Abstract
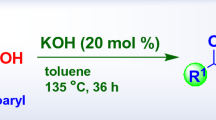
Transformation of enolizable α-haloketones to esters, carboxylic acids, or amides via alkoxide-, hydroxide-, or amine-catalyzed rearrangements, respectively.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
(a) Favorskii, A. E. J. Prakt. Chem. 1895, 51, 533–563. Aleksei E. Favorskii (1860–1945), born in Selo Pavlova, Russia, studied at St. Petersburg State University, where he became a professor since 1900. (b) Favorskii, A. E. J. Prakt. Chem. 1913, 88, 658.
Wagner, R. B.; Moore, J. A. J. Am. Chem. Soc. 1950, 72, 3655–3658.
Wenkert, E.; Bakuzis, P.; Baumgarten, R. J.; Leicht, C. L.; Schenk, H. P. J. Am. Chem. Soc. 1971, 93, 3208–3216.
Chenier, P. J. J. Chem. Ed. 1978, 55, 286–291. (Review).
Barreta, A.; Waegell, B. In Reactive Intermediates; Abramovitch, R. A., ed.; Plenum Press: New York, 1982, 2, pp 527–585. (Review).
White, J. D.; Dillon, M. P.; Butlin, R. J. J. Am. Chem. Soc. 1992, 114, 9673–9674.
Dhavale, D. D.; Mali, V. P.; Sudrik, S. G.; Sonawane, H. R. Tetrahedron 1997, 53, 16789–16794.
Kitayama, T.; Okamoto, T. J. Org. Chem. 1999, 64, 2667–2672.
Mamedov, V. A.; Tsuboi, S.; Mustakimova, L. V.; Hamamoto, H.; Gubaidullin, A. T.; Litvinov, I. A.; Levin, Y. A. Chem. Heterocyclic Compd. 2001, 36, 911. (Review).
Harmata, M.; Wacharasindhu, S. Org. Lett. 2005, 7, 2563–2565.
Pogrebnoi, S.; Saraber, F. C. E.; Jansen, B. J. M.; de Groot, A. Tetrahedron 2006, 62, 1743–1748.
Filipski, K.J.; Pfefferkorn, J. A. Favorskii Rearrangement. In Name Reactions for Homologations-Part II; Li, J. J., Ed.; Wiley: Hoboken, NJ, 2009, pp 238–252. (Review).
Kammath, V. B.; Šolomek, T.; Ngoy, B. P.; Heger, D.; Klán, P.; Rubina, M.; Givens, R. S. J. Org. Chem. 2013, 78, 1718–1729.
References
Cope, A. C.; Graham, E. S. J. Am. Chem. Soc. 1951, 73, 4702–4706.
Smissman, E. E.; Diebold, J. L. J. Org. Chem. 1965, 30, 4005–4007.
Sasaki, T.; Eguchi, S.; Toru, T. J. Am. Chem. Soc. 1969, 91, 3390–3391.
Baudry, D.; Begue, J. P.; Charpentier-Morize, M. Tetrahedron Lett. 1970, 2147–2150.
Stevens, C. L.; Pillai, P. M.; Taylor, K. G. J. Org. Chem. 1974, 39, 3158–3161.
Harmata, M.; Wacharasindhu, S. J. Org. Chem. 2005, 70, 725–728.
Filipski, K.J.; Pfefferkorn, J. A. Favorskii Rearrangement. In Name Reactions for Homologations-Part II; Li, J. J., Ed.; Wiley: Hoboken, NJ, 2009, pp 438–452. (Review).
Harmata, M.; Wacharasindhu, S. Synthesis 2007, 2365–2369.
Ross, A. G.; Townsend, S. D.; Danishefsky, S. J. J. Org. Chem. 2013, 78, 204–210.
Author information
Authors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Li, J.J. (2014). Favorskii rearrangement. In: Name Reactions. Springer, Cham. https://doi.org/10.1007/978-3-319-03979-4_100
Download citation
DOI: https://doi.org/10.1007/978-3-319-03979-4_100
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-03978-7
Online ISBN: 978-3-319-03979-4
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)