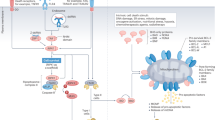
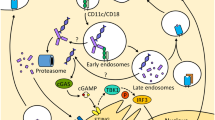
Abstract
TLR3 is a pattern-recognition receptor specialized in detecting extracellular double-stranded RNAs (dsRNAs) that sign the presence of virus. Accordingly, TLR3 triggers a Type I-IFN dominated inflammatory response in immune and nonimmune normal cells. Remarkably, TLR3 behaves also as a death receptor activating the extrinsic pathway of apoptosis in a broad variety of human epithelial cancer cells. In addition, TLR3 is highly expressed by myeloid dendritic cells that undergo maturation in the presence of Poly(I:C), a synthetic dsRNA that mimics viral dsRNA. In particular, the BDCA3+ subset of human dendritic cells that is specialized in antigen cross-presentation express the highest level of TLR3. Therefore, the double activity of TLR3 ligand to trigger apoptosis and inflammation of epithelial cancer cells and to initiate dendritic cells maturation suggests that TLR3 ligand may provide simultaneously the two components required for tumor autovaccination: the tumor-associated antigens released in immunogenic apoptotic bodies and the adjuvant to enhance their cross-presentation by dendritic cells to T cells. Here, we review the rationale for targeting TLR3 in cancer and for its combination with immune-checkpoint inhibitors.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Nagata S (1997) Apoptosis by death factor. Cell 88(3):355–365
Ashkenazi A (2015) Targeting the extrinsic apoptotic pathway in cancer: lessons learned and future directions. J Clin Invest 125(2):487–489
Bell JK, Botos I, Hall PR, Askins J, Shiloach J, Segal DM et al (2005) The molecular structure of the Toll-like receptor 3 ligand-binding domain. Proc Natl Acad Sci U S A 102(31):10976–10980
Liu L, Botos I, Wang Y, Leonard JN, Shiloach J, Segal DM et al (2008) Structural basis of toll-like receptor 3 signaling with double-stranded RNA. Science 320(5874):379–381
Kang JY, Lee J-O (2011) Structural biology of the Toll-like receptor family. Annu Rev Biochem 80:917–941
Mikami T, Miyashita H, Takatsuka S, Kuroki Y, Matsushima N (2012) Molecular evolution of vertebrate Toll-like receptors: evolutionary rate difference between their leucine-rich repeats and their TIR domains. Gene 503(2):235–243
Barreiro LB, Ben-Ali M, Quach H, Laval G, Patin E, Pickrell JK et al (2009) Evolutionary dynamics of human Toll-like receptors and their different contributions to host defense. PLoS Genet 5(7):e1000562
Alexopoulou L, Holt AC, Medzhitov R, Flavell RA (2001) Recognition of double-stranded RNA and activation of NF-kappaB by Toll-like receptor 3. Nature 413(6857):732–738
Yamamoto M, Sato S, Mori K, Hoshino K, Takeuchi O, Takeda K et al (2002) Cutting edge: a novel Toll/IL-1 receptor domain-containing adapter that preferentially activates the IFN-beta promoter in the Toll-like receptor signaling. J Immunol 169(12):6668–6672
Oshiumi H, Matsumoto M, Funami K, Akazawa T, Seya T (2003) TICAM-1, an adaptor molecule that participates in Toll-like receptor 3-mediated interferon-beta induction. Nat Immunol 4(2):161–167
Botos I, Liu L, Wang Y, Segal DM, Davies DR (2009) The toll-like receptor 3:dsRNA signaling complex. Biochim Biophys Acta 1789(9–10):667–674
Wang Y, Liu L, Davies DR, Segal DM (2010) Dimerization of Toll-like receptor 3 (TLR3) is required for ligand binding. J Biol Chem 285(47):36836–36841
Fukuda K, Tsujita T, Matsumoto M, Seya T, Sakiyama H, Nishikawa F et al (2006) Analysis of the interaction between human TLR3 ectodomain and nucleic acids. Nucleic Acids Symp Ser 50(1):249–250
Leonard JN, Ghirlando R, Askins J, Bell JK, Margulies DH, Davies DR et al (2008) The TLR3 signaling complex forms by cooperative receptor dimerization. Proc Natl Acad Sci U S A 105(1):258–263
de Bouteiller O, Merck E, Hasan UA, Hubac S, Benguigui B, Trinchieri G et al (2005) Recognition of double-stranded RNA by human toll-like receptor 3 and downstream receptor signaling requires multimerization and an acidic pH. J Biol Chem 280(46):38133–38145
Muzio M, Bosisio D, Polentarutti N, D’amico G, Stoppacciaro A, Mancinelli R et al (2000) Differential expression and regulation of toll-like receptors (TLR) in human leukocytes: selective expression of TLR3 in dendritic cells. J Immunol 164(11):5998–6004
Visintin A, Mazzoni A, Spitzer JH, Wyllie DH, Dower SK, Segal DM (2001) Regulation of Toll-like receptors in human monocytes and dendritic cells. J Immunol 166(1):249–255
Heinz S, Haehnel V, Karaghiosoff M, Schwarzfischer L, Müller M, Krause SW et al (2003) Species-specific regulation of Toll-like receptor 3 genes in men and mice. J Biol Chem 278(24):21502–21509
Miettinen M, Sareneva T, Julkunen I, Matikainen S (2001) IFNs activate toll-like receptor gene expression in viral infections. Genes Immun 2(6):349–355
Edwards AD, Diebold SS, Slack EMC, Tomizawa H, Hemmi H, Kaisho T et al (2003) Toll-like receptor expression in murine DC subsets: lack of TLR7 expression by CD8 alpha+ DC correlates with unresponsiveness to imidazoquinolines. Eur J Immunol 33(4):827–833
Fujita H, Asahina A, Mitsui H, Tamaki K (2004) Langerhans cells exhibit low responsiveness to double-stranded RNA. Biochem Biophys Res Commun 319(3):832–839
Kulka M, Alexopoulou L, Flavell RA, Metcalfe DD (2004) Activation of mast cells by double-stranded RNA: evidence for activation through Toll-like receptor 3. J Allergy Clin Immunol 114(1):174–182
Tohyama M, Dai X, Sayama K, Yamasaki K, Shirakata Y, Hanakawa Y et al (2005) dsRNA-mediated innate immunity of epidermal keratinocytes. Biochem Biophys Res Commun 335(2):505–511
Abrahams VM, Visintin I, Aldo PB, Guller S, Romero R, Mor G (2005) A role for TLRs in the regulation of immune cell migration by first trimester trophoblast cells. J Immunol 175(12):8096–8104
Pineda A, Verdin-Terán SL, Camacho A, Moreno-Fierros L (2011) Expression of toll-like receptor TLR-2, TLR-3, TLR-4 and TLR-9 is increased in placentas from patients with preeclampsia. Arch Med Res 42(5):382–391
Tanabe M, Kurita-Taniguchi M, Takeuchi K, Takeda M, Ayata M, Ogura H et al (2003) Mechanism of up-regulation of human Toll-like receptor 3 secondary to infection of measles virus-attenuated strains. Biochem Biophys Res Commun 311(1):39–48
Wen L, Peng J, Li Z, Wong FS (2004) The effect of innate immunity on autoimmune diabetes and the expression of Toll-like receptors on pancreatic islets. J Immunol 172(5):3173–3180
Patole PS, Gröne H-J, Segerer S, Ciubar R, Belemezova E, Henger A et al (2005) Viral double-stranded RNA aggravates lupus nephritis through Toll-like receptor 3 on glomerular mesangial cells and antigen-presenting cells. J Am Soc Nephrol 16(5):1326–1338
Young SL, Lyddon TD, Jorgenson RL, Misfeldt ML (2004) Expression of Toll-like receptors in human endometrial epithelial cells and cell lines. Am J Reprod Immunol 52(1):67–73
Schaefer TM, Fahey JV, Wright JA, Wira CR (2005) Innate immunity in the human female reproductive tract: antiviral response of uterine epithelial cells to the TLR3 agonist poly(I:C). J Immunol 174(2):992–1002
Tissari J, Sirén J, Meri S, Julkunen I, Matikainen S (2005) IFN-alpha enhances TLR3-mediated antiviral cytokine expression in human endothelial and epithelial cells by up-regulating TLR3 expression. J Immunol 174(7):4289–4294
Kumar MV, Nagineni CN, Chin MS, Hooks JJ, Detrick B (2004) Innate immunity in the retina: Toll-like receptor (TLR) signaling in human retinal pigment epithelial cells. J Neuroimmunol 153(1–2):7–15
Takii Y, Nakamura M, Ito M, Yokoyama T, Komori A, Shimizu-Yoshida Y et al (2005) Enhanced expression of type I interferon and toll-like receptor-3 in primary biliary cirrhosis. Lab Invest 85(7):908–920
Cario E, Podolsky DK (2000) Differential alteration in intestinal epithelial cell expression of toll-like receptor 3 (TLR3) and TLR4 in inflammatory bowel disease. Infect Immun 68(12):7010–7017
Fransson M, Adner M, Erjefält J, Jansson L, Uddman R, Cardell L-O (2005) Up-regulation of Toll-like receptors 2, 3 and 4 in allergic rhinitis. Respir Res 6:100
Andersen JM, Al-Khairy D, Ingalls RR (2006) Innate immunity at the mucosal surface: role of toll-like receptor 3 and toll-like receptor 9 in cervical epithelial cell responses to microbial pathogens. Biol Reprod 74(5):824–831
Kumar A, Zhang J, Yu F-SX (2006) Toll-like receptor 3 agonist poly(I:C)-induced antiviral response in human corneal epithelial cells. Immunology 117(1):11–21
Wörnle M, Schmid H, Banas B, Merkle M, Henger A, Roeder M et al (2006) Novel role of toll-like receptor 3 in hepatitis C-associated glomerulonephritis. Am J Pathol 168(2):370–385
Brentano F, Schorr O, Gay RE, Gay S, Kyburz D (2005) RNA released from necrotic synovial fluid cells activates rheumatoid arthritis synovial fibroblasts via Toll-like receptor 3. Arthritis Rheum 52(9):2656–2665
Matsumoto M, Kikkawa S, Kohase M, Miyake K, Seya T (2002) Establishment of a monoclonal antibody against human Toll-like receptor 3 that blocks double-stranded RNA-mediated signaling. Biochem Biophys Res Commun 293(5):1364–1369
Bsibsi M, Ravid R, Gveric D, van Noort JM (2002) Broad expression of Toll-like receptors in the human central nervous system. J Neuropathol Exp Neurol 61(11):1013–1021
Farina C, Krumbholz M, Giese T, Hartmann G, Aloisi F, Meinl E (2005) Preferential expression and function of Toll-like receptor 3 in human astrocytes. J Neuroimmunol 159(1–2):12–19
Jackson AC, Rossiter JP, Lafon M (2006) Expression of Toll-like receptor 3 in the human cerebellar cortex in rabies, herpes simplex encephalitis, and other neurological diseases. J Neurovirol 12(3):229–234
Taura M, Eguma A, Suico MA, Shuto T, Koga T, Komatsu K et al (2008) p53 regulates Toll-like receptor 3 expression and function in human epithelial cell lines. Mol Cell Biol 28(21):6557–6567
Taura M, Suico MA, Koyama K, Komatsu K, Miyakita R, Matsumoto C et al (2012) Rb/E2F1 regulates the innate immune receptor Toll-like receptor 3 in epithelial cells. Mol Cell Biol 32(8):1581–1590
Kim Y-M, Brinkmann MM, Paquet M-E, Ploegh HL (2008) UNC93B1 delivers nucleotide-sensing toll-like receptors to endolysosomes. Nature 452(7184):234–238
Toscano F, Estornes Y, Virard F, Garcia-Cattaneo A, Pierrot A, Vanbervliet B et al (2013) Cleaved/associated TLR3 represents the primary form of the signaling receptor. J Immunol 190(2):764–773
Ewald SE, Engel A, Lee J, Wang M, Bogyo M, Barton GM (2011) Nucleic acid recognition by Toll-like receptors is coupled to stepwise processing by cathepsins and asparagine endopeptidase. J Exp Med 208(4):643–651
Yamashita M, Chattopadhyay S, Fensterl V, Saikia P, Wetzel JL, Sen GC (2012) Epidermal growth factor receptor is essential for Toll-like receptor 3 signaling. Sci Signal 5(233):ra50
Johnsen IB, Nguyen TT, Ringdal M, Tryggestad AM, Bakke O, Lien E et al (2006) Toll-like receptor 3 associates with c-Src tyrosine kinase on endosomes to initiate antiviral signaling. EMBO J 25(14):3335–3346
Sarkar SN, Peters KL, Elco CP, Sakamoto S, Pal S, Sen GC (2004) Novel roles of TLR3 tyrosine phosphorylation and PI3 kinase in double-stranded RNA signaling. Nat Struct Mol Biol 11(11):1060–1067
Lee K-G, Xu S, Kang Z-H, Huo J, Huang M, Liu D et al (2012) Bruton’s tyrosine kinase phosphorylates Toll-like receptor 3 to initiate antiviral response. Proc Natl Acad Sci U S A 109(15):5791–5796
Verstak B, Arnot CJ, Gay NJ (2013) An alanine-to-proline mutation in the BB-loop of TLR3 Toll/IL-1R domain switches signalling adaptor specificity from TRIF to MyD88. J Immunol 191(12):6101–6109
Sasai M, Oshiumi H, Matsumoto M, Inoue N, Fujita F, Nakanishi M et al (2005) Cutting edge: NF-kappaB-activating kinase-associated protein 1 participates in TLR3/Toll-IL-1 homology domain-containing adapter molecule-1-mediated IFN regulatory factor 3 activation. J Immunol 174(1):27–30
Oganesyan G, Saha SK, Guo B, He JQ, Shahangian A, Zarnegar B et al (2006) Critical role of TRAF3 in the Toll-like receptor-dependent and -independent antiviral response. Nature 439(7073):208–211
Sharma S, tenOever BR, Grandvaux N, Zhou G-P, Lin R, Hiscott J (2003) Triggering the interferon antiviral response through an IKK-related pathway. Science 300(5622):1148–1151
Fitzgerald KA, McWhirter SM, Faia KL, Rowe DC, Latz E, Golenbock DT et al (2003) IKKepsilon and TBK1 are essential components of the IRF3 signaling pathway. Nat Immunol 4(5):491–496
Ng CT, Mendoza JL, Garcia KC, Oldstone MBA (2016) Alpha and Beta type 1 interferon signaling: passage for diverse biologic outcomes. Cell 164(3):349–352
Ritter M, Mennerich D, Weith A, Seither P (2005) Characterization of Toll-like receptors in primary lung epithelial cells: strong impact of the TLR3 ligand poly(I:C) on the regulation of Toll-like receptors, adaptor proteins and inflammatory response. J Inflamm (Lond) 2:16
Saitoh T, Yamamoto M, Miyagishi M, Taira K, Nakanishi M, Fujita T et al (2005) A20 is a negative regulator of IFN regulatory factor 3 signaling. J Immunol 174(3):1507–1512
Jiang Z, Mak TW, Sen G, Li X (2004) Toll-like receptor 3-mediated activation of NF-kappaB and IRF3 diverges at Toll-IL-1 receptor domain-containing adapter inducing IFN-beta. Proc Natl Acad Sci U S A 101(10):3533–3538
Rydberg C, Månsson A, Uddman R, Riesbeck K, Cardell L-O (2009) Toll-like receptor agonists induce inflammation and cell death in a model of head and neck squamous cell carcinomas. Immunology 128(1 Suppl):e600–e611
Umemura N, Zhu J, Mburu YK, Forero A, Hsieh PN, Muthuswamy R et al (2012) Defective NF-κB signaling in metastatic head and neck cancer cells leads to enhanced apoptosis by dsRNA. Cancer Res 72(1):45–55
Sha Q, Truong-Tran AQ, Plitt JR, Beck LA, Schleimer RP (2004) Activation of airway epithelial cells by toll-like receptor agonists. Am J Respir Cell Mol Biol 31(3):358–364
Perales-Linares R, Navas-Martin S (2013) Toll-like receptor 3 in viral pathogenesis: friend or foe? Immunology 140(2):153–167
Ruckdeschel K, Pfaffinger G, Haase R, Sing A, Weighardt H, Häcker G et al (2004) Signaling of apoptosis through TLRs critically involves toll/IL-1 receptor domain-containing adapter inducing IFN-beta, but not MyD88, in bacteria-infected murine macrophages. J Immunol 173(5):3320–3328
Rasschaert J, Ladrière L, Urbain M, Dogusan Z, Katabua B, Sato S et al (2005) Toll-like receptor 3 and STAT-1 contribute to double-stranded RNA+ interferon-gamma-induced apoptosis in primary pancreatic beta-cells. J Biol Chem 280(40):33984–33991
Dogusan Z, García M, Flamez D, Alexopoulou L, Goldman M, Gysemans C et al (2008) Double-stranded RNA induces pancreatic beta-cell apoptosis by activation of the toll-like receptor 3 and interferon regulatory factor 3 pathways. Diabetes 57(5):1236–1245
Klettner A, Koinzer S, Meyer T, Roider J (2013) Toll-like receptor 3 activation in retinal pigment epithelium cells – mitogen-activated protein kinase pathways of cell death and vascular endothelial growth factor secretion. Acta Ophthalmol 91(3):e211–e218
Sato A, Iizuka M, Nakagomi O, Suzuki M, Horie Y, Konno S et al (2006) Rotavirus double-stranded RNA induces apoptosis and diminishes wound repair in rat intestinal epithelial cells. J Gastroenterol Hepatol 21(3):521–530
Zhou R, Wei H, Sun R, Zhang J, Tian Z (2007) NKG2D recognition mediates Toll-like receptor 3 signaling-induced breakdown of epithelial homeostasis in the small intestines of mice. Proc Natl Acad Sci U S A 104(18):7512–7515
Yaddanapudi K, De Miranda J, Hornig M, Lipkin WI (2011) Toll-like receptor 3 regulates neural stem cell proliferation by modulating the Sonic Hedgehog pathway. PLoS One 6(10):e26766
Jacobs BL, Langland JO (1996) When two strands are better than one: the mediators and modulators of the cellular responses to double-stranded RNA. Virology 219(2):339–349
Tatematsu M, Nishikawa F, Seya T, Matsumoto M (2013) Toll-like receptor 3 recognizes incomplete stem structures in single-stranded viral RNA. Nat Commun 4:1833
Iwakiri D, Zhou L, Samanta M, Matsumoto M, Ebihara T, Seya T et al (2009) Epstein-Barr virus (EBV)–encoded small RNA is released from EBV-infected cells and activates signaling from toll-like receptor 3. J Exp Med 206(10):2091
Zhang S-Y, Herman M, Ciancanelli MJ, Pérez de Diego R, Sancho-Shimizu V, Abel L et al (2013) TLR3 immunity to infection in mice and humans. Curr Opin Immunol 25(1):19–33
Zhang S-Y, Jouanguy E, Ugolini S, Smahi A, Elain G, Romero P et al (2007) TLR3 deficiency in patients with herpes simplex encephalitis. Science 317(5844):1522–1527
Casrouge A, Zhang S-Y, Eidenschenk C, Jouanguy E, Puel A, Yang K et al (2006) Herpes simplex virus encephalitis in human UNC-93B deficiency. Science 314(5797):308–312
Sancho-Shimizu V, Pérez de Diego R, Lorenzo L, Halwani R, Alangari A, Israelsson E et al (2011) Herpes simplex encephalitis in children with autosomal recessive and dominant TRIF deficiency. J Clin Invest 121(12):4889–4902
Pérez de Diego R, Sancho-Shimizu V, Lorenzo L, Puel A, Plancoulaine S, Picard C et al (2010) Human TRAF3 adaptor molecule deficiency leads to impaired Toll-like receptor 3 response and susceptibility to herpes simplex encephalitis. Immunity 33(3):400–411
Herman M, Ciancanelli M, Ou Y-H, Lorenzo L, Klaudel-Dreszler M, Pauwels E et al (2012) Heterozygous TBK1 mutations impair TLR3 immunity and underlie herpes simplex encephalitis of childhood. J Exp Med 209(9):1567–1582
Aksoy E, Zouain CS, Vanhoutte F, Fontaine J, Pavelka N, Thieblemont N et al (2005) Double-stranded RNAs from the helminth parasite Schistosoma activate TLR3 in dendritic cells. J Biol Chem 280(1):277–283
Lathia JD, Okun E, Tang S-C, Griffioen K, Cheng A, Mughal MR et al (2008) Toll-like receptor 3 is a negative regulator of embryonic neural progenitor cell proliferation. J Neurosci 28(51):13978–13984
Karikó K, Ni H, Capodici J, Lamphier M, Weissman D (2004) mRNA is an endogenous ligand for Toll-like receptor 3. J Biol Chem 279(13):12542–12550
Koski GK, Karikó K, Xu S, Weissman D, Cohen PA, Czerniecki BJ (2004) Cutting edge: innate immune system discriminates between RNA containing bacterial versus eukaryotic structural features that prime for high-level IL-12 secretion by dendritic cells. J Immunol 172(7):3989–3993
Karikó K, Buckstein M, Ni H, Weissman D (2005) Suppression of RNA recognition by Toll-like receptors: the impact of nucleoside modification and the evolutionary origin of RNA. Immunity 23(2):165–175
Lee H, Jo E-K, Choi S-Y, Oh SB, Park K, Kim JS et al (2006) Necrotic neuronal cells induce inflammatory Schwann cell activation via TLR2 and TLR3: implication in Wallerian degeneration. Biochem Biophys Res Commun 350(3):742–747
Cavassani KA, Ishii M, Wen H, Schaller MA, Lincoln PM, Lukacs NW et al (2008) TLR3 is an endogenous sensor of tissue necrosis during acute inflammatory events. J Exp Med 205(11):2609–2621
Lin Q, Fang D, Fang J, Ren X, Yang X, Wen F et al (2011) Impaired wound healing with defective expression of chemokines and recruitment of myeloid cells in TLR3-deficient mice. J Immunol 186(6):3710–3717
Shiose S, Chen Y, Okano K, Roy S, Kohno H, Tang J et al (2011) Toll-like receptor 3 is required for development of retinopathy caused by impaired all-trans-retinal clearance in mice. J Biol Chem 286(17):15543–15555
Bernard JJ, Cowing-Zitron C, Nakatsuji T, Muehleisen B, Muto J, Borkowski AW et al (2012) Ultraviolet radiation damages self noncoding RNA and is detected by TLR3. Nat Med 18(8):1286–1290
Hoffman RW, Gazitt T, Foecking MF, Ortmann RA, Misfeldt M, Jorgenson R et al (2004) U1 RNA induces innate immunity signaling. Arthritis Rheum 50(9):2891–2896
Malmegrim KCR, Pruijn GJM, van Venrooij WJ (2002) The fate of the U1 snRNP autoantigen during apoptosis: implications for systemic autoimmunity. Isr Med Assoc J 4(9):706–712
Liu Y, Gu Y, Han Y, Zhang Q, Jiang Z, Zhang X et al (2016) Tumor Exosomal RNAs promote lung pre-metastatic niche formation by activating alveolar epithelial TLR3 to recruit neutrophils. Cancer Cell 30(2):243–256
Kato H, Takeuchi O, Mikamo-Satoh E, Hirai R, Kawai T, Matsushita K et al (2008) Length-dependent recognition of double-stranded ribonucleic acids by retinoic acid-inducible gene-I and melanoma differentiation-associated gene 5. J Exp Med 205(7):1601–1610
Kato H, Takeuchi O, Sato S, Yoneyama M, Yamamoto M, Matsui K et al (2006) Differential roles of MDA5 and RIG-I helicases in the recognition of RNA viruses. Nature 441(7089):101–105
Gitlin L, Barchet W, Gilfillan S, Cella M, Beutler B, Flavell RA et al (2006) Essential role of mda-5 in type I IFN responses to polyriboinosinic:polyribocytidylic acid and encephalomyocarditis picornavirus. Proc Natl Acad Sci U S A 103(22):8459–8464
Levy HB, Baer G, Baron S, Buckler CE, Gibbs CJ, Iadarola MJ et al (1975) A modified polyriboinosinic-polyribocytidylic acid complex that induces interferon in primates. J Infect Dis 132(4):434–439
Gowen BB, Wong M-H, Jung K-H, Sanders AB, Mitchell WM, Alexopoulou L et al (2007) TLR3 is essential for the induction of protective immunity against Punta Toro virus infection by the double-stranded RNA (dsRNA), poly(I:C12U), but not poly(I:C): differential recognition of synthetic dsRNA molecules. J Immunol 178(8):5200–5208
Okahira S, Nishikawa F, Nishikawa S, Akazawa T, Seya T, Matsumoto M (2005) Interferon-beta induction through toll-like receptor 3 depends on double-stranded RNA structure. DNA Cell Biol 24(10):614–623
Jeung H-C, Moon YW, Rha SY, Yoo NC, Roh JK, Noh SH et al (2008) Phase III trial of adjuvant 5-fluorouracil and adriamycin versus 5-fluorouracil, adriamycin, and polyadenylic-polyuridylic acid (poly A:U) for locally advanced gastric cancer after curative surgery: final results of 15-year follow-up. Ann Oncol 19(3):520–526
Salaun B, Zitvogel L, Asselin-Paturel C, Morel Y, Chemin K, Dubois C et al (2011) TLR3 as a biomarker for the therapeutic efficacy of double-stranded RNA in breast cancer. Cancer Res 71(5):1607–1614
Chen L, Xu Y-Y, Zhou J-M, Wu Y-Y, E Q, Zhu Y-Y (2012) TLR3 dsRNA agonist inhibits growth and invasion of HepG2.2.15 HCC cells. Oncol Rep 28(1):200–206
Xu Y-Y, Chen L, Zhou J-M, Wu Y-Y, Zhu Y-Y (2013) Inhibitory effect of dsRNA TLR3 agonist in a rat hepatocellular carcinoma model. Mol Med Rep 8(4):1037–1042
Matsumoto M, Tatematsu M, Nishikawa F, Azuma M, Ishii N, Morii-Sakai A et al (2015) Defined TLR3-specific adjuvant that induces NK and CTL activation without significant cytokine production in vivo. Nat Commun 6:6280
Seya T, Takeda Y, Matsumoto M (2016) Tumor vaccines with dsRNA adjuvant ARNAX induces antigen-specific tumor shrinkage without cytokinemia. Oncoimmunology 5(2):e1043506
Naumann K, Wehner R, Schwarze A, Petzold C, Schmitz M, Rohayem J (2013) Activation of dendritic cells by the novel Toll-like receptor 3 agonist RGC100. Clin Dev Immunol 2013:283649
Duffy KE, Lamb RJ, San Mateo LR, Jordan JL, Canziani G, Brigham-Burke M et al (2007) Down modulation of human TLR3 function by a monoclonal antibody. Cell Immunol 248(2):103–114
Levine AS, Levy HB (1978) Phase I-II trials of poly IC stabilized with poly-L-lysine. Cancer Treat Rep 62(11):1907–1912
Lampkin BC, Levine AS, Levy H, Krivit W, Hammond D (1985) Phase II trial of a complex polyriboinosinic-polyribocytidylic acid with poly-L-lysine and carboxymethyl cellulose in the treatment of children with acute leukemia and neuroblastoma: a report from the Children’s Cancer Study Group. Cancer Res 45(11 Pt 2):5904–5909
Seya T, Azuma M, Matsumoto M (2013) Targeting TLR3 with no RIG-I/MDA5 activation is effective in immunotherapy for cancer. Expert Opin Ther Targets 17(5):533–544
Vérillaud B, Gressette M, Morel Y, Paturel C, Herman P, Lo KW et al (2012) Toll-like receptor 3 in Epstein-Barr virus-associated nasopharyngeal carcinomas: consistent expression and cytotoxic effects of its synthetic ligand poly(A:U) combined to a Smac-mimetic. Infect Agent Cancer 7(1):36
Hasimu A, Ge L, Li Q-Z, Zhang R-P, Guo X (2011) Expressions of Toll-like receptors 3, 4, 7, and 9 in cervical lesions and their correlation with HPV16 infection in Uighur women. Chin J Cancer 30(5):344–350
Luo Q, Hu S, Yan M, Sun Z, Chen W, Chen F (2012) Activation of Toll-like receptor 3 induces apoptosis of oral squamous carcinoma cells in vitro and in vivo. Int J Biochem Cell Biol 44(8):1266–1275
He Z, Huang X, Ni Y, Shi P, Wang Z, Han W et al (2014) Functional toll-like receptor 3 expressed by oral squamous cell carcinoma induced cell apoptosis and decreased migration. Oral Surg Oral Med Oral Pathol Oral Radiol 118(1):92–100
Chuang H-C, Huang C-C, Chien C-Y, Chuang J-H (2012) Toll-like receptor 3-mediated tumor invasion in head and neck cancer. Oral Oncol 48(3):226–232
Nojiri K, Sugimoto K, Shiraki K, Tameda M, Inagaki Y, Kusagawa S et al (2013) The expression and function of Toll-like receptors 3 and 9 in human colon carcinoma. Oncol Rep 29(5):1737–1743
Chew V, Tow C, Huang C, Bard-Chapeau E, Copeland NG, Jenkins NA et al (2012) Toll-like receptor 3 expressing tumor parenchyma and infiltrating natural killer cells in hepatocellular carcinoma patients. J Natl Cancer Inst 104(23):1796–1807
Yuan M-M, Xu Y-Y, Chen L, Li X-Y, Qin J, Shen Y (2015) TLR3 expression correlates with apoptosis, proliferation and angiogenesis in hepatocellular carcinoma and predicts prognosis. BMC Cancer 15:245
Li D, Gu R, Yang X, Hu C, Li Y, Gao M et al (2014) TLR3 correlated with cervical lymph node metastasis in patients with papillary thyroid cancer. Int J Clin Exp Med 7(12):5111
Gambara G, Desideri M, Stoppacciaro A, Padula F, Cesaris PD, Starace D et al (2015) TLR3 engagement induces IRF-3-dependent apoptosis in androgen-sensitive prostate cancer cells and inhibits tumour growth in vivo. J Cell Mol Med 19(2):327
Zhou M, McFarland-Mancini MM, Funk HM, Husseinzadeh N, Mounajjed T, Drew AF (2009) Toll-like receptor expression in normal ovary and ovarian tumors. Cancer Immunol Immunother 58(9):1375–1385
Helminen O, Huhta H, Lehenkari PP, Saarnio J, Karttunen TJ, Kauppila JH (2016) Nucleic acid-sensing toll-like receptors 3, 7 and 8 in esophageal epithelium, barrett’s esophagus, dysplasia and adenocarcinoma. Oncoimmunology 5(5):e1127495
Combaz-Lair C, Galateau-Sallé F, McLeer-Florin A, Le Stang N, David-Boudet L, Duruisseaux M et al (2016) Immune biomarkers PD-1/PD-L1 and TLR3 in malignant pleural mesotheliomas. Hum Pathol 52:9–18
Allhorn S, Böing C, Koch AA, Kimmig R, Gashaw I (2008) TLR3 and TLR4 expression in healthy and diseased human endometrium. Reprod Biol Endocrinol 6:40
Hasan UA, Caux C, Perrot I, Doffin AC, Menetrier-Caux C, Trinchieri G, Tommasino M, Vlach J (2007) Cell proliferation and survival induced by Toll-like receptors is antagonized by type I IFNs. Proc Natl Acad Sci U S A 104(19):8047–8052
Salaun B, Coste I, Rissoan M-C, Lebecque SJ, Renno T (2006) TLR3 can directly trigger apoptosis in human cancer cells. J Immunol 176(8):4894–4901
Paone A, Starace D, Galli R, Padula F, De Cesaris P, Filippini A et al (2008) Toll-like receptor 3 triggers apoptosis of human prostate cancer cells through a PKC-alpha-dependent mechanism. Carcinogenesis 29(7):1334–1342
Nomi N, Kodama S, Suzuki M (2010) Toll-like receptor 3 signaling induces apoptosis in human head and neck cancer via survivin associated pathway. Oncol Rep 24(1):225–231
Hughes MA, Harper N, Butterworth M, Cain K, Cohen GM, MacFarlane M (2009) Reconstitution of the death-inducing signaling complex reveals a substrate switch that determines CD95-mediated death or survival. Mol Cell 35(3):265–279
Oberst A, Pop C, Tremblay AG, Blais V, Denault J-B, Salvesen GS et al (2010) Inducible dimerization and inducible cleavage reveal a requirement for both processes in caspase-8 activation. J Biol Chem 285(22):16632–16642
Dickens LS, Boyd RS, Jukes-Jones R, Hughes MA, Robinson GL, Fairall L et al (2012) A death effector domain chain DISC model reveals a crucial role for caspase-8 chain assembly in mediating apoptotic cell death. Mol Cell 47(2):291–305
Schleich K, Warnken U, Fricker N, Oztürk S, Richter P, Kammerer K et al (2012) Stoichiometry of the CD95 death-inducing signaling complex: experimental and modeling evidence for a death effector domain chain model. Mol Cell 47(2):306–319
Estornes Y, Toscano F, Virard F, Jacquemin G, Pierrot A, Vanbervliet B et al (2012) dsRNA induces apoptosis through an atypical death complex associating TLR3 to caspase-8. Cell Death Differ 19(9):1482–1494
Feoktistova M, Geserick P, Kellert B, Dimitrova DP, Langlais C, Hupe M et al (2011) cIAPs block Ripoptosome formation, a RIP1/caspase-8 containing intracellular cell death complex differentially regulated by cFLIP isoforms. Mol Cell 43(3):449–463
Kaiser WJ, Offermann MK (2005) Apoptosis induced by the toll-like receptor adaptor TRIF is dependent on its receptor interacting protein homotypic interaction motif. J Immunol 174(8):4942–4952
Weiss R, Sachet M, Zinngrebe J, Aschacher T, Krainer M, Hegedus B et al (2013) IL-24 sensitizes tumor cells to TLR3-mediated apoptosis. Cell Death Differ 20(6):823–833
Tenev T, Bianchi K, Darding M, Broemer M, Langlais C, Wallberg F et al (2011) The Ripoptosome, a signaling platform that assembles in response to genotoxic stress and loss of IAPs. Mol Cell 43(3):432–448
Friboulet L, Pioche-Durieu C, Rodriguez S, Valent A, Souquère S, Ripoche H et al (2008) Recurrent overexpression of c-IAP2 in EBV-associated nasopharyngeal carcinomas: critical role in resistance to Toll-like receptor 3-mediated apoptosis. Neoplasia 10(11):1183–1194
Weber A, Kirejczyk Z, Besch R, Potthoff S, Leverkus M, Häcker G (2010) Proapoptotic signalling through Toll-like receptor-3 involves TRIF-dependent activation of caspase-8 and is under the control of inhibitor of apoptosis proteins in melanoma cells. Cell Death Differ 17(6):942–951
O’Donnell MA, Legarda-Addison D, Skountzos P, Yeh WC, Ting AT (2007) Ubiquitination of RIP1 regulates an NF-kappaB-independent cell-death switch in TNF signaling. Curr Biol 17(5):418–424
Varfolomeev E, Goncharov T, Fedorova AV, Dynek JN, Zobel K, Deshayes K et al (2008) c-IAP1 and c-IAP2 are critical mediators of tumor necrosis factor alpha (TNFalpha)-induced NF-kappaB activation. J Biol Chem 283(36):24295–24299
Boatright KM, Deis C, Denault J-B, Sutherlin DP, Salvesen GS (2004) Activation of caspases-8 and -10 by FLIP(L). Biochem J 382(Pt 2):651–657
Pop C, Oberst A, Drag M, Van Raam BJ, Riedl SJ, Green DR et al (2011) FLIP(L) induces caspase 8 activity in the absence of interdomain caspase 8 cleavage and alters substrate specificity. Biochem J 433(3):447–457
Salaun B, Lebecque S, Matikainen S, Rimoldi D, Romero P (2007) Toll-like receptor 3 expressed by melanoma cells as a target for therapy? Clin Cancer Res 13(15 Pt 1):4565–4574
Jiang Q, Wei H, Tian Z (2008) Poly I:C enhances cycloheximide-induced apoptosis of tumor cells through TLR3 pathway. BMC Cancer 8:12
Zinngrebe J, Rieser E, Taraborrelli L, Peltzer N, Hartwig T, Ren H et al (2016) LUBAC deficiency perturbs TLR3 signaling to cause immunodeficiency and autoinflammation. J Exp Med 213(12):2671–2689
McCormick KD, Ghosh A, Trivedi S, Wang L, Coyne CB, Ferris RL et al (2016) Innate immune signaling through differential RIPK1 expression promote tumor progression in head and neck squamous cell carcinoma. Carcinogenesis 37(5):522–529
Cho YS, Challa S, Moquin D, Genga R, Ray TD, Guildford M et al (2009) Phosphorylation-driven assembly of the RIP1-RIP3 complex regulates programmed necrosis and virus-induced inflammation. Cell 137(6):1112–1123
He S, Wang L, Miao L, Wang T, Du F, Zhao L et al (2009) Receptor interacting protein kinase-3 determines cellular necrotic response to TNF-alpha. Cell 137(6):1100–1111
Zhang D-W, Shao J, Lin J, Zhang N, Lu B-J, Lin S-C et al (2009) RIP3, an energy metabolism regulator that switches TNF-induced cell death from apoptosis to necrosis. Science 325(5938):332–336
Vanden Berghe T, Linkermann A, Jouan-Lanhouet S, Walczak H, Vandenabeele P (2014) Regulated necrosis: the expanding network of non-apoptotic cell death pathways. Nat Rev Mol Cell Biol 15(2):135–147
Chen W, Zhou Z, Li L, Zhong C-Q, Zheng X, Wu X et al (2013) Diverse sequence determinants control human and mouse receptor interacting protein 3 (RIP3) and mixed lineage kinase domain-like (MLKL) interaction in necroptotic signaling. J Biol Chem 288(23):16247–16261
Cai Z, Jitkaew S, Zhao J, Chiang H-C, Choksi S, Liu J et al (2014) Plasma membrane translocation of trimerized MLKL protein is required for TNF-induced necroptosis. Nat Cell Biol 16(1):55–65
Dondelinger Y, Declercq W, Montessuit S, Roelandt R, Goncalves A, Bruggeman I et al (2014) MLKL compromises plasma membrane integrity by binding to phosphatidylinositol phosphates. Cell Rep 7(4):971–981
Hildebrand JM, Tanzer MC, Lucet IS, Young SN, Spall SK, Sharma P et al (2014) Activation of the pseudokinase MLKL unleashes the four-helix bundle domain to induce membrane localization and necroptotic cell death. Proc Natl Acad Sci U S A 111(42):15072–15077
He S, Liang Y, Shao F, Wang X (2011) Toll-like receptors activate programmed necrosis in macrophages through a receptor-interacting kinase-3-mediated pathway. Proc Natl Acad Sci U S A 108(50):20054–20059
Kaiser WJ, Sridharan H, Huang C, Mandal P, Upton JW, Gough PJ et al (2013) Toll-like receptor 3-mediated necrosis via TRIF, RIP3, and MLKL. J Biol Chem 288(43):31268–31279
Kim SJ, Li J (2013) Caspase blockade induces RIP3-mediated programmed necrosis in Toll-like receptor-activated microglia. Cell Death Dis 4:e716
Mandal P, Berger SB, Pillay S, Moriwaki K, Huang C, Guo H et al (2014) RIP3 induces apoptosis independent of pronecrotic kinase activity. Mol Cell 56(4):481–495
Takemura R, Takaki H, Okada S, Shime H, Akazawa T, Oshiumi H et al (2015) PolyI:C-induced, TLR3/RIP3-dependent Necroptosis backs up immune effector-mediated tumor elimination in vivo. Cancer Immunol Res 3(8):902–914
Meylan E, Burns K, Hofmann K, Blancheteau V, Martinon F, Kelliher M et al (2004) RIP1 is an essential mediator of Toll-like receptor 3-induced NF-kappa B activation. Nat Immunol 5(5):503–507
Degterev A, Hitomi J, Germscheid M, Ch’en IL, Korkina O, Teng X et al (2008) Identification of RIP1 kinase as a specific cellular target of necrostatins. Nat Chem Biol 4(5):313–321
Koo G-B, Morgan MJ, Lee D-G, Kim W-J, Yoon J-H, Koo JS et al (2015) Methylation-dependent loss of RIP3 expression in cancer represses programmed necrosis in response to chemotherapeutics. Cell Res 25(6):707–725
Geserick P, Wang J, Schilling R, Horn S, Harris PA, Bertin J et al (2015) Absence of RIPK3 predicts necroptosis resistance in malignant melanoma. Cell Death Dis 6:e1884
Bondhopadhyay B, Moirangthem A, Basu A (2015) Innate adjuvant receptor Toll-like receptor 3 can promote breast cancer through cell surface. Tumour Biol 36(2):1261–1271
Pries R, Hogrefe L, Xie L, Frenzel H, Brocks C, Ditz C et al (2008) Induction of c-Myc-dependent cell proliferation through toll-like receptor 3 in head and neck cancer. Int J Mol Med 21(2):209–215
Chiron D, Pellat-Deceunynck C, Amiot M, Bataille R, Jego G (2009) TLR3 ligand induces NF-{kappa}B activation and various fates of multiple myeloma cells depending on IFN-{alpha} production. J Immunol 182(7):4471–4478
Matijevic T, Pavelic J (2011) The dual role of TLR3 in metastatic cell line. Clin Exp Metastasis 28(7):701–712
Conforti R, Ma Y, Morel Y, Paturel C, Terme M, Viaud S et al (2010) Opposing effects of toll-like receptor (TLR3) signaling in tumors can be therapeutically uncoupled to optimize the anticancer efficacy of TLR3 ligands. Cancer Res 70(2):490–500
Taura M, Fukuda R, Suico MA, Eguma A, Koga T, Shuto T et al (2010) TLR3 induction by anticancer drugs potentiates poly I:C-induced tumor cell apoptosis. Cancer Sci 101(7):1610–1617
Menendez D, Lowe JM, Snipe J, Resnick MA (2016) Ligand dependent restoration of human TLR3 signaling and death in p53 mutant cells. Oncotarget 7(38):61630–61642
Ultimo A, Giménez C, Bartovsky P, Aznar E, Sancenón F, Marcos MD et al (2016) Targeting innate immunity with dsRNA-conjugated Mesoporous silica nanoparticles promotes antitumor effects on breast cancer cells. Chemistry 22(5):1582–1586
Park J-H, Jeon D-I, Yoon H-E, Kwon S-M, Kim S-A, Ahn S-G et al (2012) Poly I:C inhibits cell proliferation and enhances the growth inhibitory effect of paclitaxel in oral sqaumous cell carcinoma. Acta Odontol Scand 70(3):241–245
Matijević T, Kirinec G, Pavelić J (2011) Antitumor activity from the combined application of poly(I:C) and chemotherapeutics in human metastatic pharyngeal cell lines. Chemotherapy 57(6):460–467
Le UM, Yanasarn N, Löhr CV, Fischer KA, Cui Z (2008) Tumor chemo-immunotherapy using gemcitabine and a synthetic dsRNA. Cancer Biol Ther 7(3):440–447
Van DN, Roberts CF, Marion JD, Lépine S, Harikumar KB, Schreiter J et al (2012) Innate immune agonist, dsRNA, induces apoptosis in ovarian cancer cells and enhances the potency of cytotoxic chemotherapeutics. FASEB J 26(8):3188–3198
Li L, Thomas RM, Suzuki H, De Brabander JK, Wang X, Harran PG (2004) A small molecule Smac mimic potentiates TRAIL- and TNFalpha-mediated cell death. Science 305(5689):1471–1474
Brands RC, Herbst F, Hartmann S, Seher A, Linz C, Kübler AC et al (2016) Cytotoxic effects of SMAC-mimetic compound LCL161 in head and neck cancer cell lines. Clin Oral Investig 20(9):2325–2332
Friboulet L, Gourzones C, Tsao SW, Morel Y, Paturel C, Témam S et al (2010) Poly(I:C) induces intense expression of c-IAP2 and cooperates with an IAP inhibitor in induction of apoptosis in cancer cells. BMC Cancer 10:327
Bernardo AR, Cosgaya JM, Aranda A, Jiménez-Lara AM (2013) Synergy between RA and TLR3 promotes type I IFN-dependent apoptosis through upregulation of TRAIL pathway in breast cancer cells. Cell Death Dis 4:e479
Ming Lim C, Stephenson R, Salazar AM, Ferris RL (2013) TLR3 agonists improve the immunostimulatory potential of cetuximab against EGFR(+) head and neck cancer cells. Oncoimmunology. 2(6):e24677
Xu Y-Y, Chen L, Wang G-L, Zhou J-M, Zhang Y-X, Wei Y-Z et al (2013) A synthetic dsRNA, as a TLR3 pathwaysynergist, combined with sorafenib suppresses HCC in vitro and in vivo. BMC Cancer 13:527
Ho V, Lim TS, Lee J, Steinberg J, Szmyd R, Tham M et al (2015) TLR3 agonist and Sorafenib combinatorial therapy promotes immune activation and controls hepatocellular carcinoma progression. Oncotarget 6(29):27252–27266
Ayari C, Besançon M, Bergeron A, LaRue H, Bussières V, Fradet Y (2016) Poly(I:C) potentiates Bacillus Calmette-Guérin immunotherapy for bladder cancer. Cancer Immunol Immunother 65(2):223–234
Le UM, Kaurin DGL, Sloat BR, Yanasarn N, Cui Z (2009) Localized irradiation of tumors prior to synthetic dsRNA therapy enhanced the resultant anti-tumor activity. Radiother Oncol 90(2):273–279
Nagato T, Lee Y-R, Harabuchi Y, Celis E (2014) Combinatorial immunotherapy of polyinosinic-polycytidylic acid and blockade of programmed death-ligand 1 induce effective CD8 T-cell responses against established tumors. Clin Cancer Res 20(5):1223–1234
Trumpfheller C, Longhi MP, Caskey M, Idoyaga J, Bozzacco L, Keler T et al (2012) Dendritic cell-targeted protein vaccines: a novel approach to induce T-cell immunity. J Intern Med 271(2):183–192
Jongbloed SL, Kassianos AJ, McDonald KJ, Clark GJ, Ju X, Angel CE et al (2010) Human CD141+ (BDCA-3)+ dendritic cells (DCs) represent a unique myeloid DC subset that cross-presents necrotic cell antigens. J Exp Med 207(6):1247–1260
Lauterbach H, Bathke B, Gilles S, Traidl-Hoffmann C, Luber CA, Fejer G et al (2010) Mouse CD8alpha+ DCs and human BDCA3+ DCs are major producers of IFN-lambda in response to poly IC. J Exp Med 207(12):2703–2717
Azuma M, Takeda Y, Nakajima H, Sugiyama H, Ebihara T, Oshiumi H et al (2016) Biphasic function of TLR3 adjuvant on tumor and spleen dendritic cells promotes tumor T cell infiltration and regression in a vaccine therapy. Oncoimmunology. 5(8):e1188244
Forte G, Rega A, Morello S, Luciano A, Arra C, Pinto A et al (2012) Polyinosinic-polycytidylic acid limits tumor outgrowth in a mouse model of metastatic lung cancer. J Immunol 188(11):5357–5364
Salem ML, Kadima AN, Cole DJ, Gillanders WE (2005) Defining the antigen-specific T-cell response to vaccination and poly(I:C)/TLR3 signaling: evidence of enhanced primary and memory CD8 T-cell responses and antitumor immunity. J Immunother 28(3):220–228
Trumpfheller C, Caskey M, Nchinda G, Longhi MP, Mizenina O, Huang Y et al (2008) The microbial mimic poly IC induces durable and protective CD4+ T cell immunity together with a dendritic cell targeted vaccine. Proc Natl Acad Sci U S A 105(7):2574–2579
Forghani P, Waller EK (2015) Poly (I: C) modulates the immunosuppressive activity of myeloid-derived suppressor cells in a murine model of breast cancer. Breast Cancer Res Treat 153(1):21–30
Shime H, Matsumoto M, Oshiumi H, Tanaka S, Nakane A, Iwakura Y et al (2012) Toll-like receptor 3 signaling converts tumor-supporting myeloid cells to tumoricidal effectors. Proc Natl Acad Sci U S A 109(6):2066–2071
Diamond MS, Kinder M, Matsushita H, Mashayekhi M, Dunn GP, Archambault JM et al (2011) Type I interferon is selectively required by dendritic cells for immune rejection of tumors. J Exp Med 208(10):1989–2003
Curtsinger JM, Valenzuela JO, Agarwal P, Lins D, Mescher MF (2005) Type I IFNs provide a third signal to CD8 T cells to stimulate clonal expansion and differentiation. J Immunol 174(8):4465–4469
Zitvogel L, Galluzzi L, Kepp O, Smyth MJ, Kroemer G (2015) Type I interferons in anticancer immunity. Nat Rev Immunol 15(7):405–414
Schulz O, Diebold SS, Chen M, Näslund TI, Nolte MA, Alexopoulou L et al (2005) Toll-like receptor 3 promotes cross-priming to virus-infected cells. Nature 433(7028):887–892
Boes M, Meyer-Wentrup F (2015) TLR3 triggering regulates PD-L1 (CD274) expression in human neuroblastoma cells. Cancer Lett 361(1):49–56
Chiappinelli KB, Strissel PL, Desrichard A, Li H, Henke C, Akman B et al (2015) Inhibiting DNA methylation causes an interferon response in cancer via dsRNA including endogenous retroviruses. Cell 162(5):974–986
Iribarren K, Bloy N, Buqué A, Cremer I, Eggermont A, Fridman WH et al (2016) Trial watch: Immunostimulation with Toll-like receptor agonists in cancer therapy. Oncoimmunology. 5(3):e1088631
Shime H, Matsumoto M, Seya T (2016) Double-stranded RNA promotes CTL-independent tumor cytolysis mediated by CD11b(+)Ly6G(+) intratumor myeloid cells through the TICAM-1 signaling pathway. Cell Death Differ 24(3):385–396
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer International Publishing AG
About this chapter
Cite this chapter
Estornes, Y., Renno, T., Lebecque, S. (2017). TLR3 Is a Death Receptor Target in Cancer Therapy. In: Micheau, O. (eds) TRAIL, Fas Ligand, TNF and TLR3 in Cancer. Resistance to Targeted Anti-Cancer Therapeutics, vol 12. Springer, Cham. https://doi.org/10.1007/978-3-319-56805-8_8
Download citation
DOI: https://doi.org/10.1007/978-3-319-56805-8_8
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-56804-1
Online ISBN: 978-3-319-56805-8
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)