Abstract
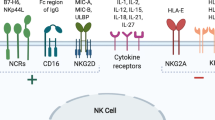
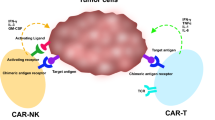
Human pluripotent stems cells provide an ideal source for the study of hematopoietic differentiation. Natural killer (NK) cells are lymphocytes that play a key role in innate immunity against viral infections as well as malignancies. The development and differentiation of NK cells have been an area of increasing research interest due to their clinical utility in treating multiple types of cancer and potentially infectious disease. Our initial studies to derive NK cells from human embryonic stem cells (hESCs) and induced pluripotent stem cells (iPSCs) used a stromal cell co-culture method with relatively poor-defined conditions. Subsequent studies have utilized a stroma-free embryoid body (EB) method to generate hemato-endothelial precursor cells followed by in vitro NK cell differentiation in defined conditions. Further expansion of these hESC- and iPSC-derived NK cells can be done through the use of interleukin (IL)-21 expressing artificial antigen-presenting cells (aAPCs). Combining these methods, we can efficiently generate enough NK cells required for clinical therapies from a small number of undifferentiated human pluripotent stem cells. These methods enable hESCs and iPSCs to be used to produce an essentially unlimited amount of homogenous NK cells that can be used as a standardized, off-the-shelf immunotherapy for the treatment of refractory cancers and other diseases.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Ljunggren HG, Malmberg KJ. Prospects for the use of NK cells in immunotherapy of human cancer. Nat Rev Immunol. 2007;7:329–39.
Miller JS, Soignier Y, Panoskaltsis-Mortari A, et al. Successful adoptive transfer and in vivo expansion of human haploidentical NK cells in patients with cancer. Blood. 2005;105:3051–7.
Geller MA, Cooley S, Judson PL, et al. A phase II study of allogeneic natural killer cell therapy to treat patients with recurrent ovarian and breast cancer. Cytotherapy. 2011;13:98–107.
Miller JS, Alley KA, McGlave P. Differentiation of natural killer (NK) cells from human primitive marrow progenitors in a stroma-based long-term culture system: identification of a CD34 + 7 + NK progenitor. Blood. 1994;83:2594–601.
Miller JS, McCullar V, Verfaillie CM. Ex vivo culture of CD34 +/Lin-/DR- cells in stroma-derived soluble factors, interleukin-3, and macrophage inflammatory protein-1alpha maintains not only myeloid but also lymphoid progenitors in a novel switch culture assay. Blood. 1998;91:4516–22.
Silva MR, Kessler S, Ascensao JL. Hematopoietic origin of human natural killer (NK) cells: generation from immature progenitors. Pathobiology. 1993;61:247–55.
Mrózek E, Anderson P, Caligiuri MA. Role of interleukin-15 in the development of human CD56 + natural killer cells from CD34 + hematopoietic progenitor cells. Blood. 1996;87:2632–40.
Sivori S, Cantoni C, Parolini S, et al. IL-21 induces both rapid maturation of human CD34 + cell precursors towards NK cells and acquisition of surface killer Ig-like receptors. Eur J Immunol. 2003;33:3439–47.
Perez SA, Mahaira LG, Sotiropoulou PA, et al. Effect of IL-21 on NK cells derived from different umbilical cord blood populations. Int Immunol. 2006;18:49–58.
Wilber A, Linehan JL, Tian X, et al. Efficient and stable transgene expression in human embryonic stem cells using transposon-mediated gene transfer. Stem Cells. 2007;25:2919–27.
Giudice A, Trounson A. Genetic modification of human embryonic stem cells for derivation of target cells. Cell Stem Cell. 2008;2:422–33.
Woll PS, Martin CH, Miller JS, Kaufman DS. Human embryonic stem cell-derived NK cells acquire functional receptors and cytolytic activity. J Immunol. 2005;175:5095–103.
Ni Z, Knorr DA, Clouser CL, et al. Human pluripotent stem cells produce natural killer cells that mediate anti-HIV-1 activity by utilizing diverse cellular mechanisms. J Virol. 2011;85:43–50.
Woll PS, Grzywacz B, Tian X, et al. Human embryonic stem cells differentiate into a homogeneous population of natural killer cells with potent in vivo antitumor activity. Blood. 2009;113:6094–101.
Ng ES, Davis R, Stanley EG, Elefanty AG. A protocol describing the use of a recombinant protein-based, animal product-free medium (APEL) for human embryonic stem cell differentiation as spin embryoid bodies. Nat Protoc. 2008;3:768–76.
Ng ES, Davis RP, Azzola L, Stanley EG, Elefanty AG. Forced aggregation of defined numbers of human embryonic stem cells into embryoid bodies fosters robust, reproducible hematopoietic differentiation. Blood. 2005;106:1601–3.
Kaufman DS, Hanson ET, Lewis RL, Auerbach R, Thomson JA. Hematopoietic colony-forming cells derived from human embryonic stem cells. Proc Natl Acad Sci USA. 2001;98:10716–21.
Oostendorp RA, Robin C, Steinhoff C, et al. Long-term maintenance of hematopoietic stem cells does not require contact with embryo-derived stromal cells in cocultures. Stem Cells. 2005;23:842–51.
Ledran MH, Krassowska A, Armstrong L, et al. Efficient hematopoietic differentiation of human embryonic stem cells on stromal cells derived from hematopoietic niches. Cell Stem Cell. 2008;3:85–98.
Denman CJ, Senyukov VV, Somanchi SS, et al. Membrane-bound IL-21 promotes sustained ex vivo proliferation of human natural killer cells. PLoS ONE. 2012;7:e30264.
Ng ES, Davis RP, Hatzistavrou T, Stanley EG, Elefanty AG. Directed differentiation of human embryonic stem cells as spin embryoid bodies and a description of the hematopoietic blast colony forming assay. Curr Protoc Stem Cell Biol Chapter. 2008;1: Unit 1D 3.
Hexum MK, Tian X, Kaufman DS. In vivo evaluation of putative hematopoietic stem cells derived from human pluripotent stem cells. Methods Mol Biol. 2011;767:433–47.
Knorr DA, Ni Z, Hermanson D, et al. Clinical-scale derivation of natural killer cells from human pluripotent stem cells for cancer therapy. Stem Cells Transl Med. 2013;2:274–83.
Acknowledgments
Support for studies of NK cell development have come from National Institutes of Health/NHLBI, the University of Minnesota Masonic Cancer Center, the William Lawrence & Blanche Hughes Foundation, the State of Minnesota Partnership for Biotechnology and Medical Genomics, the Minnesota Ovarian Cancer Alliance, and the International Clinical Research Center.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 The Author(s)
About this chapter
Cite this chapter
Hermanson, D.L., Ni, Z., Kaufman, D.S. (2015). Human Pluripotent Stem Cells as a Renewable Source of Natural Killer Cells. In: Cheng, T. (eds) Hematopoietic Differentiation of Human Pluripotent Stem Cells. SpringerBriefs in Stem Cells, vol 6. Springer, Dordrecht. https://doi.org/10.1007/978-94-017-7312-6_5
Download citation
DOI: https://doi.org/10.1007/978-94-017-7312-6_5
Published:
Publisher Name: Springer, Dordrecht
Print ISBN: 978-94-017-7311-9
Online ISBN: 978-94-017-7312-6
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)