Abstract
In recent years interest in blood viscosity has increased significantly in different biomedical areas. Blood viscosity, a measure of the resistance of blood flow, related to its thickness and stickiness, is one of the main biophysical properties of blood. Many factors affect blood viscosity, both in physiological and in pathological conditions.
The aim of this study is to estimate blood viscosity by using the regression equation of viscosity which is based on hematocrit and total plasma proteins. It can be used to perform several observations regards the main factors which can influence blood viscosity. The main contribution regards the correlation between viscosity values and other important biological parameters such as cholesterol. This correlation has been supported by performing statistical tests and it suggest that the viscosity could be the main risk factor in cardiovascular diseases. Moreover, it is the only biological measure being correlated with the other cardiovascular risk factors. Results obtained are compliant with values obtained by using the standard viscosity measurement through a viscometer.
You have full access to this open access chapter, Download conference paper PDF
Similar content being viewed by others
Keywords
1 Introduction
Blood is a fluid in a viscous, red and opaque state, which is distributed in the body through the circulatory system. It is a non-Newtonian fluid composed by many cells with different features and tasks [1]. Blood consists of two parts: (i) a liquid part, called plasma, and (ii) a corpuscular part, formed by cells or fragments of cells. The former represents the 54% of the circulating blood mass and it is composed by 90% of water and 10% of inorganic substances, including critical proteins which are fundamental for sustaining health and life. The latter contains the so-called figurative elements including red blood cells (or erythrocytes), white blood cells (or leukocytes) and platelets (thrombocytes), as well as some liquid particles. The platelets are cellular fragments essential for coagulation and in case of vessel lesion. The white blood cells defense the organism in the case of pathogens aggression. The red blood cells represent the 40–50% of total volume so that this high concentration influences the behavior of the blood more than the other blood cells. Moreover, they are particularly important in the study of rheology and blood flow dynamics, but also in hemorrhage which is specifically aimed at the study of blood and viscosity. The volumetric concentration of erythrocytes in the blood is commonly known as hematocrit which is responsible for blood viscosity. In fact, it indicates the relationship between the liquid and the corpuscular parts of the blood expressed as a percentage or a fraction. Blood viscosity represents the inherent resistance of blood to flow and it is obtained by summing the viscosity of hematocrit with the viscosity of plasma [2]. In the International System, the unit of measure used for viscosity is “Pascal-Second” (\(Pa\cdot s\)). In general, the viscosity coefficient of a fluid indicates the ratio between the force applied per unit area and the relative velocity gradient, called shear rate and expressed in s\(^{-1}\). The force per unit area that produces the shear rate is called shear stress and is expressed in dyne/cm\(^{2}\). The relationship between shear stress and shear rate corresponds to viscosity. The main factor that influences blood viscosity is hematocrit. Red blood cells, in contrast to white blood cells and platelets, strongly influence viscosity by: (i) volumetric fraction: its increase leads to an increase in viscosity; (ii) rouleaux development: the aggregation of red blood cells increases viscosity; (iii) cells warping: a decrease in the deformability of red blood cells leads to an increase in viscosity [3].
The relationship between volumetric fraction and dynamic viscosity is a non-linear relationship. Viscosity grows rapidly for hematocrit values above 45%.
1.1 Related Work
Recently, the interest in studying blood viscosity has significantly increased because it has been shown that alterations in its normal values can be related to diseases [4,5,6,7]. Viscosity can be determined by (i) a reduction in plasma, (ii) a change in the volume or number of red blood cells, (iii) an increase in blood coagulation capacity [8,9,10]. These events cause greater resistance in blood flow, forcing the heart to increase its contraction force. Conventional methods for measuring blood viscosity include the use of different types of viscometers [11]. They cannot perform a direct measure but they estimate an experimental parameter related to viscosity in a known manner. Moreover, they present disadvantages in terms of avoiding to deal with heavy and expensive equipment, large sample consumption, and long measurement time. To overcome these limitations, new measurement techniques have been proposed in literature [12]. A quick method to measure blood viscosity, which allows to verify the accuracy of predictions and to biologically determine the variability of the blood viscosity and its components, is based on a regression equation [13,14,15,16]. These study are based on this regression equation and they are been conducted on demographic and laboratory variables in normal subjects to identify deviations related to cardiovascular diseases.
2 Methods
The dataset used for the experiment contains features from 4320 subjects who enrolled at the Laboratory of Clinical Biochemistry in University of Magna Graecia. The subjects attended the laboratory from January to December 2016 for routine blood tests. Every subject, between 12 and 100 years old, gave written informed consent to this research conforming to the ethical guidelines of the Declaration of Helsinki, as reflected in a priori approval by the Ethical Committee of Mater Domini Hospital in Catanzaro, Italy. Moreover, features of the dataset have been acquired by taking into consideration all clinical variabilities (e.g. temperature, use of anticoagulants). For each subject, the number of clinical record has been linked to an identifier associated with personal data (e.g. date of birth, sex) and the examination date. In this study, the regression equation has been applied to the dependent variable (blood viscosity) and has been correlated to the independent variables (e.g. proteins, hematocrit). The goal is to estimate a functional relationship between the dependent variable and the independent variables. Blood viscosity have been calculated for different shear rates:
-
Shear rate 208 s\(^{-1}\) with (x, y, z) = (0.12, 0.17, 2.07);
-
Shear rate 104 s\(^{-1}\) with (x, y, z) = (0.12, 0.19, 2.13);
-
Shear rate 52 s\(^{-1}\) with (x, y, z) = (0.14, 0.22, 2.6);
-
Shear rate 5.2 s\(^{-1}\) with (x, y, z) = (0.46, 0.62, 14.25).
The Eq. 1 used for the Whole Blood Viscosity (WBV) is [17]:
WBV is the dependent variable and HCT (hematocrit, expressed in percentage) and TP (plasma protein concentration, expressed in g/dL) are the independent clinical variables. Blood viscosity is dependent because it depends on the value of proteins and hematocrit and also on the variables x, y, z, (related to the thickness of vessels) defined according to the shear rate. Moreover, viscosity also depends on the cutting speed because blood is a non-Newtonian fluid, due to the presence of cells, so the viscosity is closely linked to the speed gradient.
All the variables have been measured in the laboratories of Clinical Biochemistry and Clinical Pathology of the Mater Domini Polyclinic. Possible correlations between blood viscosity and hematocrit, proteins and shear rate have been evaluated for two groups of subjects divided by sex and age. The viscosity correlation with other important variables has been analyzed to validate the correct functioning of the organism. In particular, the serum calcium has been examined and the analysis has been performed based on five different serum calcium values:
-
Group 1: serum calcium \(\le \) 9 mg/dl;
-
Group 2: serum calcium > 9 mg/dl and \(\le \) 9.3 mg/dl;
-
Group 3: serum calcium > 9.3 mg/dl and \(\le \) 9.6 mg/dl;
-
Group 4: serum calcium > 9.6 mg/dl and \(\le \) 9.8 mg/dl;
-
Group 5: serum calcium > 9.8 mg/dl.
Statistical analysis has been performed by using T-test with a significance level of 0.05. The association between viscosity and calcium has been studied by using a Pearson correlation. Multiple regression analysis has been used to evaluate the correlation adjusted for age between viscosity and hematocrit, proteins and share rate. The analysis of variance ANOVA has been performed to compare the multivariate means among the 5 calcium groups.
3 Results
The overall population consists of 4320 subjects (1922 women and 2398 men) in an age range between 12 and 100 years. In order to manage the data, apply the regression equation and perform the analysis, IBM Watson (www.ibm.com/watson-analytics) has been used. Watson Analytics is a cloud-based software for data analysis and visualization containing modules able to find useful information through statistical and machine learning models. Table 1 reports mean and standard deviation values for age, hematocrit, proteins and serum calcium variables. Women are younger than men and show significantly lower hematocrit. Proteins and serum calcium are similar for women and men.
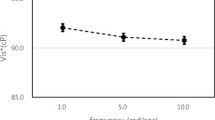
The higher values of hematocrit in men is due to the higher testosterone levels. In fact, erythrocytes are produced in the bone marrow thanks to the stimulating action of erythropoietin (EPO), an action that depends on several factors, including the concentration of testosterone. Table 2 reports the viscosity calculated by using the regression equation and related to the different values of shear-rate. Viscosity increases significantly and progressively as the shear-rate decreases, both for men and women. Since blood is a non-Newtonian fluid, viscosity increases as the cutting speed decreases.
Pearson correlation and T-test have been performed to evaluate correlations between viscosity and age, hematocrit, proteins and calcium. These results are reported in Table 3. A weak correlation between age and viscosity can be observed and the T-test produces a result statistically significant with a p-value < 0.001, confirming the weak relation. A significant and direct association between hematocrit and viscosity can be highlighted. This is a direct relationship, hence viscosity increases with the increase of the hematocrit. By considering the gender, higher values are reported in male, which can be explained by viscosity being closely related to hematocrit. This strong correlation is statistically confirmed by a p-value < 0.001. Moreover, Pearson correlation confirms a moderate correlation between proteins and viscosity and between calcium and viscosity, both with a p-value < 0.001. Blood viscosity increases with the increase of proteins but the presence of proteins is not highly influential, therefore it does not influence the viscosity trend excessively. The correlation between the serum calcium and blood viscosity has been calculated to evaluate the presence of a major cardiovascular risk. The dataset has been subdivided into five groups with respect to the different calcium values (as reported in the previous section). Table 4 reports the dimension of each group in term of number of subjects, mean and standard deviation (SD) value for blood viscosity.
We note a mean blood viscosity that increases with the increase of the calcium values in each group. ANOVA statistical test has been applied between the mean values and the result indicates a significant difference between the means of the groups. Then, the Turkey-Kramer Post-Hoc test has been applied because the groups present a different dimension and also in this case a significant difference between means is reported, especially between the first and the last group (Groups 1 and 5). Hematocrit values have been evaluated related to the five groups for their strong correlation with blood viscosity. Results are reported in Table 5.
4 Discussion
The main result of this study is that blood viscosity can be calculated by applying the regression equation proposed in this paper. The obtained results have been compared to the ones obtained through a viscometer instrument and they show a statistical similarity (test results and details not reported), thus confirming the validity of the method. The second result is the evaluation and the identification of the main factors influencing blood viscosity. Hematocrit, plasma viscosity and erythrocyte deformability are factors independently associated with viscosity at high and low shear rates. Viscosity increases significantly as shear rate decreases and hematocrit increases. Viscosity at high shear rates is strongly influenced by erythrocyte deformability, while at low shear rates it is influenced by the aggregation of red blood cells. These characteristics also depend on the properties of erythrocytes and plasma. Another result is the evidence of a significant correlation between serum calcium and viscosity. Results also show that blood viscosity is one of the risk factors for cardiovascular diseases and it is a biological measure correlated with all the other major cardiovascular risk factors, such as an excessive calcium increase. In particular, a higher blood viscosity is due to an increase in calcium level. By acting on the amount of calcium, this reflects on cholesterol and, consequently, on viscosity.
5 Conclusion
We presented a cross-sectional study to evaluate the application of the regression equation on blood viscosity measurements. The increase in blood viscosity as a pathogenetic factor for the development of cardiovascular complications and diseases has been evaluated with respect to specific clinical and biological parameters. A significant correlation between serum calcium and viscosity, both in men and women, is reported. This suggests that the calcium supplement in the population should be done with caution. The result of this study is in line with results obtained by using the standard blood viscosity measurement, validating the proposed method and verifying the effect of calcium on cardiovascular risks.
References
Rogers, K.: Blood: Physiology and Circulation (The Human Body). The Rosen Publishing Group, New York (2010)
Goslinga, H.: Blood Viscosity and Shock: The Role of Hemodilution, Hemoconcentration and Defibrination. Springer, Heidelberg (1984). https://doi.org/10.1007/978-3-642-69260-4
Chien, S.: Determinants of blood viscosity and red cell deformability. Scand. J. Clin. Lab. Invest. 41(1981), 712 (2009)
Chen, G., Zhao, L., Liu, Y., Liao, F., Han, D., Zhou, H.: Regulation of blood viscosity in disease prevention and treatment. Chin. Sci. Bull. 57(16), 1946–1952 (2012)
Mazza, A., Fruci, B., Guzzi, P., D’Orrico, B., Malaguarnera, R., Veltri, P., Fava, A., Belfiore, A.: In PCOS patients the addition of low-dose spironolactone induces a more marked reduction of clinical and biochemical hyperandrogenism than metformin alone. Nutr. Metab. Cardiovasc. Dis. 24(2), 132–139 (2014)
Palopoli, L., Rombo, S.E., Terracina, G., Tradigo, G., Veltri, P.: Improving protein secondary structure predictions by prediction fusion. Inf. Fus. 10(3), 217–232 (2009)
Vizza, P., Curcio, A., Tradigo, G., Indolfi, C., Veltri, P.: A framework for the atrial fibrillation prediction in electrophysiological studies. Comput. Methods Program. Biomed. 120(2), 65–76 (2015)
Sloop, G.D.: Blood Viscosity: Its Role in Cardiovascular Pathophysiology and Hematology. Nova Science Publishers, Inc., Hauppauge (2017)
De Napoli, I.E., Zanetti, E.M., Fragomeni, G., Audenino, A.L., Catapano, G.: Transport modeling of convection-enhanced hollow fiber membrane bioreactors for therapeutic applications. J. Membr. Sci. 471, 347–361 (2014)
Caruso, M.V., Gramigna, V., Renzulli, A., Fragomeni, G.: Computational analysis of aortic hemodynamics during total and partial extra-corporeal membrane oxygenation and intra-aortic balloon pump support. Acta Bioeng. Biomech. 18(3), 3–9 (2016)
Schramm, G.A.: A Practical Approach to Rheology and Rheometry, 2nd edn. Thermo Haake Rheology, Karlsruhe (1994)
Kim, B.J., Lee, S.Y., Jee, S., Atajanov, A., Yang, S.: Micro-viscometer for measuring shear-varying blood viscosity over a wide-ranging shear rate. Sensors (Basel.) 17(6), E1442 (2017)
Stoeff, S., Jovtchev, S., Trifonova, N.: Whole blood viscosity assessment in arterial hypertension: a mathematical approach. Measurements 6(9), 10 (2012)
Sahin, B., Yigitarslan, S.: The equation for prediction of blood viscosity from biochemical laboratory data. In: AIP Conference Proceedings, vol. 1653, no. 1 (2015)
Ruef, P., Gehm, J., Gehm, L., Felbinger, C., Pöschl, J., Kuss, N.: Determination of whole blood and plasma viscosity by means of flow curve analysis. Gen. Physiol. Biophys. 33(3), 285–293 (2014)
Çinar, Y., Şenyol, A.M., Duman, K.: Blood viscosity and blood pressure: role of temperature and hyperglycemia. Am. J. Hypertens. 14(5), 433–438 (2001)
Pop, G.A., Chang, Z.Y., Slager, C.J., Kooij, B.J., van Deel, E.D., Moraru, L., Quak, J., Meijer, G.C., Duncker, D.J.: Catheter-based impedance measurements in the right atrium for continuously monitoring hematocrit and estimating blood viscosity changes; an in vivo feasibility study in swine. Biosens. Bioelectron. 19(12), 1685–1693 (2004)
Acknowledgments
This contribution has been funded by grant POR FESR/FSE Regione Calabria for SISTABENE and PIHGIS projects.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer International Publishing AG, part of Springer Nature
About this paper
Cite this paper
Vizza, P., Tradigo, G., Parrilla, M., Guzzi, P.H., Gnasso, A., Veltri, P. (2018). On Blood Viscosity and Its Correlation with Biological Parameters. In: Shi, Y., et al. Computational Science – ICCS 2018. ICCS 2018. Lecture Notes in Computer Science(), vol 10861. Springer, Cham. https://doi.org/10.1007/978-3-319-93701-4_26
Download citation
DOI: https://doi.org/10.1007/978-3-319-93701-4_26
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-93700-7
Online ISBN: 978-3-319-93701-4
eBook Packages: Computer ScienceComputer Science (R0)