Abstract
Human induced pluripotent stem cells (iPSCs) have highly promising applications in regenerative medicine, drug discovery, and disease modeling, as well as in investigations of developmental biology. Although iPSC can theoretically be differentiated into any cell type, further developments are required for application to tissue replacement. In particular, most differentiation protocols produce fully matured populations of single cell types in two-dimensional cultures.
Some tissues, including retinal pigment epithelium, comprise relatively simple structures of single cell types and can be replaced by monocultures of matured cells. However, gastrointestinal and liver tissues comprise multiple cell types in complex architectures. Therefore, the lack of blood perfusion and three-dimensional interactions between multiple cell types are fundamental obstacles to the application of iPSC to regenerative medicine.
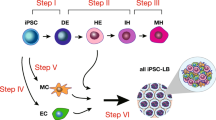
To address these critical technological gaps, we investigated in vivo developmental processes in the context of essential interactions between cell types. In particular, the development of endodermal organs starts with organ specification as monocellular epithelium and subsequent multicellular interactions result in three-dimensional organs. We previously demonstrated the development of three-dimensional functional human liver buds (iPSC-LBs) from iPSC by coculturing iPSC-derived hepatic endodermal cells with endothelial and mesenchymal cells. This “organ bud technology” has high potential for the application of regenerative medicine to complex tissues and provides a unique model system for analyzing complex human tissues that comprise multiple cell types.
In this review, we present the current understanding of iPSC-LB in regenerative medicine and discuss its potential use as a tool for investigating the developmental biology of liver tissues.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Assawachananont J, Mandai M, Okamoto S et al (2014) Transplantation of embryonic and induced pluripotent stem cell-derived 3D retinal sheets into retinal degenerative mice. Stem Cell Reports 2:662–674. doi:10.1016/j.stemcr.2014.03.011
Bhatia SN, Underhill GH, Zaret KS, Fox IJ (2014) STATE OF THE ART REVIEW Cell and tissue engineering for liver disease
Burke Z, Oliver G (2002) Prox1 is an early specific marker for the developing liver and pancreas in the mammalian foregut endoderm. Mech Dev 118:147–155. doi:10.1016/S0925-4773(02)00240-X
Camp JG, Badsha F, Florio M, et al (2015) Human cerebral organoids recapitulate gene expression programs of fetal neocortex development. 1–6. doi: 10.1073/pnas.1520760112
Chi X, Michos O, Shakya R et al (2009) Ret-Dependent Cell Rearrangements in the Wolffian Duct Epithelium initiate ureteric bud morphogenesis. Dev Cell 17:199–209. doi:10.1016/j.devcel.2009.07.013
Collin de l’Hortet A, Takeishi K, Guzman-Lepe J et al (2016) Liver-regenerative transplantation: regrow and reset. Am J Transplant 16:1688–1696. doi:10.1111/ajt.13678
Daley GQ (2012) The promise and perils of stem cell therapeutics. Cell Stem Cell 10:740–749
Eiraku M, Takata N, Ishibashi H et al (2011) Self-organizing optic-cup morphogenesis in three-dimensional culture. Nature 472:51–56. doi:10.1038/nature09941
Franklin V, Khoo PL, Bildsoe H et al (2008) Regionalisation of the endoderm progenitors and morphogenesis of the gut portals of the mouse embryo. Mech Dev 125:587–600. doi:10.1016/j.mod.2008.04.001
Gordillo M, Evans T, Gouon-Evans V (2015) Orchestrating liver development. Development 142:2094–2108. doi:10.1242/dev.114215
Hannoun Z, Steichen C, Dianat N et al (2016) The potential of induced pluripotent stem cell derived hepatocytes. J Hepatol. doi:10.1016/j.jhep.2016.02.025
Harrelson Z, Kaestner KH, Evans SM (2012) Foxa2 mediates critical functions of prechordal plate in patterning and morphogenesis and is cell autonomously required for early ventral endoderm morphogenesis. Biol Open 1:173–181. doi:10.1242/bio.2011040
Hirayama M, Ogawa M, Oshima M et al (2013) Functional lacrimal gland regeneration by transplantation of a bioengineered organ germ. Nat Commun 4:2497. doi:10.1038/ncomms3497
Ichikawa T, Nakazato K, Keller PJ et al (2014) Live imaging and quantitative analysis of gastrulation in mouse embryos using light-sheet microscopy and 3D tracking tools. Nat Protoc 9:575–585. doi:10.1038/nprot.2014.035
Ikonomou L, Kotton DN (2015) Derivation of endodermal progenitors from pluripotent stem cells. J Cell Physiol 230:246–258. doi:10.1002/jcp.24771
Ishikawa M, Sekine K, Okamura A et al (2011) Reconstitution of hepatic tissue architectures from fetal liver cells obtained from a three-dimensional culture with a rotating wall vessel bioreactor. J Biosci Bioeng 111:711–718. doi:10.1016/j.jbiosc.2011.01.019
Ito K, Yanagida A, Okada K et al (2014) Mesenchymal progenitor cells in mouse foetal liver regulate differentiation and proliferation of hepatoblasts. Liver Int 34:1378–1390. doi:10.1111/liv.12387
Kajiwara M, Aoi T, Okita K et al (2012) Donor-dependent variations in hepatic differentiation from human-induced pluripotent stem cells. Proc Natl Acad Sci USA 109:12538–12543. doi:10.1073/pnas.1209979109
Kamiya A, Kinoshita T, Ito Y et al (1999) Fetal liver development requires a paracrine action of oncostatin M through the gp130 signal transducer. EMBO J 18:2127–2136. doi:10.1093/emboj/18.8.2127
Kinoshita T, Sekiguchi T, Xu MJ et al (1999) Hepatic differentiation induced by oncostatin M attenuates fetal liver hematopoiesis. Proc Natl Acad Sci USA 96:7265–7270. doi:10.1073/pnas.96.13.7265
Kopp JL, Grompe M, Sander M (2016) Stem cells versus plasticity in liver and pancreas regeneration. Nat Cell Biol 18:238–245. doi:10.1038/ncb3309
Kuroda T, Yasuda S, Kusakawa S et al (2012) Highly sensitive in vitro methods for detection of residual undifferentiated cells in retinal pigment epithelial cells derived from human iPS cells. PLoS One. doi:10.1371/journal.pone.0037342
Kuroda T, Yasuda S, Sato Y (2014) In vitro detection of residual undifferentiated cells in retinal pigment epithelial cells derived from human induced pluripotent stem cells. Methods Mol Biol 1210:183–192. doi:10.1007/978-1-4939-1435-7_14
Lancaster MA, Knoblich JA (2014) Generation of cerebral organoids from human pluripotent stem cells. Nat Protoc 9:2329–2340. doi: 10.1038/nprot.2014.158
Lancaster MA, Renner M, Martin C et al (2013) Cerebral organoids model human brain development and microcephaly. Nature 501:373–379. doi:10.1038/nature12517
Le lay J, Kaestner KH (2010) The Fox genes in the liver: from organogenesis to functional integration. Physiol Rev 90:1–22. doi:10.1152/physrev.00018.2009
Lee CS, Friedman JR, Fulmer JT, Kaestner KH (2005) The initiation of liver development is dependent on Foxa transcription factors. Nature 435:944–947. doi:10.1038/nature03649
Loh KM, Ang LT, Zhang J et al (2014) Efficient endoderm induction from human pluripotent stem cells by logically directing signals controlling lineage bifurcations. Cell Stem Cell 14:237–252. doi:10.1016/j.stem.2013.12.007
Lokmane L, Haumaitre C, Garcia-Villalba P et al (2008) Crucial role of vHNF1 in vertebrate hepatic specification. Development 135:2777–2786. doi:10.1242/dev.023010
Matsumoto K, Yoshitomi H, Rossant J, Zaret KS (2001) Liver organogenesis promoted by endothelial cells prior to vascular function. Science 294(80):559–563. doi: 10.1126/science.1063889
Miyajima A, Tanaka M, Itoh T (2014) Stem/progenitor cells in liver development, homeostasis, regeneration, and reprogramming. Cell Stem Cell 14:561–574. doi:10.1016/j.stem.2014.04.010
Nagamoto Y, Takayama K, Ohashi K et al (2016) Transplantation of a human iPSC-derived hepatocyte sheet increases survival in mice with acute liver failure. J Hepatol 64:1068–1075. doi:10.1016/j.jhep.2016.01.004
Nakano T, Ando S, Takata N et al (2012) Self-formation of optic cups and storable stratified neural retina from human ESCs. Cell Stem Cell 10:771–785. doi:10.1016/j.stem.2012.05.009
Nicolas C, Wang Y, Luebke-Wheeler J, Nyberg S (2016) Stem cell therapies for treatment of liver disease. Biomedicines 4:2. doi:10.3390/biomedicines4010002
Okabe M, Tsukahara Y, Tanaka M et al (2009) Potential hepatic stem cells reside in EpCAM+ cells of normal and injured mouse liver. Development 136:1951–1960. doi:10.1242/dev.031369
Osafune K (2010) In vitro regeneration of kidney from pluripotent stem cells. Exp. Cell Res. 316:2571–2577
Oshima M, Inoue K, Nakajima K et al (2014) Functional tooth restoration by next-generation bio-hybrid implant as a bio-hybrid artificial organ replacement therapy. Sci Rep 4:1–10. doi:10.1038/srep06044
Ozone C, Suga H, Eiraku M et al (2016) Functional anterior pituitary generated in self-organizing culture of human embryonic stem cells. Nat Commun 7:10351. doi:10.1038/ncomms10351
Sasai Y (2013) Next-generation regenerative medicine: organogenesis from stem cells in 3D culture. Cell Stem Cell 12:520–530. doi:10.1016/j.stem.2013.04.009
Sato T, Vries RG, Snippert HJ et al (2009) Single Lgr5 stem cells build crypt-villus structures in vitro without a mesenchymal niche. Nature 459:262–265. doi:10.1038/nature07935
Sato T, van Es JH, Snippert HJ et al (2011) Paneth cells constitute the niche for Lgr5 stem cells in intestinal crypts. Nature 469:415–418. doi:10.1038/nature09637
Sekine K, Takebe T, Suzuki Y et al (2012) Highly efficient generation of definitive endoderm lineage from human induced pluripotent stem cells. Transplant Proc 44:1127–1129. doi:10.1016/j.transproceed.2012.03.001
Si-Tayeb K, Lemaigre FP, Duncan SA (2010a) Organogenesis and development of the liver. Dev Cell 18:175–189. doi:10.1016/j.devcel.2010.01.011
Si-Tayeb K, Noto FK, Nagaoka M et al (2010b) Highly efficient generation of human hepatocyte-like cells from induced pluripotent stem cells. Hepatology 51:297–305. doi:10.1002/hep.23354
Sosa-Pineda B, Wigle JT, Oliver G (2000) Hepatocyte migration during liver development requires Prox1. Nat Genet 25:254–255. doi:10.1038/76996
Spence JR, Mayhew CN, Rankin SA et al (2010) Directed differentiation of human pluripotent stem cells into intestinal tissue in vitro. Nature 470:105–109. doi:10.1038/nature09691
Spence JR, Mayhew CN, Rankin SA et al (2011) Directed differentiation of human pluripotent stem cells into intestinal tissue in vitro. Nature 470:105–109. doi:10.1038/nature09691
Sudo R (2014) Multiscale tissue engineering for liver reconstruction. Organogenesis 10:1–9. doi:10.4161/org.27968
Suga H, Kadoshima T, Minaguchi M et al (2011) Self-formation of functional adenohypophysis in three-dimensional culture. Nature 480:57–62. doi:10.1038/nature10637
Suzuki K, Tanaka M, Watanabe N et al (2008) p75 Neurotrophin receptor is a marker for precursors of stellate cells and portal fibroblasts in mouse fetal liver. Gastroenterology. doi:10.1053/j.gastro.2008.03.075
Takagi R, Ishimaru J, Sugawara A et al (2016) Bioengineering a 3D integumentary organ system from iPS cells using an in vivo transplantation model. Sci Adv 2:e1500887–e1500887. doi:10.1126/sciadv.1500887
Takasato M, Er PX, Becroft M et al (2014) Directing human embryonic stem cell differentiation towards a renal lineage generates a self-organizing kidney. Nat Cell Biol 16:118–126. doi:10.1038/ncb2894
Takasato M, Er PX, Chiu HS et al (2015) Kidney organoids from human iPS cells contain multiple lineages and model human nephrogenesis. Nature 526:564–568. doi:10.1038/nature15695
Takebe T, Koike N, Sekine K et al (2012) Generation of functional human vascular network. Transplant Proc 44:1130–1133. doi:10.1016/j.transproceed.2012.03.039
Takebe T, Sekine K, Enomura M et al (2013) Vascularized and functional human liver from an iPSC-derived organ bud transplant. Nature 499:481–484. doi:10.1038/nature12271
Takebe T, Zhang R-R, Koike H et al (2014) Generation of a vascularized and functional human liver from an iPSC-derived organ bud transplant. Nat Protoc 9:396–409. doi:10.1038/nprot.2014.020
Takebe T, Enomura M, Yoshizawa E et al (2015) Vascularized and complex organ buds from diverse tissues via mesenchymal cell-driven condensation. Cell Stem Cell 16:556–565. doi:10.1016/j.stem.2015.03.004
Tanimizu N, Miyajima A, Mostov KE (2007) Liver progenitor cells develop CholangiocytetType epithelial polarity in three-dimensional culture. Mol Biol Cell 18:986–994. doi:10.1091/mbc.E06
Toyohara T, Mae S, Sueta S et al (2015) Cell therapy using human induced pluripotent stem cell-derived renal progenitors Ameliorates acute kidney injury in mice. Stem Cells Transl Med 4:980–992. doi:10.5966/sctm.2014-0219
Toyoshima K, Asakawa K, Ishibashi N et al (2012) Fully functional hair follicle regeneration through the rearrangement of stem cells and their niches. Nat Commun 3:784. doi:10.1038/ncomms1784
Tremblay KD, KSZ T (2005) Distinct populations of endoderm cells converge to generate the embryonic liver bud and ventral foregut tissues. 280:87–99. doi: 10.1016/j.ydbio.2005.01.003
Uygun BE, Soto-Gutierrez A, Yagi H et al (2010) Organ reengineering through development of a transplantable recellularized liver graft using decellularized liver matrix. Nat Med 16:814–820. doi:10.1038/nm.2170
Watson CL, Mahe MM, Múnera J et al (2014) An in vivo model of human small intestine using pluripotent stem cells. doi: 10.1038/nm.3737
Yui S, Nakamura T, Sato T et al (2012) Functional engraftment of colon epithelium expanded in vitro from a single adult Lgr5+ stem cell. Nat Med 18:618–623. doi:10.1038/nm.2695
Zaret KS, Grompe M (2008) Generation and regeneration of cells of the liver and pancreas. Science 322:1490–1494. doi:10.1126/science.1161431
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Sekine, K., Takebe, T., Taniguchi, H. (2017). Liver Regeneration Using Cultured Liver Bud. In: Tsuji, T. (eds) Organ Regeneration Based on Developmental Biology. Springer, Singapore. https://doi.org/10.1007/978-981-10-3768-9_12
Download citation
DOI: https://doi.org/10.1007/978-981-10-3768-9_12
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-10-3766-5
Online ISBN: 978-981-10-3768-9
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)