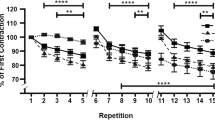
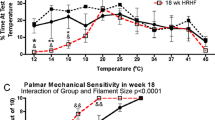
Abstract
The heat shock protein (Hsp) response is understudied with non-exercise overuse injuries. We focused on the Hsp response in muscles and tendons undergoing such injury or cyclical loading. Hsp25 mRNA and protein levels increase in muscles undergoing functional overload, and show greater increases in fast type muscles. In an operant rat model of reaching and grasping, the inducible form of Hsp70 increased in muscles and tendons showing injury, with the greatest increase in rats performing a high repetition high force for 12 weeks, compared to easier repetition/loading paradigms. These increases were paralleled by increases in several repair-associated proteins (osteoactivin, MMPs, and TGFB1). Trapezius biopsies from patients with myalgia show increased mRNA levels of Hsp72 and decreased levels of growth and metabolism regulators. Prolonged exercise interventions in general, when provided to subjects with trapezius myalgia, decreased Hsp72 mRNA levels, while specific strength training of shoulder and neck muscles increased mRNA levels of analytes related to carbohydrate oxidation. In a rat model of supraspinatus injury, the Hsp response appeared related to the cascade of stress-related programmed cell death in torn tendons. A mild mechanical stimulation of cultured tendon fibroblasts reduced apoptosis and increased cell proliferation and may be helpful for tissue regeneration.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Bae, Y. H., & Min, K. S. (2016). Associations between work-related musculoskeletal disorders, quality of life, and workplace stress in physical therapists. Industrial Health, 54, 347–353.
Banfi, G., et al. (2004). Exercise raises serum heat-shock protein 70 (Hsp70) levels. Clinical Chemistry and Laboratory Medicine: CCLM/FESCC, 42, 1445–1446.
Barbe, M. F., et al. (1988). Hyperthermia protects against light damage in the rat retina. Science, 241, 1817–1820.
Barbe, M. F., et al. (2003). Chronic repetitive reaching and grasping results in decreased motor performance and widespread tissue responses in a rat model of MSD. Journal of Orthopaedic Research, 21, 167–176.
Barbe, M. F., et al. (2013). The interaction of force and repetition on musculoskeletal and neural tissue responses and sensorimotor behavior in a rat model of work-related musculoskeletal disorders. BMC Musculoskeletal Disorders, 14, 303.
Barkhausen, T., et al. (2003). Modulation of cell functions of human tendon fibroblasts by different repetitive cyclic mechanical stress patterns. Experimental and Toxicologic Pathology, 55, 153–158.
Barr, A. E., & Barbe, M. F. (2002). Pathophysiological tissue changes associated with repetitive movement: A review of the evidence. Physical Therapy, 82, 173–187.
Barr, A. E., & Barbe, M. F. (2004). Inflammation reduces physiological tissue tolerance in the development of work-related musculoskeletal disorders. Journal of Electromyography and Kinesiology, 14, 77–85.
Bureau of Labor Statistics. (2012). Nonfatal occupational injuries and illnesses requiring days away from work, 2011. http://www.bls.gov/news.release/osh2.nr0.htm News Release USDL-12-2204, November 8, 2012.
Bureau of Labor Statistics (2014). Prevention of work-related musculoskeletal disorders. http://www.bls.gov/news.release/pdf/osh2.pdf.
Cahill, C. M., et al. (1996). Transcriptional repression of the prointerleukin 1beta gene by heat shock factor 1. The Journal of Biological Chemistry, 271, 24874–24879.
Carmeli, E., et al. (2010). Increased iNOS, MMP-2, and HSP-72 in skeletal muscle following high-intensity exercise training. Journal of Basic and Clinical Physiology and Pharmacology, 21, 127–146.
Cote, J. N. (2012). A critical review on physical factors and functional characteristics that may explain a sex/gender difference in work-related neck/shoulder disorders. Ergonomics, 55, 173–182.
Craig, E. A., et al. (1993). Heat shock proteins: Molecular chaperones of protein biogenesis. Microbiological Reviews, 57, 402–414.
De, A. K., et al. (2000). Exaggerated human monocyte IL-10 concomitant to minimal TNF-alpha induction by heat-shock protein 27 (Hsp27) suggests Hsp27 is primarily an antiinflammatory stimulus. Journal of Immunology, 165, 3951–3958.
Escobedo, J., et al. (2004). hsp25 protects skeletal muscle cells against oxidative stress. Free Radical Biology & Medicine, 37, 1455–1462.
Fine, L. J., et al. (1986). Detection of cumulative trauma disorders of upper extremities in the workplace. Journal of Occupational Medicine, 28(8), 674.
Fisher, P. W., et al. (2015). Increased CCN2, substance P and tissue fibrosis are associated with sensorimotor declines in a rat model of repetitive overuse injury. Journal of Cell Communication and Signaling, 9(1), 37–54.
Frara, N., et al. (2016). Growth and repair factors, osteoactivin, matrix metalloproteinase and heat shock protein 72, increase with resolution of inflammation in musculotendinous tissues in a rat model of repetitive grasping. BMC Musculoskeletal Disorders, 17, 34.
Frara, N., et al. (2017). Substance P increases CCN2 dependent on TGF-beta yet Collagen Type I via TGF-beta1 dependent and independent pathways in tenocytes. Connective Tissue Research, 1, 1–15.
Fujii, N., et al. (2017). Heat shock protein 90 contributes to cutaneous vasodilation through activating nitric oxide synthase in young male adults exercising in the heat. Journal of Applied Physiology, (1985), jap 00446 2017.
Fung, D. T., et al. (2009). Subrupture tendon fatigue damage. Journal of Orthopaedic Research, 27, 264–273.
Fung, D. T., et al. (2010). Early response to tendon fatigue damage accumulation in a novel in vivo model. Journal of Biomechanics, 43, 274–279.
Furochi, H., et al. (2007a). Osteoactivin fragments produced by ectodomain shedding induce MMP-3 expression via ERK pathway in mouse NIH-3T3 fibroblasts. FEBS Letters, 581, 5743–5750.
Furochi, H., et al. (2007b). Overexpression of osteoactivin protects skeletal muscle from severe degeneration caused by long-term denervation in mice. The Journal of Medical Investigation: JMI, 54, 248–254.
Gabai, V. L., et al. (1997). Hsp70 prevents activation of stress kinases. A novel pathway of cellular thermotolerance. The Journal of Biological Chemistry, 272, 18033–18037.
Gallagher, S., & Heberger, J. R. (2013). Examining the interaction of force and repetition on musculoskeletal disorder risk: A systematic literature review. Human Factors, 55, 108–124.
Gehrig, S. M., et al. (2012). Hsp72 preserves muscle function and slows progression of severe muscular dystrophy. Nature, 484, 394–398.
Hartl, F. U. (1996). Molecular chaperones in cellular protein folding. Nature, 381, 571–579.
Hirunsai, M., et al. (2015). Heat stress promotes extracellular matrix remodelling via TGF-beta1 and MMP-2/TIMP-2 modulation in tenotomised soleus and plantaris muscles. International Journal of Hyperthermia, 31(4), 336–348.
Huey, K. A., et al. (2007). Modulation of HSP25 and TNF-alpha during the early stages of functional overload of a rat slow and fast muscle. Journal of Applied Physiology. (1985), 102, 2307–2314.
Jagodzinski, M., et al. (2006). Influence of cyclic mechanical strain and heat of human tendon fibroblasts on HSP-72. European Journal of Applied Physiology, 96, 249–256.
Kannus, P., et al. (1997). Effects of training, immobilization and remobilization on tendons. Scandinavian Journal of Medicine & Science in Sports, 7, 67–71.
Kherif, S., et al. (1998). Matrix metalloproteinases MMP-2 and MMP-9 in denervated muscle and injured nerve. Neuropathology and Applied Neurobiology, 24, 309–319.
Koh, T. J. (2002). Do small heat shock proteins protect skeletal muscle from injury? Exercise and Sport Sciences Reviews, 30, 117–121.
Koh, T. J., & Escobedo, J. (2004). Cytoskeletal disruption and small heat shock protein translocation immediately after lengthening contractions. American Journal of Physiology. Cell Physiology, 286, C713–C722.
Martin-Ventura, J. L., et al. (2006). Biological significance of decreased HSP27 in human atherosclerosis. Arteriosclerosis, Thrombosis, and Vascular Biology, 26, 1337–1343.
McArdle, A., et al. (2004). Overexpression of HSP70 in mouse skeletal muscle protects against muscle damage and age-related muscle dysfunction. The FASEB Journal, 18, 355–357.
Millar, N. L., & Murrell, G. A. (2012). Heat shock proteins in tendinopathy: Novel molecular regulators. Mediators of Inflammation, 436203.
Millar, N. L., et al. (2008). Heat shock protein and apoptosis in supraspinatus tendinopathy. Clinical Orthopaedics and Related Research, 466, 1569–1576.
Miyabara, E. H., et al. (2006). Overexpression of inducible 70-kDa heat shock protein in mouse attenuates skeletal muscle damage induced by cryolesioning. American Journal of Physiology. Cell Physiology, 290, C1128–C1138.
Miyabara, E. H., et al. (2012). Overexpression of inducible 70-kDa heat shock protein in mouse improves structural and functional recovery of skeletal muscles from atrophy. Pflügers Archiv, 463, 733–741.
Morton, J. P., et al. (2009). The exercise-induced stress response of skeletal muscle, with specific emphasis on humans. Sports Medicine, 39, 643–662.
Mounier, N., & Arrigo, A. P. (2002). Actin cytoskeleton and small heat shock proteins: How do they interact? Cell Stress & Chaperones, 7, 167–176.
Neviaser, A., et al. (2012). Basic mechanisms of tendon fatigue damage. Journal of Shoulder and Elbow Surgery, 21, 158–163.
Noble, E. G., & Shen, G. X. (2012). Impact of exercise and metabolic disorders on heat shock proteins and vascular inflammation. Autoimmune Diseases, 836519.
Noble, E. G., et al. (1999). Differential expression of stress proteins in rat myocardium after free wheel or treadmill run training. Journal of Applied Physiology .1985, 86, 1696–1701.
Ogawa, T., et al. (2005). Osteoactivin upregulates expression of MMP-3 and MMP-9 in fibroblasts infiltrated into denervated skeletal muscle in mice. American Journal of Physiology. Cell Physiology, 289, C697–C707.
Paulsen, G., et al. (2007). Maximal eccentric exercise induces a rapid accumulation of small heat shock proteins on myofibrils and a delayed HSP70 response in humans. American Journal of Physiology. Regulatory, Integrative and Comparative Physiology, 293, R844–R853.
Piligian, G., et al. (2000). Evaluation and management of chronic work-related musculoskeletal disorders of the distal upper extremity. American Journal of Industrial Medicine, 37, 75–93.
Rempel, D. M., & Diao, E. (2004). Entrapment neuropathies: Pathophysiology and pathogenesis. Journal of Electromyography and Kinesiology, 14, 71–75.
Rempel, D. M., et al. (1992). Work-related cumulative trauma disorders of the upper extremity. JAMA, 267, 838–842.
Saleh, A., et al. (2000). Negative regulation of the Apaf-1 apoptosome by Hsp70. Nature Cell Biology, 2, 476–483.
Senf, S. M., et al. (2008). Hsp70 overexpression inhibits NF-kappaB and Foxo3a transcriptional activities and prevents skeletal muscle atrophy. The FASEB Journal, 22, 3836–3845.
Senf, S. M., et al. (2013). Loss of the inducible hsp70 delays the inflammatory response to skeletal muscle injury and severely impairs muscle regeneration. PLoS One, 8, e62687.
Silverstein, B. A., et al. (1986). Hand wrist cumulative trauma disorders in industry. British Journal of Industrial Medicine, 43, 779–784.
Sjogaard, G., et al. (2010). Muscle oxygenation and glycolysis in females with trapezius myalgia during stress and repetitive work using microdialysis and NIRS. European Journal of Applied Physiology, 108, 657–669.
Sjogaard, G., et al. (2013). Exercise training and work task induced metabolic and stress-related mRNA and protein responses in myalgic muscles. BioMed Research International, 2013, 984523.
Skutek, M., et al. (2003). Cyclic mechanical stretching of human patellar tendon fibroblasts: Activation of JNK and modulation of apoptosis. Knee Surgery, Sports Traumatology, Arthroscopy, 11, 122–129.
Song, J., et al. (2001). Bag1-Hsp70 mediates a physiological stress signalling pathway that regulates Raf-1/ERK and cell growth. Nature Cell Biology, 3, 276–282.
Subadi, I., et al. (2017). Pain relief with wet cupping therapy in rats is mediated by heat shock protein 70 and ss-endorphin. The Iran Journal of Medical Sciences, 42, 384–391.
Tuttle, J. A., et al. (2017). The Hsp72 and Hsp90alpha mRNA responses to hot downhill running are reduced following a prior bout of hot downhill running, and occur concurrently within leukocytes and the vastus Lateralis. Frontiers in Physiology, 8, 473.
Tytell, M., et al. (1989). Photoreceptor protection from light damage by hyperthermia. Progress in Clinical and Biological Research, 314, 523–538.
van Rijn, R.M., et al. (2009) Associations between work-related factors and specific disorders at the elbow: A systematic literature review. Rheumatology (Oxford) 48, 528–536.
Welsh, M. J., & Gaestel, M. (1998). Small heat-shock protein family: Function in health and disease. Annals of the New York Academy of Sciences, 851, 28–35.
Wieten, L., et al. (2007). Cell stress induced HSP are targets of regulatory T cells: A role for HSP inducing compounds as anti-inflammatory immuno-modulators? FEBS Letters, 581, 3716–3722.
Woolf, A. D., & Pfleger, B. (2003). Burden of major musculoskeletal conditions. Bulletin of the World Health Organization, 81, 646–656.
World Health Organization (2007) Workers’ health: Global plan of action Geneva: World Health Organization, http://www.who.int/occupational_health/publications/global_plan/en/
Xin, D. L., et al. (2011). Aging enhances serum cytokine response but not task-induced grip strength declines in a rat model of work-related musculoskeletal disorders. BMC Musculoskeletal Disorders, 12, 63.
Zeichen, J. (2000). The proliferative response of isolated human tendon fibroblasts to cyclic biaxial mechanical strain. The American Journal of Sports Medicine, 28, 888–892.
Acknowledgements
Research reported in this publication was supported by the National Institute of Arthritis and Musculoskeletal and Skin Diseases of the National Institutes of Health under Award Number AR056019 to MFB. This study utilized services offered by core facilities of the Center for Substance Abuse Research (Grant Number DA013429). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Competing Interests
None of the authors have any competing interests to declare.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer International Publishing AG, part of Springer Nature
About this chapter
Cite this chapter
Barbe, M.F., Salvadeo, D.M., Hilliard, B.A. (2018). Heat Shock Protein Response to Overuse Injuries. In: Asea, A., Kaur, P. (eds) Regulation of Heat Shock Protein Responses. Heat Shock Proteins, vol 13. Springer, Cham. https://doi.org/10.1007/978-3-319-74715-6_11
Download citation
DOI: https://doi.org/10.1007/978-3-319-74715-6_11
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-74714-9
Online ISBN: 978-3-319-74715-6
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)