Abstract
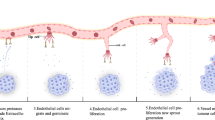
The knowledge related to angiogenesis has grown exponentially over the past few decades with the recognition that angiogenesis is essential for numerous normal and pathological processes. Very importantly, angiogenesis is required for the growth and metastasis of solid tumors in human beyond the size of 1–2 mm³ (Arap et al. 1998; Folkman 1990, 1995). Angiogenesis is the process of developing buds and outgrowth of capillaries from existing blood vessels that are derived as extensions due to hypoxia or other forms of signaling that occurs in the microenvironment that surrounds a tissue or tumors. On the other hand neovascularization is defined as the formation of functional microvascular network with red blood cell perfusion. Angiogenesis is required for invasive tumor growth and metastasis and therefore constitutes an important step in the control of cancer progression. In general vascular tumors are severely restricted in their growth potential because of the lack of a blood supply. To achieve the new blood vessel formation, endothelial cells must first escape from their stable location by breaking through the basement membrane, and this degradation is associated with migration of endothelial cells out of the vascular channel toward the angiogenic stimulus. During this process, the subendothelial basement membrane, a dense meshwork of collagen, glycoproteins, and proteoglycans are proteolytically disrupted to allow formation of new capillaries. Though it is an integral component of normal processes such as reproduction and wound healing, angiogenesis is known to play an important role in other pathological processes ranging from tumor growth and metastasis to inflammation and ocular diseases. During angiogenesis tumor cells exploit their microenvironment by releasing cytokines and growth factors to activate normal, quiescent cells around them and initiate a cascade of events that quickly becomes dysregulated. For example, tumor cell-released vascular endothelial growth factor (VEGF) stimulates the sprouting and proliferation of endothelial cells and thereby play a crucial role in neovascularization of solid tumors (Leung et al. 1989). The expression of VEGF has been shown to correlate with the density of microvessels in various tumors and exhibit higher metastatic ability (Folkman 1995; Leung et al. 1989; Toi et al. 1994). Therefore, inhibition of angiogenesis triggered by VEGF or other factors is accepted as a valuable approach to cancer therapy.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Arap W, Pasqualini R, Ruoslahti E (1998) Chemotherapy targeted to tumor vasculature. Curr Opin Oncol 10:560–565
Brekken RA, Overholser JP, Stastny VA, Waltenberger J, Minna JD, Thorpe PE (2000) Selective inhibition of vascular endothelial growth factor (VEGF) receptor 2 (KDR/Flk-1) activity by a monoclonal anti-VEGF antibody blocks tumor growth in mice. Cancer Res 60:5117–5124
Cheng OC, Han JH, Thomas HG, Balentien E, Richmond A (1992) The melanoma growth stimulatory activity receptor consists of two proteins. Ligand binding results in enhanced tyrosine phosphorylation. J Immunol 148:451–456
Fiser A, Do RK, Sali A (2000) Modeling of loops in protein structures. Protein Sci 9:1753–1773
Folkman J (1990) Endothelial cells and angiogenic growth factors in cancer growth and metastasis. Introduction. Cancer Metastasis Rev 9:171–174
Folkman J (1995) Angiogenesis in cancer, vascular, rheumatoid and other disease. Nat Med 1:27–31
Jain RK (2002) Tumor angiogenesis and accessibility: role of vascular endothelial growth factor. Semin Oncol 29:3–9
Kubo K et al (2005) Novel potent orally active selective VEGFR-2 tyrosine kinase inhibitors: synthesis, structure-activity relationships, and antitumour activities of N-phenyl-N'-{4-(4-quinolyloxy)phenyl}ureas. J Med Chem 48:1359
Laird AD, Vajkoczy P, Shawver LK, Thurnher A, Liang C et al (2000) SU6668 is a potent anti-angiogenic and antitumor agent that induces regression of established tumors. Cancer Res 60:4152–4160
Leung DW, Cachianes G, Kuang WJ, Goeddel DV, Ferrara N (1989) Vascular endothelial growth factor is a secreted angiogenic mitogen. Science 246:1306–1309
Ma J, Waxman DJ (2008) Combination of anti-angiogenesis with chemotherapy for more. Mol Cancer Ther 7(12):3670–3684
McMohan G (2000) VEGF receptor signaling in tumor angiogenesis. Oncologist 5(Suppl 1):3–10
Mendel DB et al (2003) In vivo antitumor activity of SU11248, a novel tyrosine kinase inhibitor targeting vascular endothelial growth factor and platelet-derived growth factor receptors: determination of a pharmacokinetic/pharmacodynamic relationship. Clin Cancer Res 9:327
Meyer M, Clauss M, Lepple-Wienhues A et al (1999) A novel vascular endothelial growth factor encoded by Orf virus, VEGF- E, mediates angiogenesis via signalling through VEGFR-2 (KDR) but not VEGFR-1 (Flt-1) receptor tyrosine kinases. EMBO J 18:363–374
Miao RQ, Agata J, Chao L, Chao J (2002) Kallistatin is a new inhibitor of angiogenesis and tumor growth. Blood 100:3245–3252
Mohammadi M, McMahon G, Sun L, Tang C, Hirth P, Yeh BK, Hubbard SR, Schlessinger J (1997) Structures of the tyrosine kinase domain of fibroblast growth factor receptor in complex with inhibitors. Science 276:955–960
Sonpavde G, Hutson TE (2008) Sternberg CN Pazopanib, a potent orally administered small-molecule multitargeted tyrosine kinase inhibitor for renal cell carcinoma. Expert Opin Investig Drugs 17(2):253–261
Toi M, Hoshina S, Takayanagi T, Tominaga T (1994) Association of vascular endothelial growth factor expression with tumor angiogenesis and with early relapse in primary breast cancer. Jpn J Cancer Res 85:1045–1049
Ueda Y, Yamagishi T, Samata K, Ikeya H, Hirayama N et al (2003) A novel low molecular weight antagonist of vascular endothelial growth factor receptor binding: VGA1155. Mol Cancer Ther 2:1105–1111
Wedge SR, Ogilvie DJ, Dukes M, Kendrew J, Curwen JO et al (2000) ZD4190: an orally active inhibitor of vascular endothelial growth factor signaling with broad-spectrum antitumor efficacy. Cancer Res 60:970–975
Hu-Lowe DD et al (2008) Non-clinical anti-angiogenesis and antitumor activities of axitinib (AG-013736), an oral, potent, and selective inhibitor of vascular endothelial growth factor receptor tyrosine kinases 1, 2 and 3. Clin Cancer Res 14:7272
Sridhar J, Akula N, Sivanesan D, Narasimhan M, Rathinavelu A, Pattabiraman N (2005) Identification of novel angiogenesis inhibitors. Bioorg Med Chem Lett 15:4125–4129
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 Springer International Publishing Switzerland
About this paper
Cite this paper
Rathinavelu, A. (2015). Tumor Angiogenesis and Novel Vascular Endothelial Receptor (VEGFR)-Specific Small Molecule Inhibitors. In: Gandhi, V., Mehta, K., Grover, R., Pathak, S., Aggarwal, B. (eds) Multi-Targeted Approach to Treatment of Cancer. Adis, Cham. https://doi.org/10.1007/978-3-319-12253-3_15
Download citation
DOI: https://doi.org/10.1007/978-3-319-12253-3_15
Published:
Publisher Name: Adis, Cham
Print ISBN: 978-3-319-12252-6
Online ISBN: 978-3-319-12253-3
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)