate-based biomineralizing surface hydrogels for bone tissue engineering. ACS Appl Mater Interfaces 9:21639–21650
Downey PA, Siegel MI (2006) Bone biology and the clinical implications for osteoporosis. Phys Ther 86:77–91
Cancedda R, Giannoni P, Mastrogiacomo M (2007) A tissue engineering approach to bone repair in large animal models and in clinical practice. Biomaterials 28:4240–4250
Heath CA (2000) Cells for tissue engineering. Trends Biotechnol 18:17–19
Krampera M, Pizzolo G, Aprili G, Franchini M (2006) Mesenchymal stem cells for bone, cartilage, tendon and skeletal muscle repair. Bone 39:678–683
Uccelli A, Moretta L, Pistoia V (2008) Mesenchymal stem cells in health and disease. Nat Rev Immunol 8:726–736
Hanson S, Souza RND, Hematti P (2014) Biomaterial-mesenchymal stem cell constructs for immunomodulation in composite tissue engineering. Tissue Eng Part A 20:2162–2168
Nassif L, Jurjus A, Nassar J (2012) Enhanced in vivo bone formation by bone marrow differentiated mesenchymal stem cells grown in chitosan scaffold. J Bioeng Biomed Sci 2:2–7
Yu L, Wu Y, Liu J (2018) 3D culture of bone marrow-derived mesenchymal stem cells (BMSCs) could improve bone regeneration in 3D-printed porous Ti6Al4V scaffolds. Stem Cells Int 2018:2074021
Krampera M, Pasini A, Pizzolo G (2006) Regenerative and immunomodulatory potential of mesenchymal stem cells. Curr Opin Pharmacol 6:435–441
Caplan AI (2007) Adult mesenchymal stem cells for tissue engineering versus regenerative medicine. J Cell Physiol 213:341–347
Frese L, Dijkman E, Hoerstrup SP (2016) Adipose tissue-derived stem cells in regenerative medicine. Transfus Med Hemotherapy 43:268–274
Calabrese G, Giuff R, Forte S (2017) Human adipose-derived mesenchymal stem cells seeded into a collagen-hydroxyapatite scaffold promote bone augmentation after implantation in the mouse. Sci Rep 7:7110
Sayin E, Rashid RH, Rodríguez-Cabello JC (2017) Human adipose derived stem cells are superior to human osteoblasts (HOB) in bone tissue engineering on a collagen- fibroin-ELR blend. Bioact Mater 1–11
Oryan A, Kamali A, Moshiri A, Eslaminejad MB (2017) Role of mesenchymal stem cells in bone regenerative medicine: what is the evidence? Cells Tissues Organs 204:59–83
Yu J, Wang Y, Deng Z (2007) Odontogenic capability: bone marrow stromal stem cells versus dental pulp stem cells. Biol Cell 99:465–474
El-gendy R, Yang XB, Newby PJ (2013) Osteogenic differentiation of human dental pulp stromal cells on 45S5 bioglass based scaffolds in vitro and in vivo. Tissue Eng Part A 19:707–715
Petridis X, Diamanti E, Trigas GC, Kalyvas D (2015) Bone regeneration in critical-size calvarial defects using human dental pulp cells in an extracellular matrix-based scaffold. J Craniomaxillofac Surg
La M, Paino F, Spina A (2014) Dental pulp stem cells: state of the art and suggestions for a true translation of research into therapy. J Dent 42:761–768
Tian X, Heng B, Ge Z (2008) Comparison of osteogenesis of human embryonic stem cells within 2D and 3D culture systems. Scand J Clin Lab Investig 68:58–67
De Peppo GM, Marcos-campos I, John D (2013) Engineering bone tissue substitutes from human induced pluripotent stem cells. Proc Natl Acad Sci 110:8680–8685
Marolt D, Marcos I, Bhumiratana S (2012) Engineering bone tissue from human embryonic stem cells. Proc Natl Acad Sci 109:1–5
Vats A, Tolley NS, Bishop AE, Polak JM (2005) Embryonic stem cells and tissue engineering: delivering stem cells to the clinic. J R Soc Med 98:346–350
Swijnenburg R, Schrepfer S, Govaert JA (2008) Immunosuppressive therapy mitigates immunological rejection of human embryonic stem cell xenografts. Proc Natl Acad Sci 105:12991–12996
Wu Q, Yang B, Hu K (2017) Deriving osteogenic cells from induced pluripotent stem cells for bone tissue engineering. Tissue Eng Part B Rev 23:1–8
Jin G, Kim T, Kim J (2012) Bone tissue engineering of induced pluripotent stem cells cultured with macrochanneled polymer scaffold. J Biomed Mater Res Part A 101:1283–1291
Hayashi K, Ochiai-shino H, Shiga T (2016) Transplantation of human-induced pluripotent stem cells carried by self-assembling peptide nanofi ber hydrogel improves bone regeneration in rat calvarial bone defects. BDJOpen 2:1–7
Csobonyeiova M, Polak S, Zamborsky R, Danisovic L (2017) iPS cell technologies and their prospect for bone regeneration and disease modeling: a mini review. J Adv Res 8:321–327
Xie J, Peng C, Zhao Q (2015) Osteogenic differentiation and bone regeneration of the iPSC-mscs supported by a biomimetic nanofibrous scaffold. Acta Biomater 29:365–379
Jung Y, Bauer G, Nolta JA (2012) Concise review: induced pluripotent stem cell-derived mesenchymal stem cells: progress toward safe clinical products. Stem Cells 30:42–47
Reible B, Schmidmaier G, Prokscha M, Westhauser F (2017) Continuous stimulation with differentiation factors is necessary to enhance osteogenic differentiation of human mesenchymal stem cells. Growth Factors 35:179–188
Kimelman N, Pelled G, Helm GA (2007) Review: gene- and stem cell-based therapeutics for bone regeneration and repair. Tissue Eng 13:1135–1150
Huynh NPT, Brunger JM, Gloss CC (2018) Genetic engineering of mesenchymal stem cells for differential matrix deposition on 3D woven scaffolds. Tissue Eng Part A 00:1–14
Kuttappan S, Anitha A, Minsha MG (2018) BMP2 expressing genetically engineered mesenchymal stem cells on composite fibrous scaffolds for enhanced bone regeneration in segmental defects. Mater Sci Eng, C 85:239–248
Kargozar S, Hashemian SJ, Soleimani M (2017) Acceleration of bone regeneration in bioactive glass/gelatin composite scaffolds seeded with bone marrow-derived mesenchymal stem cells over-expressing bone morphogenetic protein-7. Mater Sci Eng, C 75:688–698
Sayed N, Liu C, Wu JC (2016) Translation of human-induced pluripotent stem cells from clinical trial in a dish to precision medicine. J Am Coll Cardiol 67:2161–2176
Martin U (2017) Therapeutic application of pluripotent stem cells: challenges and risks. Front Med 4:229
Raggatt LJ, Partridge NC (2010) Cellular and molecular mechanisms of bone remodeling. J Biol Chem 285:25103–25108
Kook Y, Jeong Y, Lee K, Koh W (2017) Design of biomimetic cellular scaffolds for co-culture system and their application. J Tissue Eng 8:1–17
Beşkardeş IG, Hayden RS, Glettig DL (2017) Bone tissue engineering with scaffold-supported perfusion co-cultures of human stem cell-derived osteoblasts and cell line-derived osteoclasts. Process Biochem 59:303–311
Jeon OH, Panicker LM, Lu Q (2016) Human iPSC-derived osteoblasts and osteoclasts together promote bone regeneration in 3D biomaterials. 1–11
Lovett M, Ph D, Lee K (2009) Vascularization strategies for tissue engineering. Tissue Eng Part B 15:353–370
Gurel G, Torun G, Hasirci V (2016) In fl uence of co-culture on osteogenesis and angiogenesis of bone marrow mesenchymal stem cells and aortic endothelial cells. Microvasc Res 108:1–9
Jin G, Kim H (2017) Co-culture of human dental pulp stem cells and endothelial cells using porous biopolymer microcarriers: a feasibility study for bone tissue engineering. Tissue Eng anf Regen Med 14:393–401
Nguyen BB, Moriarty RA, Kamalitdinov T (2018) Collagen hydrogel scaffold promotes mesenchymal stem cell and endothelial cell coculture for bone tissue engineering. J Biomed Mater Res Part A 105:1123–1131
Lee K, Silva EA, Mooney DJ (2011) Growth factor delivery-based tissue engineering: general approaches and a review of recent developments. J R Soc Interface 8:153–170
Canalis E (2009) Prospect-growth factor control of bone mass. J Cell Biochem 108:769–777
Li F, Niyibizi C (2012) Cells derived from murine induced pluripotent stem cells (iPSC) by treatment with members of TGF-beta family give rise to osteoblasts differentiation and form bone in vivo. BMC Cell Biol 13:35
Nyberg E, Holmes C, Witham T, Grayson WL (2015) Growth factor-eluting technologies for bone tissue engineering. Drug Deliv Transl Res 6:184–194
Vo TN, Kasper FK, Mikos AG (2012) Strategies for controlled delivery of growth factors and cells for bone regeneration. Adv Drug Deliv Rev. https://doi.org/10.1016/j.addr.2012.01.016
Hankenson K, Gagne K, Shaughnessy M (2015) Extracellular signaling molecules to promote fracture healing and bone regeneration. Adv Drug Deliv Rev 94:3–12. https://doi.org/10.1016/j.addr.2015.09.008
Lienemann PS, Lutolf MP, Ehrbar M (2012) Biomimetic hydrogels for controlled biomolecule delivery to augment bone regeneration. Adv Drug Deliv Rev 64:1078–1089
Kowalczewski CJ, Saul JM (2018) Biomaterials for the delivery of growth factors and other therapeutic agents in tissue engineering approaches to bone regeneration. Front Pharmacol 9:513
Marenzana M, Arnett TR (2013) The key role of the blood supply to bone. Bone Res 1:203–215
Nauta TD, Van Hinsbergh VWM, Koolwijk P (2014) Hypoxic signaling during tissue repair and regenerative medicine. Int J Mol Sci 15:19791–19815
Mercado-Pagan AE, Stahl AM, Shanjani Y, Yang Y (2015) Vascularization in bone tissue engineering constructs. Ann Biomed Eng 43:718–729
Nikitovic D, Zafiropoulos A, Tzanakakis GN (2005) Effects of glycosaminoglycans on cell proliferation of normal osteoblasts and human osteosarcoma cells depend on their type and fine chemical compositions. Anticancer Res 25:2851–2856
Nauth A, Ristevski B, Li R, Schemitsch EH (2011) Growth factors and bone regeneration: how much bone can we expect? Injury 42:574–579
Farokhi M, Mottaghitalab F, Shokrgozar MA (2016) Importance of dual delivery systems for bone tissue engineering. J Control Release 225:152–169
Carragee EJ, Comer G, Chu G (2013) Cancer risk after use of recombinant bone. J Bone Jt Surg 95:1537–1545
Wang W, Yeung KWK (2017) Bioactive materials bone grafts and biomaterials substitutes for bone defect repair: a review. Bioact Mater 2:224–247
Fernandez-Yague MA, Abbah SA, McNamara L (2014) Biomimetic approaches in bone tissue engineering: integrating biological and physicomechanical strategies. Adv Drug Deliv Rev 84:1–29
Brown KV, Lon M, Li B (2011) Improving bone formation in a rat femur segmental defect by controlling bone morphogenetic protein-2 release. Tissue Eng Part A 17:1735–1746
Ziegler J, Anger D, Krummenauer F (2007) Biological activity of recombinant human growth factors released from biocompatible bone implants. https://doi.org/10.1002/jbm.a.31625
Murphy WL, Peters MC, Kohn DH, Mooney DJ (2000) Sustained release of vascular endothelial growth factor from mineralized poly (lactide-co-glycolide) scaffolds for tissue engineering. Biomater 21:2521–2527
Reyes R, De B, Delgado A (2012) Effect of triple growth factor controlled delivery by a brushite—PLGA system on a bone defect. Injury 43:334–342
Wang Y, Angelatos AS, Caruso F (2008) Template synthesis of nanostructured materials via layer-by-layer. Chem Mater 20:848–858
Wang Z, Wang Z, Lu WW (2017) Novel biomaterial strategies for controlled growth factor delivery for biomedical applications. NPG Asia Mater 9:e435
Richardson JJ, Björnmalm M, Caruso F (2015) Technology-driven layer-by-layer assembly of nanofilms. Science 80(348):411–424
Macdonald ML, Samuel RE, Shah NJ (2011) Tissue integration of growth factor-eluting layer-by-layer polyelectrolyte multilayer coated implants. Biomaterials 32:1446–1453
Shah NJ, Macdonald ML, Beben YM (2011) Tunable dual growth factor delivery from polyelectrolyte multilayer films. Biomaterials 32:6183–6193
Bouyer M, Guillot R, Jonathan L (2016) Surface delivery of tunable doses of BMP-2 from an adaptable polymeric scaffold induces volumetric bone regeneration. Biomaterials 104:168–181
Newman MR, Benoit DSW (2016) Local and targeted drug delivery for bone regeneration. Curr Opin Biotechnol 40:125–132
Draenert FG, Nonnenmacher A, Ka PW (2012) BMP-2 and bFGF release and in vitro effect on human osteoblasts after adsorption to bone grafts and biomaterials. Clin Oral Implant Res 24:750–757
Masters KS (2011) Covalent growth factor immobilization strategies for tissue repair and regeneration. Macromol Biosci 11:1149–1163
Di Luca A, Klein-gunnewiek M, Vancso JG (2017) Covalent binding of bone morphogenetic protein-2 and transforming growth factor-β3 to 3D plotted scaffolds for osteochondral tissue regeneration. Biotechnol J 12:1700072
Madl CM, Mehta M, Duda GN (2013) Presentation of BMP-2 mimicking peptides in 3D hydrogels directs cell fate commitment in osteoblasts and mesenchymal stem cells. Biomacromol 15:445–455
Karageorgiou V, Meinel L, Hofmann S (2004) Bone morphogenetic protein-2 decorated silk fibroin films induce osteogenic differentiation of human bone marrow stromal cells. J Biomed Mater Res Part A 71A:528–537
Lee H, Dellatore SM, Miller WM, Messersmith PB (2007) Mussel-inspired surface chemistry for multifunctional coatings. Science (80-) 318:426–431
Lee GH, Paul K, Lee B (2017) Development of BMP-2 immobilized polydopamine mediated multichannelled biphasic calcium phosphate granules for improved bone regeneration. Mater Lett 208:122–125
Geiger M, Li RH, Friess W (2003) Collagen sponges for bone regeneration with rhBMP-2. Adv Drug Deliv Rev 55:1613–1629
Upton Z, Cuttle L, Noble A (2008) Vitronectin: growth factor complexes hold potential as a wound therapy approach. J Invest Dermatol 128:1534–1544
Schultz GS, Wysocki A (2009) Interactions between extracellular matrix and growth factors in wound healing. Wound Repair Regen 17:153–162
Martino MM, Briquez PS, Ranga A (2013) Heparin-binding domain of fibrin(ogen) binds growth factors and promotes tissue repair when incorporated within a synthetic matrix. Proc Natl Acad Sci 110:4563–4568
Yue B (2014) Biology of the extracellular matrix: an overview. J Glaucoma 23:S20–S23
Capila I, Linhardt RJ (2002) Heparin-protein interactions. Angew Chem Int Ed 41:390–412
Macri L, Silverstein D, Clark RAF (2007) Growth factor binding to the pericellular matrix and its importance in tissue engineering. Adv Drug Deliv Rev 59:1366–1381
Billings PC, Yang E, Mundy C, Pacifici M (2018) Domains with highest heparan sulfate-binding affinity reside at opposite ends in BMP2/4 versus BMP5/6/7: implications for function. J Biol Chem 293:14371–14383
Kim T, Yun Y, Park Y, Lee S (2014) In vitro and in vivo evaluation of bone formation using solid freeform fabrication-based bone morphogenic protein-2 releasing PCL/PLGA scaffolds. Biomed Mater 9:025008
Martino MM, Tortelli F, Mochizuki M (2011) Engineering the growth factor microenvironment with fibronectin domains to promote wound and bone tissue healing. Sci Transl Med 3:100ra89
Sakiyama-elbert SE, Hubbell JA (2000) Development of fibrin derivatives for controlled release of heparin-binding growth factors. J Control Release 65:389–402
Jha AK, Mathur A, Svedlund FL (2015) Molecular weight and concentration of heparin in hyaluronic acid-based matrices modulates growth factor retention kinetics and stem cell fate. J Control Release 209:308–316
Vieira S, Vial S, Reis RL, Oliveira JM (2017) Nanoparticles for bone tissue engineering. Biotechnol Prog 33:590–611
Chiellini F, Piras AM, Errico C (2008) Micro/nanostructured polymeric systems for biomedical and pharmaceutical applications. Nanomedicine 3:367–393
Wang Z, Wang K, Lu X (2014) BMP-2 encapsulated polysaccharide nanoparticle modified biphasic calcium phosphate scaffolds for bone tissue regeneration. J Biomed Mater Res Part A 103:1520–1532
Kim B, Yang S, Sang C (2018) Incorporation of BMP-2 nanoparticles on the surface of a 3D-printed hydroxyapatite scaffold using an ε -polycaprolactone polymer emulsion coating method for bone tissue engineering. Colloids Surf B 170:421–429
Eimori K, Endo N, Uchiyama S, Takahashi Y (2016) Disrupted bone metabolism in long-term bedridden patients. PLoS ONE 11:e0156991
Wittkowske C, Reilly GC, Lacroix D, Perrault CM (2016) In vitro bone cell models: impact of fluid shear stress on bone formation. Front Bioeng Biotechnol 4:87
Mishra R, Bishop T, Valerio IL (2016) The potential impact of bone tissue engineering in the clinic. Regen Med 11:571–587
Volkmer E, Drosse I, Otto S (2008) Hypoxia in static and dynamic 3D culture systems for tissue engineering of bone. Tissue Eng Part A 14:1331–1340
Grellier M, Bareille R, Bourget C (2009) Responsiveness of human bone marrow stromal cells to shear stress. J Tissue Eng Regen Med 2:302–309
Yourek G, McCormick SM, Mao JJ, Reilly GC (2010) Shear stress induces osteogenic differentiation of human mesenchymal stem cells. Regen Med 5:713–724
Singh H, Hutmacher DW (2009) Bioreactor studies and computational fluid dynamics. In: Advances in biochemical engineering/biotechnology series, pp 231–250
Rauh J, Ph D, Milan F (2011) Bioreactor systems for bone tissue engineering. Tissue Eng Part B Rev 17:263–280
Sladkova M, De Peppo GM (2014) Bioreactor systems for human bone tissue engineering. Processes 2:494–525
Martin I, Wendt D, Heberer M (2004) The role of bioreactors in tissue engineering. Trends Biotechnol 22:10–12
Gaspar DA, Gomide V, Monteiro FJ (2012) The role of perfusion bioreactors in bone tissue engineering the role of perfusion bioreactors in bone tissue engineering. Biomatter 2:1–9
Sikavitsas VI, Bancroft GN, Mikos AG (2002) Formation of three-dimensional cell/polymer constructs for bone tissue engineering in a spinner flask and a rotating wall vessel bioreactor. J Biomed Mater Res 62:136–148
Meinel L, Karageorgiou V, Fajardo R (2004) Bone tissue engineering using human mesenchymal stem cells: effects of scaffold material and medium flow. Ann Biomed Eng 32:112–122
Kim HJ, Kim U, Leisk GG (2007) Bone regeneration on macroporous aqueous-derived silk 3-D scaffolds. Macromol Biosci 7:643–655
Stiehler M, Bunger C, Baatrup A (2008) Effect of dynamic 3-D culture on proliferation, distribution, and osteogenic differentiation of human mesenchymal stem cells. J Biomed Mater Res, Part A 89:96–107
Song K, Liu T, Cui Z (2007) Three-dimensional fabrication of engineered bone with human bio-derived bone scaffolds in a rotating wall vessel bioreactor. J Biomed Mater Res, Part A 86A:323–332
Wang T, Wu H, Wang H (2009) Regulation of adult human mesenchymal stem cells into osteogenic and chondrogenic lineages by different bioreactor systems. J Biomed Mater Res, Part A 88A:935–946
Mccoy RJ, Eng D, Brien FJO, Ph D (2010) Influence of shear stress in perfusion bioreactor cultures for the development of three-dimensional bone tissue constructs: a review. Tissue Eng Part B 16:587–601
Wang Y, Uemura T, Dong J (2003) Application of perfusion culture system improves in vitro and in vivo osteogenesis of bone marrow-derived osteoblastic cells in porous ceramic materials. Tissue Eng 9:1205–1214
Bancroft GN, Sikavitsas VI, Mikos AG (2003) Design of a flow perfusion bioreactor system for bone tissue-engineering applications. Tissue Eng 9:549–554
Bhaskar B, Owen R, Bahmaee H (2017) Design and assessment of a dynamic perfusion bioreactor for large bone tissue engineering scaffolds. Appl Biochem Biotechnol 185:555–563
Bouet G, Marchat D, Cruel M (2015) In vitro three-dimensional bone tissue models: from cells to controlled and dynamic environment. Tissue Eng Part B 21:133–156
Nokhbatolfoghahaei H, Rad MR, Khani M-M (2017) Application of bioreactors to improve functionality of bone tissue engineering constructs: a systematic review. Curr Stem Cell Res Ther 12:564–599
Matziolis D, Tuischer J, Matziolis G (2011) Osteogenic predifferentiation of human bone marrow-derived stem cells by short-term mechanical stimulation. Open Orthop J 5:1–6
Bölgen N, Yang Y, Korkusuz P (2008) Three-dimensional ingrowth of bone cells within biodegradable cryogel scaffolds in bioreactors at different regimes. Tissue Eng Part A 14:1743–1750
Aaron RK, Ciombor DM, Simon BJ (2004) Treatment of nonunions with electric and electromagnetic fields. Clin Orthop Relat Res 02906:21–291
Chalidis B, Sachinis N, Hospital SM, Hospital AG (2011) Stimulation of bone formation and fracture healing with pulsed electromagnetic fields: biologic responses and clinical implications. Int J Immunopathol Pharmacol 24:17–20
Funk RHW, Monsees T, Nurdan O (2009) Electromagnetic effects-from cell biology to medicine. Prog Histochem Cytochem 43:177–264
Sun L, Hsieh D, Lin P (2010) Pulsed electromagnetic fields accelerate proliferation and osteogenic gene expression in human bone marrow mesenchymal stem cells during osteogenic differentiation. Bioelectromagnetics 31:209–219
Tsai M, Chang WH, Chang K (2007) Pulsed electromagnetic fields affect osteoblast proliferation and differentiation in bonetissue engineering. Bioelectromagnetics 28:519–528
Fassina L, Visai L, De Angelis MGC (2007) Surface modification of a porous polyurethane through a culture of human osteoblasts and an electromagnetic bioreactor. Technol Heal Care 15:33–45
Liu C, Abedian R, Meister R (2012) Influence of perfusion and compression on the proliferation and differentiation of bone mesenchymal stromal cells seeded on polyurethane scaffolds. Biomaterials 33:1052–1064
Petri M, Ufer K, Toma I (2012) Effects of perfusion and cyclic compression on in vitro tissue engineered meniscus implants. Knee Surg, Sport Traumatol Arthrosc 20:223–231
Kang KS, Hong JM, Jeong YH (2014) Combined effect of three types of biophysical stimuli for bone regeneration. Tissue Eng Part A 20:1767–1777
Huang R-L, Liu K, Li Q (2016) Bone regeneration following the in vivo bioreactor principle: is in vitro manipulation of exogenous elements still needed? Regen Med 11:475–481
Huang R, Kobayashi E, Liu K, Li Q (2016) Bone graft prefabrication following the in vivo bioreactor principle. EBioMedicine 12:43–54
Tatara AM, Wong ME, Mikos AG (2014) In vivo bioreactors for mandibular reconstruction. J Dent Res 93:1196–1202
Stevens MM, Marini RP, Schaefer D (2005) In vivo engineering of organs: the bone bioreactor. Proc Natl Acad Sci 102:11450–11455
Holt GE, Halpern JL, Dovan TT (2005) Evolution of an in vivo bioreactor. J Orthop Res 23:916–923
Huang R-L, Tremp M, Ho C-K (2017) Prefabrication of a functional bone graft with a pedicled periosteal flap as an in vivo bioreactor. Sci Rep 7:1–11
Zhang H, Mao X, Zhao D (2017) Three dimensional printed polylactic acid-hydroxyapatite composite scaffolds for prefabricating vascularized tissue engineered bone: an in vivo bioreactor model. Sci Rep 7:1–13
Akar B, Tatara AM, Sutradhar A (2018) Large animal models of an in vivo bioreactor for engineering. Tissue Eng 24:317–325
Cheng M-H, Brey EM, Allori AC (2009) Periosteum-guided prefabrication of vascularized bone of clinical shape and volume. Plast Reconstr Surg 124:787–795
Brey EM, Cheng M-H, Allori A (2007) Comparison of guided bone formation from periosteum and muscle fascia. Plast Reconstr Surg 119:1216–1222
Cheng M, Brey EM, Ph D (2005) Ovine model for engineering bone segments. Tissue Eng 11:214–225
Warnke PH, Springer ING, Wiltfang J (2004) Growth and transplantation of a custom vascularised bone graft in a man. Lancet 364:766–770
Wiltfang J, Rohnen M, Egberts A-H (2016) Man as a living bioreactor: prefabrication of a custom vascularized bone graft in the gastrocolic omentum. Tissue Eng Part C Methods 22:740–746
Warnke PH, Kosmahl M, Russo PAJ (2006) Man as living bioreactor: fate of an exogenously prepared customized tissue-engineered mandible. Biomaterials 27:3163–3167
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Thilakan, J., Mishra, R., Goel, S.K., Arya, N. (2019). Engineering of Bone: Uncovering Strategies of Static and Dynamic Environments. In: Bains, P., Sidhu, S., Bahraminasab, M., Prakash, C. (eds) Biomaterials in Orthopaedics and Bone Regeneration . Materials Horizons: From Nature to Nanomaterials. Springer, Singapore. https://doi.org/10.1007/978-981-13-9977-0_12
Download citation
DOI: https://doi.org/10.1007/978-981-13-9977-0_12
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-13-9976-3
Online ISBN: 978-981-13-9977-0
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)