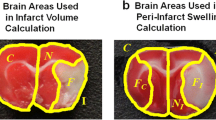
Abstract
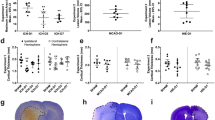
The primary measure for experimental stroke studies, infarct volume, can be affected by brain swelling. The algorithm by Lin et al. was developed to correct for brain swelling, however, the correction is not adequate. This chapter presents a new infarct volume algorithm that more appropriately corrects for brain hemisphere volume changes (swelling and stunted growth). Fifty-one adult rats were sacrificed 24 h after middle cerebral artery occlusion (MCAO). Forty-four P10 rat pups were sacrificed 48 h after hypoxia-ischemia (HI). Infarct volumes for 2,3,5-triphenyl-2H-tetrazolium chloride (TTC) stained brains were calculated using our algorithm and that of Lin and colleagues. For MCAO animals, the algorithm of Lin et al. computed smaller infarct volumes than those of our algorithm. For HI animals, Lin et al.’s algorithm’s infarct volumes were greater than those of our algorithm. For sham animals, Lin et al.’s algorithm’s computed infarct volumes were significantly different from those of our algorithm. Our algorithm produces a more robust estimation of infarct volume than Lin et al.’s algorithm because the effects of ipsilesional hemisphere volume changes are minimized. Herein, our algorithm yields an infarct volume that better corrects for brain swelling and stunted brain growth compared with the algorithm of Lin et al.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Bederson JB, Pitts LH, Germano SM, Nishimura MC, Davis RL, Bartkowski HM (1986) Evaluation of 2,3,5-triphenyltetrazolium chloride as a stain for detection and quantification of experimental cerebral infarction in rats. Stroke 17:1304–1308
Bederson JB, Pitts LH, Tsuji M, Nishimura MC, Davis RL, Bartkowski H (1986) Rat middle cerebral artery occlusion: evaluation of the model and development of a neurologic examination. Stroke 17:472–476
Belayev L, Khoutorova L, Deisher TA, Belayev A, Busto R, Zhang Y, Zhao W, Ginsberg MD (2003) Neuroprotective effect of SolCD39, a novel platelet aggregation inhibitor, on transient middle cerebral artery occlusion in rats. Stroke 34:758–763
Bhattacharya P, Pandey AK, Paul S, Patnaik R, Yavagal DR (2013) Aquaporin-4 inhibition mediates piroxicam-induced neuroprotection against cerebral ischemia/reperfusion injury in rodents. PLoS One 8, e73481
Calvert JW, Cahill J, Yamaguchi-Okada M, Zhang JH (2006) Oxygen treatment after experimental hypoxia-ischemia in neonatal rats alters the expression of HIF-1 alpha and its downstream target genes. J Appl Physiol 101:853–865
Carano RA, Li F, Irie K, Helmer KG, Silva MD, Fisher M, Sotak CH (2000) Multispectral analysis of the temporal evolution of cerebral ischemia in the rat brain. J Magn Reson Imaging 12:842–858
Chen H, Burris M, Fajilan A, Spangnoli F, Tang J, Zhang JH (2011) Prolonged exposure to isoflurane ameliorates infarction severity in the rat pup model of neonatal hypoxia-ischemia. Transl Stroke Res 2011:382–390
Chen ST, Hsu CY, Hogan EL, Juan HY, Banik NL, Balentine JD (1987) Brain calcium content in ischemic infarction. Neurology 37:1227–1229
Chen ST, Hsu CY, Hogan EL, Maricq H, Balentine JD (1986) A model of focal ischemic stroke in the rat: reproducible extensive cortical infarction. Stroke 17:738–743
Duverger D, MacKenzie ET (1988) The quantification of cerebral infarction following focal ischemia in the rat: influence of strain, arterial pressure, blood glucose concentration, and age. J Cereb Blood Flow Metab 8:449–461
Fabian R, Kent T (2012) Hyperglycemia accentuates persistent “functional uncoupling” of cerebral microvascular nitric oxide and superoxide following focal ischemia/reperfusion in rats. Transl Stroke Res 3:482–490
Foley LM, Hitchens TK, Barbe B, Zhang F, Ho C, Rao GR, Nemoto EM (2010) Quantitative temporal profiles of penumbra and infarction during permanent middle cerebral artery occlusion in rats. Transl Stroke Res 1:220–229
Garcia JH (1984) Experimental ischemic stroke: a review. Stroke 15:5–14
Gartshore G, Patterson J, Macrae IM (1997) Influence of ischemia and reperfusion on the course of brain tissue swelling and blood–brain barrier permeability in a rodent model of transient focal cerebral ischemia. Exp Neurol 147:353–360
Hafez S, Coucha M, Bruno A, Fagan SC, Ergul A (2014) Hyperglycemia, acute ischemic stroke, and thrombolytic therapy. Transl Stroke Res 5:442–453
Harvey J, Rasmussen T (1951) Occlusion of the middle cerebral artery: an experimental study. Arch Neurol Psychiatry 66:20–29
Hoff JT, Nishimura M, Newfield P (1982) Pentobarbital protection from cerebral infarction without suppression of edema. Stroke 13:623–628
Jiang Q, Chopp M, Zhang ZG, Knight RA, Jacobs M, Windham JP, Peck D, Ewing JR, Welch KM (1997) The temporal evolution of MRI tissue signatures after transient middle cerebral artery occlusion in rat. J Neurol Sci 145:15–23
Jones PG, Coyle P (1984) Microcomputer assisted lesion size measurements in spontaneously hypertensive stroke-prone rats. J Electrophysiol Tech 11:71–78
Kawamura S, Yasui N, Shirasawa M, Fukasawa H (1991) Rat middle cerebral artery occlusion using an intraluminal thread technique. Acta Neurochir 109:126–132
Khanna A, Kahle KT, Walcott BP, Gerzanich V, Simard JM (2014) Disruption of ion homeostasis in the neurogliovascular unit underlies the pathogenesis of ischemic cerebral edema. Transl Stroke Res 5:3–16
Kondo T, Reaume AG, Huang TT, Carlson E, Murakami K, Chen SF, Hoffman EK, Scott RW, Epstein CJ, Chan PH (1997) Reduction of CuZn-superoxide dismutase activity exacerbates neuronal cell injury and edema formation after transient focal cerebral ischemia. J Neurosci 17:4180–4189
Lee J, Lee JK, Han K (2011) InfarctSizer: computing infarct volume from brain images of a stroke animal model. Comput Methods Biomech Biomed Engin 14:497–504
Lin TN, He YY, Wu G, Khan M, Hsu CY (1993) Effect of brain edema on infarct volume in a focal cerebral ischemia model in rats. Stroke 24:117–121
Lundy EF, Solik BS, Frank RS, Lacy PS, Combs DJ, Zelenock GB, D’Alecy LG (1986) Morphometric evaluation of brain infarcts in rats and gerbils. J Pharmacol Methods 16:201–214
Mandava P, Martini SR, Munoz M, Dalmeida W, Sarma AK, Anderson JA, Fabian RH, Kent TA (2014) Hyperglycemia worsens outcome after rt-PA primarily in the large-vessel occlusive stroke subtype. Transl Stroke Res 5:519–525
Meyer JS, Denny-Brown D (1957) The cerebral collateral circulation. I. Factors influencing collateral blood flow. Neurology 7:447–458
Nemoto E, Mendez O, Kerr M, Firlik A, Stevenson K, Jovin T, Yonas H (2012) CT density changes with rapid onset acute, severe, focal cerebral ischemia in monkeys. Transl Stroke Res 3:369–374
Osborne KA, Shigeno T, Balarsky AM, Ford I, McCulloch J, Teasdale GM, Graham DI (1987) Quantitative assessment of early brain damage in a rat model of focal cerebral ischaemia. J Neurol Neurosurg Psychiatry 50:402–410
Rice JE, Vannucci RC, Brierley JB (1981) The influence of immaturity on hypoxic-ischemic brain-damage in the rat. Ann Neurol 9:131–141
Robinson RG, Shoemaker WJ, Schlumpf M, Valk T, Bloom FE (1975) Effect of experimental cerebral infarction in rat brain on catecholamines and behaviour. Nature 255:332–334
Shimamura N, Matsuda N, Kakuta K, Narita A, Ohkuma H (2013) A model of rat embolic cerebral infarction with a quantifiable, autologous arterial blood clot. Transl Stroke Res 4:564–570
Song M, Yu SP (2014) Ionic regulation of cell volume changes and cell death after ischemic stroke. Transl Stroke Res 5:17–27
Sundt TM Jr, Waltz AG (1966) Experimental cerebral infarction: retro-orbital, extradural approach for occluding the middle cerebral artery. Mayo Clin Proc 41:159–168
Swanson RA, Morton MT, Tsao-Wu G, Savalos RA, Davidson C, Sharp FR (1990) A semiautomated method for measuring brain infarct volume. J Cereb Blood Flow Metab 10:290–293
Tajiri N, Dailey T, Metcalf C, Mosley Y, Lau T, Staples M, van Loveren H, Kim S, Yamashima T, Yasuhara T, Date I, Kaneko Y, Borlongan C (2013) In vivo animal stroke models. Transl Stroke Res 4:308–321
Watson BD, Dietrich WD, Busto R, Wachtel MS, Ginsberg MD (1985) Induction of reproducible brain infarction by photochemically initiated thrombosis. Ann Neurol 17:497–504
Acknowledgments
This work was supported by NIH R01 NS043338 grant (J.H.Z.). The authors thank Xiping Liang and Brandon Dixon.
Disclosure
The authors have no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 Springer International Publishing Switzerland
About this chapter
Cite this chapter
McBride, D.W., Tang, J., Zhang, J.H. (2016). Development of an Infarct Volume Algorithm to Correct for Brain Swelling After Ischemic Stroke in Rats. In: Applegate, R., Chen, G., Feng, H., Zhang, J. (eds) Brain Edema XVI. Acta Neurochirurgica Supplement, vol 121. Springer, Cham. https://doi.org/10.1007/978-3-319-18497-5_18
Download citation
DOI: https://doi.org/10.1007/978-3-319-18497-5_18
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-18496-8
Online ISBN: 978-3-319-18497-5
eBook Packages: MedicineMedicine (R0)