Abstract
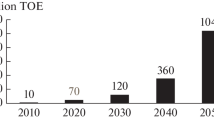
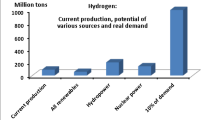
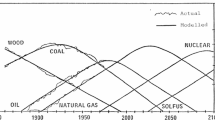
The idea of an economy supported by hydrogen is still being considered by government bodies and major oil companies. The extensive use of hydrogen as a fuel has many applications in pollution-free technologies which could be of every-day use in society. Examples are in transportation and power generation; other uses include chemical technology, metallurgy, effluent processing and water production. For mass application the most appropriate process for hydrogen production is water electrolysis. To this end, a simplified description of the hydrogen and oxygen evolution mechanisms is presented. Understanding these mechanisms will provide a basis for making water electrolysis more efficient. For this reason special attention is given to the design of catalysts for water electrolysis.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Bockris, J.O.M.: The Solar-hydrogen Alternative. Architectural Press, London (1975)
Meadows, D.H., Meadows, D.L., Randers, J., Behrens, W.W.: The Limits to Growth. Universe Books, New York (1972)
Hubbert, M.K.: Resources and Man. W.H Freeman, San Francisco (1969)
The Hydrogen Economy.: The National Academies Press, Washington, D.C (2004)
Colton, W.M.: The Outlook for Energy a view to 2030. Exxon Mobil Corporation (2011)
Voser, P.: Shell Energy Scenarios to 2050—Signals and Signposts. Royal Dutch Shell plc (2011)
Grundy, P.: Shell Energy Scenarios to 2050—Energy. Royal Dutch Shell plc (2011)
Dudley, B.: BP Energy Outlook 2030. BP plc (2011)
Lide, D.R.: The Elements. CRC Handbook of Chemistry and Physics. 86th edn. CRC Press, New York (2005)
Needham, S.: The Potential for Renewable Energy to Provide Baseload Power in Australia. Parliament of Australia, Commonwealth of Australia (2011)
El-Moneim, A.A., Bhattarai, J., Kato, Z., Izumiya, K., Kumagai, N., Hashimoto, K.: Mn-Mo-Sn oxide anodes for oxygen evolution in seawater electrolysis for hydrogen production. ECS Trans. 25, 127–137 (2009)
Bockris, J.O.M., Reddy, A.K.N., Gamboa-Aldeco, M.: Modern Electrochemistry. vol. 2A. 2nd edn. Kluwer Academic/Plenum Publishers, New York (2000)
Millet, P., Ngameni, R., Grigoriev, S.A., Mbemba, N., Brisset, F., Ranjbari, A., Etievant, C.: PEM water electrolyzers: from electrocatalysis to stack development. Int. J. Hydrogen Energy 35, 5043–5052 (2010)
Jackson, T.: XI.13 High-efficiency, ultra-high pressure electrolysis with direct linkage to photovoltaic arrays (Phase II Project) (2005)
Dresselhaus, M.: Basic research needs for the hydrogen economy (2003)
Brown, D.E., Mahmood, M.N., Man, M.C.M., Turner, A.K.: Preparation and characterization of low overvoltage transition metal alloy electrocatalysts for hydrogen evolution in alkaline solutions. Electrochim. Acta 29, 1551–1556 (1984)
Hashimoto, K., Sasaki, T., Meguro, S., Asami, K.: Nanocrystalline electrodeposited Ni-Mo-C cathodes for hydrogen production. Mat. Sci. Eng. A-Struct. 375–377, 942–945 (2004)
Chen, L., Lasia, A.: Study of the kinetics of hydrogen evolution reaction on nickel-zinc alloy electrodes. J. Electrochem. Soc. 138, 3321–3328 (1991)
Vandenborre, H., Vermeiren, P., Leysen, R.: Hydrogen evolution at nickel sulphide cathodes in alkaline medium. Electrochim. Acta 29, 297–301 (1984)
Yamashita, H., Yamamura, T., Yoshimoto, K.: The relation between catalytic ability for hydrogen evolution reaction and characteristics of nickel-tin alloys. J. Electrochem. Soc. 140, 2238–2243 (1993)
Conway, B.E., Liu, T.-C.: Characterization of electrocatalysis in the oxygen evolution reaction at platinum by evaluation of behavior of surface intermediates states at the oxide film. Langmuir 6, 268–276 (1990)
Birss, V.I.: Oxygen evolution at platinum electrodes in alkaline solutions. J. Electrochem. Soc. 133, 1621–1625 (1986)
Hansen, H.A., Man, I.C., Studt, F., Abild-Pedersen, F., Rossmeisl, J.: Electrochemical chlorine evolution at rutile oxide (110) surfaces. Phys. Chem. Chem. Phys. 12, 283–290 (2010)
Rossmeisl, J., Qu, Z.-W., Zhu, H., Kroes, G.-J., Norskov, J.K.: Electrolysis of water on oxides surfaces. J. Electroanal. Chem. 607, 83–89 (2007)
Matsumoto, Y., Sato, E.: Electrocatalytic properties of transition metal oxides for oxygen evolution reaction. Mater. Chem. Phys. 14, 397–426 (1986)
Bockris, J.O.M.: Kinetics of activation-controlled consecutive electrochemical reactions: anodic evolution of oxygen. J. Chem. Phys. 1956, 817–827 (1956)
Krasilshchikov, A.I.: Intermediate stages of anodic oxygen evolution. Russ. J. Phys. Chem. 37, 273 (1963)
Wade, W.H., Hackerman, N.: Anodic phenomena at an iron electrode. Trans. Faraday Soc. 53, 1636–1647 (1957)
O’Grady, W., Iwakura, C., Huang, J., Yeager, E.: Ruthenium oxide catalysts for the oxygen electrode. In: Breiter, M.W. (ed.) Proceedings of Symposium Electrocatalyst, pp. 286–302 (1974)
O’Grady, W.E., Iwakura, C., Yeager, E.: Oxygen electrocatalysts for life support systems. Am. Soc. Mech. Eng. (76-ENAs-37), 11 (1976)
Bockris, J.O.M., Otagawa, T.: The electrocatalysis of oxygen evolution on perovskites. J. Electrochem. Soc. 131, 290–302 (1984)
Kelly, E.J., Heatherly, D.E., Vallet, C.E., White, C.W.: Application of ion implantation to the study of electrocatalysis. I. Chlorine evolution at ruthenium-implanted titanium electrodes. J. Electrochem. Soc. 134, 1667–1675 (1987)
Trasatti, S., O’Gradey, W.E.: Properties and applications of ruthenium dioxide-based electrodes. Adv. Electroch. El. Eng. 1981, 177–261 (1981)
Trasatti, S.: Studies in physical and theoretical chemistry. vol. 11. Electrodes of conductive metallic oxides, Pt. A (1980)
Krishtalik, L.I.: Kinetics and mechanism of anodic chlorine and oxygen evolution reactions on transition metal oxide electrodes. Electrochim. Acta 26, 329–337 (1981)
Hepel, T., Pollak, F.H., O’Grady, W.E.: Chlorine evolution and reduction processes at oriented single-crystal ruthenium dioxide electrodes. J. Electrochem. Soc. 133, 69–75 (1986)
Kuznetsova, E., Krtil, P., Heyrovsky, J.: Parallel oxygen and chlorine evolution on iridium based oxides. Paper presented at the electrocatalysis: Present and future and ELCAT meeting, Alicante, Spain, 14–17 (2011)
Ghany, N.A.A., Kumagai, N., Meguro, S., Asami, K., Hashimoto, K.: Oxygen evolution anodes composed of anodically deposited Mn-Mo-Fe oxides for seawater electrolysis. Electrochim. Acta 48, 21–28 (2002)
Vondrak, J., Klapste, B., Velicka, J., Sedlarikova, M., Reiter, J., Roche, I., Chainet, E., Fauvarque, J.F., Chatenet, M.: Electrochemical activity of manganese oxide/carbon-based electrocatalysts. J. New Mat. Elect. Syst. 8, 209–212 (2005)
Gatt, P., Petrie, S., Stranger, R., Pace, R.J.: Rationalizing the 1.9 A crystal structure of photosystem II—a remarkable Jahn-Teller balancing act induced by a single proton transfer. Angew. Chem. Int. Ed. 51, 1–5 (2012)
Guizard, C., Princivalle, A.: Preparation and characterization of catalyst thin films. Catal. Today 146, 367–377 (2009)
Djokic, S.S.: Modern Aspects of Electrochemistry, vol. 48. Springer, New York (2010)
Chen, Y.-C., Hsu, Y.-K., Lin, Y.-G., Lin, Y.-K., Horng, Y–.Y., Chen, L.-C., Chen, K.-H.: Highly flexible supercapacitors with manganese oxide nanoshhet/carbon cloth electrode. Electrochim. Acta 56, 7124–7130 (2011)
Kawashima, A., Sakaki, T., Habazaki, H., Hashimoto, K.: Ni-Mo-O alloy cathodes for hydrogen evolution in hot concentrated NaOH solution. Mat. Chem. Eng. A 267, 246–253 (1999)
Choquette, Y., Menard, H., Brossard, L.: Hydrogen discharge on a Raney nickel composite-coated electrode. Int. J. Hydrogen Energy 14, 637–642 (1989)
Raush, S., Wendt, H.: Morphology and utilization of smooth hydrogen-evolving Raney nickel cathode coatings and porous sintered-nickel cathodes. J. Electrochem. Soc. 143, 2852–2862 (1996)
Endoh, E., Otouma, H., Morimoto, T., Oda, Y.: New Raney nickel composite-coated electrode for hydrogen evolution. Int. J. Hydrogen Energy 12, 473–479 (1987)
Paunovic, P.: Toward sustainable hydrogen economy: electrode materials-non-platinum electrocatalytic systems. J. Univ. Chem. Tech. Metall. 45, 149–160 (2010)
Jaksic, M.M., Lacnjevac, C.M., Grgur, B.N., Krstajic, N.V.: Volcano plots along intermetallic hypo-hyper-d-electronic phase diagrams and electrocatalysis for hydrogen electrode reactions. J. New Mat. Elect. Syst. 3, 169–182 (2000)
Paunovic, P., Popovski, O., Dimitrov, A., Slavkov, D., Lefterova, E., Jordanov, Sh: Improvement of performances of complex non-platinum electrode materials for hydrogen evolution. Electrochim. Acta 52, 1810–1817 (2006)
Trasatti, S., Buzzanca, G.: Ruthenium dioxide: a new interesting electrode material. Solid state structure and electrochemical behavior. J. Electroanal. Chem. 29, 1–2 (1971)
Thompson, R.: The Modern Inorganic Chemicals Industry, vol. 31. London (1977)
Acknowledgments
The authors would like to thank AINSE Ltd for providing financial assistance (Award No ALNGRA12020/10366 and AINSE Post Graduate Research Award 10595) to enable work on the catalyst surfaces. We would also like to acknowledge the technological support from ANSTO (Australian Nuclear Science and Technology Organization) and the grant provided by Australian Synchrotron (grant AS123/HRIR 5428A) for use of far infrared beamline.
The global R&D Centers Program of NRF (National Research Foundation of Korea), funded by MSIP (Ministry of Science, ICT & Future Planning) at KIGAM (Korean Institute of Geoscience and Mineral Resources) for instrument time. Special thanks to Dr. Danielle Meyrick, Dr. Justin McGinnity, Dr. Trevor Pryor, Dr. Kim D. J., Dr. Chung K. W., Dr. Mihail Ionescu, Dr. Gamini Senanayake and Sue Farr for their suggestions and support.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2013 Springer-Verlag Berlin Heidelberg
About this chapter
Cite this chapter
Delgado, D., Hefter, G., Minakshi, M. (2013). Hydrogen Generation. In: Ferreira, G. (eds) Alternative Energies. Advanced Structured Materials, vol 34. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-40680-5_7
Download citation
DOI: https://doi.org/10.1007/978-3-642-40680-5_7
Published:
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-642-40679-9
Online ISBN: 978-3-642-40680-5
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)