Abstract
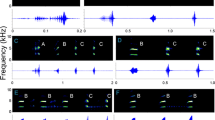
In the katydid genus Neoconocephalus, males typically produce continuous calls with an extremely fast pulse rate of about 200/s. Divergence from this ancestral pattern includes alternation of pulse periods resulting in a double-pulse pattern, and the grouping of pulses into chirps. Double-pulse patterns evolved five times independently in the genus. Analysis of the female preferences and call recognition mechanisms revealed that in three species with double-pulse pattern, females have independently evolved new mechanisms for recognizing the derived call pattern. In the remaining two species with double-pulse pattern, females retain the ancestral recognition mechanism and exhibit no preference for the derived temporal pattern. These results suggest that males are leading the evolutionary divergence of call patterns in this genus. We propose a hypothetical scenario in which genetic bottlenecks and founder effects arising from the climatic history of North America contributed to the rapid diversification of calls in this genus.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Adams J (1997) Preliminary vegetation maps of the world since the last glacial maximum: an aid to archaeological understanding. J of Archaeol Sci 24:623–647
Andersson M (2006) Condition-dependent indicators in sexual selection: development of theory and tests. In: Lucas JR, Simmons LW (eds) Essays in animal behaviour: celebrating 50 years of animal behaviour. Elsevier, Amsterdam, pp 253–267
Andersson M, Simmons LW (2006) Sexual selection and mate choice. Trends Ecol Evol (Amst) 21:296–302. doi:10.1016/j.tree.2006.03.015
Beckers OM, Schul J (2008) Developmental plasticity of mating calls enables acoustic communication in diverse environments. Proc R Soc Lond B 275:1243–1248
Bloake CRB (1991) Coevolution of sender and receiver of sexual signals: genetic coupling and genetic correlations. Trends Ecol Evol 6:225–227. doi:10.1016/0169-5347(91)90027-U
Brower AVZ (1994) Rapid morphological radiation and convergence among races of the butterfly Heliconius erato inferred from patterns of mitochondrial DNA evolution. Proc Natl Acad Sci USA 91:6491–6495
Burk TE (1982) Evolutionary significance of predation on sexually signaling males. Fla Entomol 65:90–104
Bush SL, Schul J (2006) Pulse-rate recognition in an insect: evidence of a role for oscillatory neurons. J Comp Physiol A 192:113–121. doi:10.1007/s00359-005-0053-x
Bush SL, Schul J (2010) Evolution of novel signal traits in the absence of female preferences in Neoconocephalus katydids (Orthoptera, Tettigoniidae). PLoS ONE 5(8):e12457
Bush SL, Beckers OM, Schul J (2009) A complex mechanism of call recognition in the katydid Neoconocephalus affinis (Orthoptera: Tettigoniidae). J Exp Biol 212:648–655. doi:10.1242/jeb.024786
Butlin R, Ritchie MG (1989) Genetic coupling in mate recognition systems: what is the evidence. Biol J Linn Soc 37:237–246. doi:10.1111/j.1095-8312.1989.tb01902.x
Büttner UK (2002) Charakterisierung der Gesänge von fünf in Missouri (USA) heimischen Neoconocephalus-Arten (Orthoptera, Tettigoniidae). Diplom Thesis, Friedrich Alexander University, Erlangen
Deily JA, Schul J (2004) Recognition of calls with exceptionally fast pulse rates: Female phonotaxis in the genus Neoconocephalus (Orthoptera: Tettigoniidae). J Exp Biol 207:3523–3529
Deily JA, Schul J (2006) Spectral selectivity during phonotaxis: a comparative study in Neoconocephalus (Orthoptera, Tettigoniidae). J Exp Biol 209:1757–1764
Deily JA, Schul J (2009) Selective phonotaxis in Neoconocephalus nebrascensis (Orthoptera: Tettigoniidae): call recognition at two temporal scales. J Comp Physiol A 195:31–37
Drummond AJ, Rambaut A (2007) BEAST: bayesian evolutionary analysis by sampling trees. BMC Evol Biol 7:214
Drummond AJ, Ho SYW, Phillips MJ, Rambaut A (2006) Relaxed phylogenetics and dating with confidence. PLoS Biol 4:e88
Ellison CK, Wiley C, Shaw KL (2011) The genetics of speciation: genes of small effect underlie sexual isolation in the Hawaiian cricket Laupala. J Evolution Biol 24:1110–1119. doi:10.1111/j.1420-9101.2011.02244.x
Grace JL, Shaw KL (2011) Coevolution of male mating signal and female preference during early lineage divergence ofthe Hawaiian cricket, Laupala cerasina. Evolution 65:2184–2196. doi:10.1111/j.1558-5646.2011.01278.x
Greenfield MD (1983) Unsynchronized chorusing in the coneheaded katydid Neoconocephalus affinis (Beauvois). Anim Behav 31:102–112
Greenfield MD (1990) Evolution of acoustic communication in the genus Neoconocephalus: discontinuous songs, synchrony, and interspecific interaction. In: Bailey W, Rentz D (eds) The Tettigoniidae: biology, systematics, and evolution. Springer, New York, pp 72–97
Greenfield MD (1993) Inhibition of male calling by heterospecific signals: artifact of chorusing or abstinence during suppression of female phonotaxis? Naturwissenschaften 80:570–573
Greenfield MD (2005) Mechanisms and evolution of communal sexual displays in arthropods and anurans. Adv Study Behav 35:1–62
Greenfield MD, Roizen I (1993) Katydid synchronous chorusing is an evolutionarily stable outcome of female choice. Nature 364:618–620
Greenfield MD, Schul J (2008) Mechanisms and evolution of synchronous chorusing: emergent properties and adaptive functions in Neoconocephalus katydids (Orthoptera: Tettigoniidae). J Comp Psychology 122:289–297
Heller KG (1988) Bioakustik der Europäischen Laubheuschrecken. Margraf, Weikersheim
Ov Helversen, Dv Helversen (1994) Forces driving coevolution of song and song recognition in grasshoppers. In: Schildberger K, Elsner N (eds) Neural basis of behavioural adaptations. Fischer, Stuttgart, pp 253–284
Izhikevich EM (2001) Resonate-and-fire neurons. Neural Netw 14:883–894
Lloyd JE (1984) Evolution of a firefly flash code. Florida Entomol 67:228–239
McDermott JH, Oxenham AJ (2008) Music perception, pitch, and the auditory system. Curr Opin Neurobiol 18:452–463. doi:10.1016/j.conb.2008.09.005
Mendelson T, Shaw K (2005) Rapid speciation in an arthropod. Nature 433:375–376. doi:10.1038/433375a
Meixner AJ, Shaw KC (1986) Acoustic and associated behavior of the coneheaded katydid Neoconocephalus nebrascensis (Orthoptera: Tettigoniidae). Annals Entomol Soc Am 79:554–565
Sanderson MJ (2003) r8 s: inferring absolute rates of molecular evolution and divergence times in the absence of a molecular clock. Bioinformatics 19:301–302. doi:10.1093/bioinformatics/19.2.301
Schmidt A, Ronacher B, Hennig RM (2008) The role of frequency, phase and time for processing of amplitude modulated signals by grasshoppers. J Comp Physiol A 194:221–233. doi:10.1007/s00359-007-0295-x
Schul J, Bush SL (2002) Non-parallel coevolution of sender and receiver in the acoustic communication system of treefrogs. Proc R Soc Lond B 269:1847–1852
Schul J, Patterson AC (2003) What determines the tuning of hearing organs and the frequency of calls? a comparative study in the katydid genus Neoconocephalus (Orthoptera, Tettigoniidae). J Exp Biol 206:141–152
Shaw KL, Herlihy DP (2000) Acoustic preference functions and song variability in the Hawaiian cricket Laupala cerasina. Proc Biol Sci 267:577–584. doi:10.1098/rspb.2000.1040
Shaw KL, Lesnick SC (2009) Genomic linkage of male song and female acoustic preference QTL underlying a rapid species radiation. P Natl Acad Sci Usa 106:9737–9742. doi:10.1073/pnas.0900229106
Snyder RL, Frederick-Hudson KH, Schul J (2009) Molecular phylogenetics of the genus Neoconocephalus (Orthoptera, Tettigoniidae) and the evolution of temperate life histories. PLoS ONE 4:e7203. doi:10.1371/journal.pone.0007203.t001
Talwar M (2007) Function and evolution of the call spectrum in the katydid genus Neoconocephalus. Dissertation, University of Missouri, Columbia
Thomas ES, Alexander RD (1962) Systematics and behavioral studies on the meadow grasshoppers of the Orchelimum concinnum group (Orthoptera: Tettigoniidae). Occas Pap Mus Zool Univ Mich 626:1–31
Walker TJ (1975) Stridulatory movements of eight species of Neoconocephalus (Tettigoniidae). J Insect Physiol 21:595–603
Walker TJ (2012) The singing insects of North America—Katydids. http://buzz.ifas.ufl.edu/crickets.htm. Cited 1 June 2012
Walker T, Greenfield M (1983) Songs and systematics of Caribbean Neoconocephalus (Tettigoniidae, Orthoptera). Trans Amer Ent Soc 109:357–389
Wiley C, Ellison CK, Shaw KL (2012) Widespread genetic linkage of mating signals and preferences in the Hawaiian cricket Laupala. P Roy Soc B-Biol Sci 279:1203–1209. doi:10.1098/rspb.2011.1740
Wiley C, Shaw KL (2010) Multiple genetic linkages between female preference and male signal in rapidly speciating Hawaiian crickets. Evolution 64:2238–2245. doi:10.1111/j.1558-5646.2010.01007.x
Zuk M, Kolluru GR (1998) Exploitation of sexual signals by predators and parasitoids. Q Rev Biol 73:415–438
Acknowledgments
We thank the numerous members of our lab for their contributions and support. Oli Beckers, Josh Deily, and Rob Snyder each contributed significantly to this research. Tom Walker and Michael Greenfield introduced JS and SLB to many aspects of Neoconocephalus natural history and helped locate many species for these studies. We would like to thank Peter Heinecke for his help in assembling and maintaining our walking compensator (“Kramer Kugel”), which has been instrumental for this work.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer-Verlag Berlin Heidelberg
About this chapter
Cite this chapter
Schul, J., Bush, S.L., Frederick, K.H. (2014). Evolution of Call Patterns and Pattern Recognition Mechanisms in Neoconocephalus Katydids. In: Hedwig, B. (eds) Insect Hearing and Acoustic Communication. Animal Signals and Communication, vol 1. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-40462-7_10
Download citation
DOI: https://doi.org/10.1007/978-3-642-40462-7_10
Published:
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-642-40461-0
Online ISBN: 978-3-642-40462-7
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)