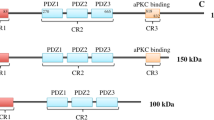
Abstract
Proteins of the evolutionary conserved family of partitioning-defective (Par) genes have emerged as key regulators of polarity and thus of cell and tissue architecture. Par proteins mediate a variety of cellular processes and couple control of cell shape to crucial signaling pathways regulating growth and survival, metabolism, cell fate, and differentiation. Alterations in adhesion, polarity, and architecture of tumor cells are hallmarks of cancer and implicated in tumor growth, invasion, and metastasis. Seminal work in Drosophila and mammalian cell culture suggested a molecular connection between the regulation of polarity and oncogenic processes. Recent advance stems from different mouse models revealing a causal link between polarity protein dysfunction and the formation and progression of cancer starting to shed light into some underlying mechanisms. It has become apparent that polarity signaling impacts on a multitude of processes involved in tumor formation and progression and that significant context dependency exists.
This chapter provides an overview of physiological processes that when disturbed facilitate tumor formation. Recent evidence from model organisms implicating Par protein dysfunction in the onset and progression of cancer is summarized. As Par proteins are tightly connected to aPKC signalling, data regarding a role of aPKC in cancer is also presented. Important findings from invertebrate tumor models will be reviewed, though the chapter focuses on functional data derived from mammalian systems.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Aprelikova O, Pace AJ, Fang B et al (2001) BRCA1 is a selective co-activator of 14-3-3 sigma gene transcription in mouse embryonic stem cells. J Biol Chem 276:25647–25650. doi:10.1074/jbc.C100265200
Atwood SX, Li M, Lee A et al (2013) GLI activation by atypical protein kinase C ι/λ regulates the growth of basal cell carcinomas. Nature 494:484–488. doi:10.1038/nature11889
Beck B, Driessens G, Goossens S et al (2011) A vascular niche and a VEGF-Nrp1 loop regulate the initiation and stemness of skin tumours. Nature 478:399–403. doi:10.1038/nature10525
Bilder D (2000) Cooperative regulation of cell polarity and growth by Drosophila tumor suppressors. Science 289:113–116. doi:10.1126/science.289.5476.113
Bilder D (2004) Epithelial polarity and proliferation control: links from the Drosophila neoplastic tumor suppressors. Genes Dev 18:1909–1925. doi:10.1101/gad.1211604
Brack AS, Rando TA (2012) Tissue-specific stem cells: lessons from the skeletal muscle satellite cell. Cell Stem Cell 10:504–514. doi:10.1016/j.stem.2012.04.001
Brumby AM, Goulding KR, Schlosser T et al (2011) Identification of novel Ras-cooperating oncogenes in Drosophila melanogaster: a RhoGEF/Rho-family/JNK pathway is a central driver of tumorigenesis. Genetics 188:105–125. doi:10.1534/genetics.111.127910
Brumby AM, Richardson HE (2003) Scribble mutants cooperate with oncogenic Ras or Notch to cause neoplastic overgrowth in Drosophila. EMBO J 22:5769–5779. doi:10.1093/emboj/cdg548
Brumby AM, Goulding KR, Schlosser T, Loi S, Galea R, Khoo P, Bolden JE, Aigaki T, Humbert PO, Richardson HE (2011) Identification of novel Ras-cooperating oncogenes in Drosophila melanogaster: a RhoGEF/Rho-family/JNK pathway is a central driver of tumorigenesis. Genetics 188(1):105–25. doi:10.1534/genetics.111.127910, Epub 2011 Mar 2. PMID: 21368274
Bu P, Chen K-Y, Chen JH et al (2013) A microRNA miR-34a-regulated bimodal switch targets notch in colon cancer stem cells. Cell Stem Cell 12:602–615. doi:10.1016/j.stem.2013.03.002
Caruso S, Bazan V, Rolfo C et al (2012) MicroRNAs in colorectal cancer stem cells: new regulators of cancer stemness? Oncogenesis 1:e32. doi:10.1038/oncsis.2012.33
Checkley LA, Rho O, Angel JM et al (2013) Metformin inhibits skin tumor promotion in overweight and obese mice. Cancer Prevention Research, Philadelphia, PA. doi:10.1158/1940-6207.CAPR-13-0110
Cicalese A, Bonizzi G, Pasi CE et al (2009) The tumor suppressor p53 regulates polarity of self-renewing divisions in mammary stem cells. Cell 138:1083–1095. doi:10.1016/j.cell.2009.06.048
Coppé JP, Desprez PY, Krtolica A, Campisi J (2010) The senescence-associated secretory phenotype: the dark side of tumor suppression. Annu Rev Pathol 5:99–118, PMID: 20078217
El-Hashash AH, Warburton D (2011) Cell polarity and spindle orientation in the distal epithelium of embryonic lung. Dev Dyn 240:441–445. doi:10.1002/dvdy.22551
Ellenbroek SIJ, Iden S, Collard JG (2012) Cell polarity proteins and cancer. Semin Cancer Biol 22:208–215. doi:10.1016/j.semcancer.2012.02.012
Freeman AK, Morrison DK (2011) 14-3-3 Proteins: diverse functions in cell proliferation and cancer progression. Semin Cell Dev Biol 22:681–687. doi:10.1016/j.semcdb.2011.08.009
Galvez AS, Duran A, Linares JF, Pathrose P, Castilla EA, Abu-Baker S, Leitges M, Diaz-Meco MT, Moscat J (2009) Protein kinase Cζ represses the interleukin-6 promoter and impairs tumorigenesis in vivo. Mol Cell Biol 29(1):104. doi:10.1128/MCB. 01294-08
Goldstein B, Macara IG (2007) The PAR proteins: fundamental players in animal cell polarization. Dev Cell 13:609–622. doi:10.1016/j.devcel.2007.10.007
Gont A, Hanson JEL, Lavictoire SJ et al (2013) PTEN loss represses glioblastoma differentiation via inactivation of Lgl1 tumor initiating cell. Oncotarget 4:1266–1279, Abstract
Gu Y, Lin S, Li J-L et al (2012) Altered LKB1/CREB-regulated transcription co-activator (CRTC) signaling axis promotes esophageal cancer cell migration and invasion. Oncogene 31:469–479. doi:10.1038/onc.2011.247
Gunaratne A, Thai BL, Di Guglielmo GM (2013) Atypical protein kinase C phosphorylates Par6 and facilitates transforming growth factor β-induced epithelial-to-mesenchymal transition. Mol Cell Biol 33:874–886. doi:10.1128/MCB. 00837-12
Gurumurthy S, Hezel AF, Sahin E et al (2008) LKB1 deficiency sensitizes mice to carcinogen-induced tumorigenesis. Cancer Res 68:55–63. doi:10.1158/0008-5472.CAN-07-3225
Hanahan D, Weinberg RA (2011) Hallmarks of cancer: the next generation. Cell 144:646–674. doi:10.1016/j.cell.2011.02.013
Hanahan D, Weinberg RA, Francisco S (2000) The Hallmarks of Cancer Review University of California at San Francisco. Cell 100:57–70
Hezel AF, Gurumurthy S, Granot Z et al (2008) Pancreatic LKB1 deletion leads to acinar polarity defects and cystic neoplasms. Mol Cell Biol 28:2414–2425. doi:10.1128/MCB. 01621-07
Hidalgo-Carcedo C, Hooper S, Chaudhry SI et al (2011) Collective cell migration requires suppression of actomyosin at cell-cell contacts mediated by DDR1 and the cell polarity regulators Par3 and Par6. Nat Cell Biol 13:49–58. doi:10.1038/ncb2133
Huang L, Muthuswamy SK (2010) Polarity protein alterations in carcinoma: a focus on emerging roles for polarity regulators. Curr Opin Genet Dev 20:41–50. doi:10.1016/j.gde.2009.12.001
Iden S, Collard JG (2008) Crosstalk between small GTPases and polarity proteins in cell polarization. Nat Rev Mol Cell Biol 9:846–859. doi:10.1038/nrm2521
Iden S, van Riel WE, Schäfer R et al (2012) Tumor type-dependent function of the Par3 polarity protein in skin tumorigenesis. Cancer Cell 22:389–403. doi:10.1016/j.ccr.2012.08.004
Jan Y-J, Ko B-S, Liu T-A et al (2013) Expression of partitioning defective 3 (par-3) for predicting extrahepatic metastasis and survival with hepatocellular carcinoma. Int J Mol Sci 14:1684–1697. doi:10.3390/ijms14011684
Ji H, Ramsey MR, Hayes DN et al (2007) LKB1 modulates lung cancer differentiation and metastasis. Nature 448:807–810. doi:10.1038/nature06030
Joshi J, Fernandez-Marcos PJ, Galvez A et al (2008) Par-4 inhibits Akt and suppresses Ras-induced lung tumorigenesis. EMBO J 27:2181–2193. doi:10.1038/emboj.2008.149
Kemphues K (2000) Minireview. Cell 101:345–348
Kemphues KJ, Priess JR, Morton DG, Cheng N (1988) Identification of genes required for cytoplasmic localization in early C. elegans embryos. Cell 52:311–320
Kirienko NV, Mani K, Fay DS (2010) Cancer models in Caenorhabditis elegans. Dev Dyn 239:1413–1448. doi:10.1002/dvdy.22247
Knust E (1994) Control of epithelial cell polarity in Drosophila. Trends Genet 10(8):275–80, Review. PMID: 7940756
Lancaster MA, Knoblich JA (2012) Spindle orientation in mammalian cerebral cortical development. Curr Opin Neurobiol 22:737–746. doi:10.1016/j.conb.2012.04.003
Lechler T, Fuchs E (2005) Asymmetric cell divisions promote stratification and differentiation of mammalian skin. Nature 437:275–280. doi:10.1038/nature03922
Linch M, Sanz-Garcia M, Soriano E et al (2013) A cancer-associated mutation in atypical protein kinase Cι occurs in a substrate-specific recruitment motif. Sci Signal 6:ra82. doi:10.1126/scisignal.2004068
Ling C, Su V-M-T, Zuo D, Muller WJ (2012) Loss of the 14-3-3σ tumor suppressor is a critical event in ErbB2-mediated tumor progression. Cancer Discov 2:68–81. doi:10.1158/2159-8290.CD-11-0189
Ling C, Zuo D, Xue B et al (2010) A novel role for 14-3-3sigma in regulating epithelial cell polarity. Genes Dev 24:947–956. doi:10.1101/gad.1896810
Liu S-G, Wang B-S, Jiang Y-Y et al (2011) Atypical protein kinase Cι (PKCι) promotes metastasis of esophageal squamous cell carcinoma by enhancing resistance to Anoikis via PKCι-SKP2-AKT pathway. Mol Cancer Res 9:390–402. doi:10.1158/1541-7786.MCR-10-0359
Liu X, Wang L, Shen Y et al (2013) Disruption of precise regulation of αPKC expression and cellular localization is associated with cervical cancer progression. Arch Gynecol Obstet 288:401–408. doi:10.1007/s00404-013-2770-7
Ma L, Tao Y, Duran A et al (2013) Control of nutrient stress-induced metabolic reprogramming by PKCζ in tumorigenesis. Cell 152:599–611. doi:10.1016/j.cell.2012.12.028
Mansfield AS, Fields AP, Jatoi A et al (2013) Phase I dose escalation study of the PKCι inhibitor aurothiomalate for advanced non-small-cell lung cancer, ovarian cancer, and pancreatic cancer. Anticancer Drugs 24:1079–1083. doi:10.1097/CAD.0000000000000009
McCaffrey LM, Macara IG (2009) The Par3/aPKC interaction is essential for end bud remodeling and progenitor differentiation during mammary gland morphogenesis. Genes Dev 23:1450–1460. doi:10.1101/gad.1795909
McCaffrey LM, Montalbano J, Mihai C, Macara IG (2012) Loss of the Par3 polarity protein promotes breast tumorigenesis and metastasis. Cancer Cell 22:601–614. doi:10.1016/j.ccr.2012.10.003
McClatchey AI, Yap AS (2012) Contact inhibition (of proliferation) redux. Curr Opin Cell Biol 24:685–694. doi:10.1016/j.ceb.2012.06.009
Mione MC, Trede NS (2010) The zebrafish as a model for cancer. Dis Model Mech 3:517–523. doi:10.1242/dmm.004747
Morén A, Raja E, Heldin C-H, Moustakas A (2011) Negative regulation of TGFβ signaling by the kinase LKB1 and the scaffolding protein LIP1. J Biol Chem 286:341–353. doi:10.1074/jbc.M110.190660
Murata T, Takayama K, Urano T et al (2012) 14-3-3Ζ, a novel androgen-responsive gene, is upregulated in prostate cancer and promotes prostate cancer cell proliferation and survival. Clin Cancer Res 18:5617–5627. doi:10.1158/1078-0432.CCR-12-0281
Murray NR, Kalari KR, Fields AP (2012) Protein kinase Cι expression and oncogenic signaling mechanisms in cancer. J Cell Physiol 226:879–887. doi:10.1002/jcp.22463.Protein
Murray NR, Weems J, Braun U et al (2009) Protein kinase C betaII and PKCiota/lambda: collaborating partners in colon cancer promotion and progression. Cancer Res 69:656–662. doi:10.1158/0008-5472.CAN-08-3001
Neal CL, Xu J, Li P et al (2012) Overexpression of 14-3-3ζ in cancer cells activates PI3K via binding the p85 regulatory subunit. Oncogene 31:897–906. doi:10.1038/onc.2011.284
Neal CL, Yu D (2010) 14-3-3Ζ as a prognostic marker and therapeutic target for cancer. Expert Opin Ther Targets 14:1343–1354. doi:10.1517/14728222.2010.531011
Nguyen HB, Babcock JT, Wells CD, Quilliam LA (2013) LKB1 tumor suppressor regulates AMP kinase/mTOR-independent cell growth and proliferation via the phosphorylation of Yap. Oncogene 32:4100–4109. doi:10.1038/onc.2012.431
Niessen MT, Iden S, Niessen CM (2012) The in vivo function of mammalian cell and tissue polarity regulators—how to shape and maintain the epidermal barrier. J Cell Sci 125(15):3501–10, Epub 2012 Aug 30
Niessen MT, Scott J, Zielinski JG et al (2013) aPKC controls epidermal homeostasis and stem cell fate through regulation of division orientation. J Cell Biol 202:887–900. doi:10.1083/jcb.201307001
Nolan ME, Aranda V, Lee S et al (2008) The polarity protein Par6 induces cell proliferation and is overexpressed in breast cancer. Cancer Res 68:8201–8209. doi:10.1158/0008-5472.CAN-07-6567
Oberholzer PA, Kee D, Dziunycz P et al (2012) RAS mutations are associated with the development of cutaneous squamous cell tumors in patients treated with RAF inhibitors. J Clin Oncol 30:316–321. doi:10.1200/JCO.2011.36.7680
Ollila S, Mäkelä TP (2011) The tumor suppressor kinase LKB1: lessons from mouse models. J Mol Cell Biol 3:330–340. doi:10.1093/jmcb/mjr016
Paget JA, Restall IJ, Daneshmand M et al (2012) Repression of cancer cell senescence by PKCι. Oncogene 31:3584–3596. doi:10.1038/onc.2011.524
Pagliarini RA, Xu T (2003) A genetic screen in Drosophila for metastatic behavior. Science (New York, NY) 302:1227–1231. doi:10.1126/science.1088474
Perez-Losada J, Balmain A (2003) Stem-cell hierarchy in skin cancer. Nat Rev Cancer 3:434–443. doi:10.1038/nrc1095
Poulson ND, Lechler T (2010) Robust control of mitotic spindle orientation in the developing epidermis. J Cell Biol 191:915–922. doi:10.1083/jcb.201008001
Quyn AJ, Appleton PL, Carey FA et al (2010) Spindle orientation bias in gut epithelial stem cell compartments is lost in precancerous tissue. Cell Stem Cell 6:175–181. doi:10.1016/j.stem.2009.12.007
Regala RP, Davis RK, Kunz A et al (2009) Atypical protein kinase C{iota} is required for bronchioalveolar stem cell expansion and lung tumorigenesis. Cancer Res 69:7603–7611. doi:10.1158/0008-5472.CAN-09-2066
Reinhardt HC, Schumacher B (2012) The p53 network: cellular and systemic DNA damage responses in aging and cancer. Trends Genet 28:128–136. doi:10.1016/j.tig.2011.12.002
Rimessi A, Zecchini E, Siviero R et al (2012) The selective inhibition of nuclear PKC ζ restores the effectiveness of chemotherapeutic agents in chemoresistant cells. Cell Cycle 11:1040–1048
Ben Sahra I, Le Marchand-Brustel Y, Tanti J-F, Bost F (2010) Metformin in cancer therapy: a new perspective for an old antidiabetic drug? Mol Cancer Ther 9:1092–1099. doi:10.1158/1535-7163.MCT-09-1186
Schultz J, Ibrahim SM, Vera J, Kunz M (2009) 14-3-3 Sigma gene silencing during melanoma progression and its role in cell cycle control and cellular senescence. Mol Cancer 8:53. doi:10.1186/1476-4598-8-53
Scotti ML, Bamlet WR, Smyrk TC et al (2010) Protein kinase Ciota is required for pancreatic cancer cell transformed growth and tumorigenesis. Cancer Res 70:2064–2074. doi:10.1158/0008-5472.CAN-09-2684
Sengupta A, Duran A, Florian MC et al (2011) Correction for Silverman et al., Metabolic cycling in single yeast cells from unsynchronized steady-state populations limited on glucose or phosphate. Proc Natl Acad Sci U S A 108:12185–12185. doi:10.1073/pnas.1109536108
Shackelford DB, Shaw RJ (2009) The LKB1-AMPK pathway: metabolism and growth control in tumour suppression. Nat Rev Cancer 9:563–575. doi:10.1038/nrc2676
Stallings-Mann M, Jamieson L, Regala RP et al (2006) A novel small-molecule inhibitor of protein kinase Ciota blocks transformed growth of non-small-cell lung cancer cells. Cancer Res 66:1767–1774. doi:10.1158/0008-5472.CAN-05-3405
Tepass U (2012) The apical polarity protein network in Drosophila epithelial cells: regulation of polarity, junctions, morphogenesis, cell growth, and survival. Annu Rev Cell Dev Biol 28:655–685. doi:10.1146/annurev-cellbio-092910-154033
Uhlirova M, Bohmann D (2006) JNK- and Fos-regulated Mmp1 expression cooperates with Ras to induce invasive tumors in Drosophila. EMBO J 25:5294–5304. doi:10.1038/sj.emboj.7601401
Van der Velden YU, Haramis A-PG (2011) Insights from model organisms on the functions of the tumor suppressor protein LKB1: zebrafish chips in. Aging 3:363–367
Van Der Velden YU, Wang L, Zevenhoven J et al (2011) The serine-threonine kinase LKB1 is essential for survival under energetic stress in zebra fish. Proc Natl Acad Sci U S A 108:4358–4363. doi:10.1073/pnas.1010210108
Velarde MC, Demaria M, Campisi J (2013) Senescent cells and their secretory phenotype as targets for cancer therapy. Interdiscip Top Gerontol 38:17–27. doi:10.1159/000343572, Epub 2013 Jan 17. Review. PMID: 23503512
Wei C, Bhattaram VK, Igwe JC et al (2012) The LKB1 tumor suppressor controls spindle orientation and localization of activated AMPK in mitotic epithelial cells. PLoS One 7:e41118. doi:10.1371/journal.pone.0041118
Williams SE, Beronja S, Pasolli HA, Fuchs E (2011) Asymmetric cell divisions promote Notch-dependent epidermal differentiation. Nature 470:353–358. doi:10.1038/nature09793
Williams SE, Fuchs E (2013) Oriented divisions, fate decisions. Curr Opin Cell Biol 25:749–758. doi:10.1016/j.ceb.2013.08.003
Wingo SN, Gallardo TD, Akbay EA et al (2009) Somatic LKB1 mutations promote cervical cancer progression. PLoS One 4:e5137. doi:10.1371/journal.pone.0005137
Xue B, Krishnamurthy K, Allred DC, Muthuswamy SK (2013) Loss of Par3 promotes breast cancer metastasis by compromising cell-cell cohesion. Nat Cell Biol 15:189–200. doi:10.1038/ncb2663
Yoshihama Y, Izumisawa Y, Akimoto K et al (2013) High expression of KIBRA in low atypical protein kinase C-expressing gastric cancer correlates with lymphatic invasion and poor prognosis. Cancer Sci 104:259–265. doi:10.1111/cas.12066
Young J, Valencia T, Abu-baker S et al (2013) c-Myc phosphorylation by PKCζ represses prostate tumorigenesis. Proc Natl Acad Sci U S A 110:6418–6423. doi:10.1073/pnas.1221799110
Acknowledgments
We are grateful for the financial and/or infrastructural support by the Excellence Initiative by the German federal and state governments (CECAD Cologne), the German Research Foundation (CRC829 and CRC832), Köln Fortune, the Stiftung Kölner Krebsforschung (EI—Exzellenz Initiieren), and the Center for Molecular Medicine Cologne (CMMC).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Mescher, M., Iden, S. (2015). Par Proteins in Tumor Formation and Progression. In: Ebnet, K. (eds) Cell Polarity 2. Springer, Cham. https://doi.org/10.1007/978-3-319-14466-5_6
Download citation
DOI: https://doi.org/10.1007/978-3-319-14466-5_6
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-14465-8
Online ISBN: 978-3-319-14466-5
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)