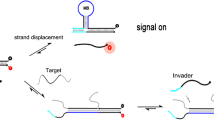
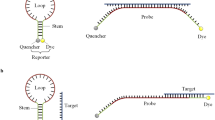
Abstract
Molecular beacons are widely used for detection of nucleic acids both in vitro and in vivo. Compared with linear probes, molecular beacons have shown enhanced sensitivity and specificity primarily due to their stem–loop hairpin structures. The hairpin structures bring new considerations on thermodynamics and kinetics for designing of nucleic acid probes. This chapter has been designed to provide a better understanding of structure–performance relationship of molecular beacons based on analysis of their thermodynamic and kinetic properties. The conformational fluctuations of molecular beacons are discussed concerning the stability and kinetics of the hairpin-coil transformation. In the presence of target nucleic acids, molecular beacons hybridize with targets to form duplex complexes. We analyzed the theoretical models and the relevant parameters used to describe the hybridization reactions. Furthermore, studies on strategies for optimization of molecular beacon performance are summarized. The systematic analysis of studies about thermodynamic and kinetic properties of molecular beacons allows for sophisticated design of better molecular beacons for specific purposes.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Tyagi S, Kramer FR (1996) Molecular beacons: probes that fluoresce upon hybridization. Nat Biotechnol 14:303–308
Tsourkas A, Behlke MA, Rose SD, Bao G (2003) Hybridization kinetics and thermodynamics of molecular beacons. Nucleic Acids Res 31:1319–1330
Bonnet G, Krichevsky O, Libchaber A (1998) Kinetics of conformational fluctuations in DNA hairpin-loops. Proc Natl Acad Sci U S A 95:8602–8606
Vallone PM, Benight AS (1999) Melting studies of short DNA hairpins containing the universal base 5-nitroindole. Nucleic Acids Res 27:3589–3596
Antao VP, Tinoco I (1992) Thermodynamic parameters for loop formation in RNA and DNA hairpin tetraloops. Nucleic Acids Res 20:819–824
Vallone PM, Paner TM, Hilario J, Lane MJ, Faldasz BD, Benight AS (1999) Melting studies of short DNA hairpins: influence of loop sequence and adjoining base pair identity on hairpin thermodynamic stability. Biopolymers 50:425–442
Rentzeperis D, Shikiya R, Maiti S, Ho J, Marky LA (2002) Folding of intramolecular DNA hairpin loops: enthalpy-entropy compensations and hydration contributions. J Phys Chem B 106:9945–9950
Breslauer KJ, Frank R, Blöcker H, Marky LA (1986) Predicting DNA duplex stability from the base sequence. Proc Natl Acad Sci U S A 83:3746–3750
SantaLucia J (1998) A unified view of polymer, dumbbell, and oligonucleotide DNA nearest-neighbor thermodynamics. Proc Natl Acad Sci U S A 95:1460–1465
Owczarzy R, Vallone PM, Gallo FJ, Paner TM, Lane MJ, Benight AS (1997) Predicting sequence‐dependent melting stability of short duplex DNA oligomers. Biopolymers 44:217–239
Jaeger JA, Turner DH, Zuker M (1989) Improved predictions of secondary structures for RNA. Proc Natl Acad Sci U S A 86:7706–7710
Haasnoot C, De Bruin S, Berendsen R, Janssen H, Binnendijk T, Hilbers C, Van der Marel G, Van Boom J (1983) Structure, kinetics and thermodynamics of DNA hairpin fragments in solution. J Biomol Struct Dyn 1:115–129
Haasnoot CA, Hilbers CW, van der Marel GA, van Boom JH, Singh UC, Pattabiraman N, Kollman PA (1986) On loop folding in nucleic acid hairpin-type structures. J Biomol Struct Dyn 3:843–857
Tinoco I, Borer PN, Dengler B, Levine MD, Uhlenbeck OC, Crothers DM, Gralla J (1973) Improved estimation of secondary structure in ribonucleic acids. Nature 246:40–41
Senior MM, Jones RA, Breslauer KJ (1988) Influence of loop residues on the relative stabilities of DNA hairpin structures. Proc Natl Acad Sci U S A 85:6242–6246
Schildkraut C, Lifson S (1965) Dependence of the melting temperature of DNA on salt concentration. Biopolymers 3:195–208
Owczarzy R, You Y, Moreira BG, Manthey JA, Huang L, Behlke MA, Walder JA (2004) Effects of sodium ions on DNA duplex oligomers: improved predictions of melting temperatures. Biochemistry (NY) 43:3537–3554
Tan ZJ, Chen SJ (2006) Nucleic acid helix stability: effects of salt concentration, cation valence and size, and chain length. Biophys J 90:1175–1190
Tan ZJ, Chen SJ (2007) RNA helix stability in mixed Na/Mg2 solution. Biophys J 92:3615–3632
Tan ZJ, Chen SJ (2008) Salt dependence of nucleic acid hairpin stability. Biophys J 95:738–752
Turner D, Sugimoto N, Freier S (1990) Thermodynamics and kinetics of base-pairing and of DNA and RNA self-assembly and helix coil transition. Nucleic Acids 1:201–227
Bloomfield VA, Crothers DM, Tinoco I (2000) Nucleic acids: structures, properties, and functions. University Science Books, Sausalito
Ansari A, Kuznetsov SV, Shen Y (2001) Configurational diffusion down a folding funnel describes the dynamics of DNA hairpins. Proc Natl Acad Sci U S A 98:7771–7776
Shen Y, Kuznetsov SV, Ansari A (2001) Loop dependence of the dynamics of DNA hairpins. J Phys Chem B 105:12202–12211
Kuznetsov SV, Shen Y, Benight AS, Ansari A (2001) A semiflexible polymer model applied to loop formation in DNA hairpins. Biophys J 81:2864–2875
Bevilacqua PC, Blose JM (2008) Structures, kinetics, thermodynamics, and biological functions of RNA hairpins. Annu Rev Phys Chem 59:79–103
Wallace MI, Ying L, Balasubramanian S, Klenerman D (2001) Non-Arrhenius kinetics for the loop closure of a DNA hairpin. Proc Natl Acad Sci U S A 98:5584–5589
Wallace MI, Ying L, Balasubramanian S, Klenerman D (2000) FRET fluctuation spectroscopy: exploring the conformational dynamics of a DNA hairpin loop. J Phys Chem B 104:11551–11555
Wemmer DE, Chou SH, Hare DR, Reid BR (1985) Duplex-hairpin transitions in DNA: NMR studies on CGCGTATACGCG. Nucleic Acids Res 13:3755–3772
Youil R, Kemper BW, Cotton R (1995) Screening for mutations by enzyme mismatch cleavage with T4 endonuclease VII. Proc Natl Acad Sci U S A 92:87–91
Nelson NC, Hammond PW, Matsuda E, Goud AA, Becker MM (1996) Detection of all single-base mismatches in solution by chemiluminescence. Nucleic Acids Res 24:4998–5003
Dubertret B, Calame M, Libchaber AJ (2001) Single-mismatch detection using gold-quenched fluorescent oligonucleotides. Nat Biotechnol 19:365–370
Kostrikis LG, Tyagi S, Mhlanga MM, Ho DD, Kramer FR (1998) Spectral genotyping of human alleles. Science 279:1228–1229
Marras SAE, Russell Kramer F, Tyagi S (1999) Multiplex detection of single-nucleotide variations using molecular beacons. Genet Anal Biomol Eng 14:151–156
Meroueh M, Chow CS (1999) Thermodynamics of RNA hairpins containing single internal mismatches. Nucleic Acids Res 27:1118–1125
Bonnet G, Tyagi S, Libchaber A, Kramer FR (1999) Thermodynamic basis of the enhanced specificity of structured DNA probes. Proc Natl Acad Sci U S A 96:6171–6176
Tsourkas A, Behlke MA, Bao G (2002) Structure–function relationships of shared‐stem and conventional molecular beacons. Nucleic Acids Res 30:4208–4215
Cantor CR (1980) Biophysical chemistry: Part III: The behavior of biological macromolecules. WH Freeman & Co, San Francisco
Marras SAE, Kramer FR, Tyagi S (2002) Efficiencies of fluorescence resonance energy transfer and contact‐mediated quenching in oligonucleotide probes. Nucleic Acids Res 30:e122
Demidov VV, Frank-Kamenetskii MD (2004) Two sides of the coin: affinity and specificity of nucleic acid interactions. Trends Biochem Sci 29:62–71
Lomakin A, Frank-Kamenetskii MD (1998) A theoretical analysis of specificity of nucleic acid interactions with oligonucleotides and peptide nucleic acids (PNAs). J Mol Biol 276:57–70
Kuhn H, Demidov VV, Coull JM, Fiandaca MJ, Gildea BD, Frank-Kamenetskii M (2002) Hybridization of DNA and PNA molecular beacons to single-stranded and double-stranded DNA targets. J Am Chem Soc 124:1097–1103
Tan L, Li Y, Drake TJ, Moroz L, Wang K, Li J, Munteanu A, Yang CJ, Martinez K, Tan W (2005) Molecular beacons for bioanalytical applications. Analyst 130:1002–1005
Goel G, Kumar A, Puniya A, Chen W, Singh K (2005) Molecular beacon: a multitask probe. J Appl Microbiol 99:435–442
Bonnet G, Libchaber A (1999) Optimal sensitivity in molecular recognition. Phys A 263:68–77
Wang K, Tang Z, Yang CJ, Kim Y, Fang X, Li W, Wu Y, Medley CD, Cao Z, Li J (2009) Molecular engineering of DNA: molecular beacons. Angew Chem Int Ed 48:856–870
Bratu DP, Cha BJ, Mhlanga MM, Kramer FR, Tyagi S (2003) Visualizing the distribution and transport of mRNAs in living cells. Proc Natl Acad Sci U S A 100:13308–13313
Beisel CL, Bayer TS, Hoff KG, Smolke CD (2008) Model-guided design of ligand-regulated RNAi for programmable control of gene expression. Mol Syst Biol 4:224
Kim JR, Ostermeier M (2006) Modulation of effector affinity by hinge region mutations also modulates switching activity in an engineered allosteric TEM1 [beta]-lactamase switch. Arch Biochem Biophys 446:44–51
Vallée-Bélisle A, Ricci F, Plaxco KW (2009) Thermodynamic basis for the optimization of binding-induced biomolecular switches and structure-switching biosensors. Proc Natl Acad Sci U S A 106:13802–13807
Tsourkas A, Behlke MA, Bao G (2002) Hybridization of 2′‐O‐methyl and 2′‐deoxy molecular beacons to RNA and DNA targets. Nucleic Acids Res 30:5168–5174
Yao G, Fang X, Yokota H, Yanagida T, Tan W (2003) Monitoring molecular beacon DNA probe hybridization at the single‐molecule level. Chem Eur J 9:5686–5692
Nakano S, Kirihata T, Fujii S, Sakai H, Kuwahara M, Sawai H, Sugimoto N (2007) Influence of cationic molecules on the hairpin to duplex equilibria of self-complementary DNA and RNA oligonucleotides. Nucleic Acids Res 35:486–494
Chen C, Wang W, Wang Z, Wei F, Zhao XS (2007) Influence of secondary structure on kinetics and reaction mechanism of DNA hybridization. Nucleic Acids Res 35:2875–2884
Porschke D, Eigen M (1971) Co-operative non-enzymatic base recognition III. Kinetics of the helix–coil transition of the oligoribouridylic · oligoriboadenylic acid system and of oligoriboadenylic acid alone at acidic pH. J Mol Biol 62:361–364
Pörschke D, Uhlenbeck O, Martin F (1973) Thermodynamics and kinetics of the helix‐coil transition of oligomers containing GC base pairs. Biopolymers 12:1313–1335
McConaughy BL, Laird CD, McCarthy BJ (1969) Nucleic acid reassociation in formamide. Biochemistry (NY) 8:3289–3295
Kohne DE, Levison SA, Byers MJ (1977) Room temperature method for increasing the rate of DNA reassociation by many thousandfold: the phenol emulsion reassociation technique. Biochemistry (NY) 16:5329–5341
Yao G, Tan W (2004) Molecular-beacon-based array for sensitive DNA analysis. Anal Biochem 331:216–223
Tsourkas A, Bao G (eds) (2001) Detecting mRNA transcripts using FRET-enhanced molecular beacons. ASME BED
Vijayanathan V, Thomas T, Sigal LH, Thomas T (2002) Direct measurement of the association constant of HER2/neu antisense oligonucleotide to its target RNA sequence using a molecular beacon. Antisense Nucleic Acid Drug Dev 12:225–233
Mhlanga MM, Vargas DY, Fung CW, Kramer FR, Tyagi S (2005) tRNA-linked molecular beacons for imaging mRNAs in the cytoplasm of living cells. Nucleic Acids Res 33:1902–1912
Santangelo PJ, Bao G (2007) Dynamics of filamentous viral RNPs prior to egress. Nucleic Acids Res 35:3602–3611
Martinez K, Estevez M, Wu Y, Phillips JA, Medley CD, Tan W (2009) Locked nucleic acid based beacons for surface interaction studies and biosensor development. Anal Chem 81:3448–3454
Wu Y, Yang CJ, Moroz LL, Tan W (2008) Nucleic acid beacons for long-term real-time intracellular monitoring. Anal Chem 80:3025–3028
Vester B, Wengel J (2004) LNA (locked nucleic acid): high-affinity targeting of complementary RNA and DNA. Biochemistry (NY) 43:13233–13241
Kurreck J, Wyszko E, Gillen C, Erdmann VA (2002) Design of antisense oligonucleotides stabilized by locked nucleic acids. Nucleic Acids Res 30:1911–1918
Aartsma-Rus A, Kaman W, Bremmer-Bout M, Janson A, Den Dunnen J, van Ommen GJB, van Deutekom J (2004) Comparative analysis of antisense oligonucleotide analogs for targeted DMD exon 46 skipping in muscle cells. Gene Ther 11:1391–1398
Romani A (2007) Regulation of magnesium homeostasis and transport in mammalian cells. Arch Biochem Biophys 458:90–102
Santangelo PJ (2010) Molecular beacons and related probes for intracellular RNA imaging. Wiley Interdiscip Rev Nanomed Nanobiotechnol 2:11–19
Koshkin AA, Nielsen P, Meldgaard M, Rajwanshi VK, Singh SK, Wengel J (1998) LNA (locked nucleic acid): an RNA mimic forming exceedingly stable LNA: LNA duplexes. J Am Chem Soc 120:13252–13253
Koshkin AA, Singh SK, Nielsen P, Rajwanshi VK, Kumar R, Meldgaard M, Olsen CE, Wengel J (1998) LNA (locked nucleic acids): synthesis of the adenine, cytosine, guanine, 5-methylcytosine, thymine and uracil bicyclonucleoside monomers, oligomerisation, and unprecedented nucleic acid recognition. Tetrahedron 54:3607–3630
Wang L, Yang CJ, Medley CD, Benner SA, Tan W (2005) Locked nucleic acid molecular beacons. J Am Chem Soc 127:15664–15665
Yoo SM, Keum KC, Yoo SY, Choi JY, Chang KH, Yoo NC, Yoo WM, Kim JM, Lee D, Lee SY (2004) Development of DNA microarray for pathogen detection. Biotechnol Bioprocess Eng 9:93–99
Gracey AY, Cossins AR (2003) Application of microarray technology in environmental and comparative physiology. Annu Rev Physiol 65:231–259
Fan J, Yang X, Wang W, Wood WH, Becker KG, Gorospe M (2002) Global analysis of stress-regulated mRNA turnover by using cDNA arrays. Proc Natl Acad Sci U S A 99:10611–10616
Fang Y, Frutos AG, Lahiri J (2003) Ganglioside microarrays for toxin detection. Langmuir 19:1500–1505
Sekar M, Bloch W, St John PM (2005) Comparative study of sequence-dependent hybridization kinetics in solution and on microspheres. Nucleic Acids Res 33:366–375
Gao Y, Wolf LK, Georgiadis RM (2006) Secondary structure effects on DNA hybridization kinetics: a solution versus surface comparison. Nucleic Acids Res 34:3370–3377
Peterson AW, Heaton RJ, Georgiadis RM (2001) The effect of surface probe density on DNA hybridization. Nucleic Acids Res 29:5163–5168
Vainrub A, Montgomery Pettitt B (2003) Surface electrostatic effects in oligonucleotide microarrays: control and optimization of binding thermodynamics. Biopolymers 68:265–270
Halperin A, Buhot A, Zhulina E (2005) Brush effects on DNA chips: thermodynamics, kinetics, and design guidelines. Biophys J 89:796–811
Tawa K, Knoll W (2004) Mismatching base‐pair dependence of the kinetics of DNA–DNA hybridization studied by surface plasmon fluorescence spectroscopy. Nucleic Acids Res 32:2372–2377
Stillman BA, Tonkinson JL (2001) Expression microarray hybridization kinetics depend on length of the immobilized DNA but are independent of immobilization substrate. Anal Biochem 295:149–157
Glazer MI, Fidanza JA, McGall GH, Trulson MO, Forman JE, Frank CW (2007) Kinetics of oligonucleotide hybridization to DNA probe arrays on high-capacity porous silica substrates. Biophys J 93:1661–1676
Liu X, Tan W (1999) A fiber-optic evanescent wave DNA biosensor based on novel molecular beacons. Anal Chem 71:5054–5059
Wang H, Li J, Liu H, Liu Q, Mei Q, Wang Y, Zhu J, He N, Lu Z (2002) Label-free hybridization detection of a single nucleotide mismatch by immobilization of molecular beacons on an agarose film. Nucleic Acids Res 30:e61
Du H, Strohsahl CM, Camera J, Miller BL, Krauss TD (2005) Sensitivity and specificity of metal surface-immobilized molecular beacon biosensors. J Am Chem Soc 127:7932–7940
Du H, Disney MD, Miller BL, Krauss TD (2003) Hybridization-based unquenching of DNA hairpins on Au surfaces: prototypical “molecular beacon” biosensors. J Am Chem Soc 125:4012–4013
Sauthier ML, Carroll RL, Gorman CB, Franzen S (2002) Nanoparticle layers assembled through DNA hybridization: characterization and optimization. Langmuir 18:1825–1830
Dave N, Liu J (2010) Fast molecular beacon hybridization fast molecular bea-con hybridization. J Phys Chem B 114:15694–15699
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2013 Springer-Verlag Berlin Heidelberg
About this chapter
Cite this chapter
Peng, L., Tan, W. (2013). Thermodynamic and Kinetic Properties of Molecular Beacons. In: Yang, C., Tan, W. (eds) Molecular Beacons. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-39109-5_2
Download citation
DOI: https://doi.org/10.1007/978-3-642-39109-5_2
Published:
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-642-39108-8
Online ISBN: 978-3-642-39109-5
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)