Abstract
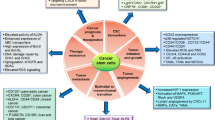
Many tumors are resistant to conventional cancer therapies because a tumor is composed of heterogeneous cell population. Especially, subpopulation of cancer stem cells, which have self-renewal and differentiation properties and responsible for the tumor initiation, is generally considered resistant to chemo-, radio-, and immune therapy. Understanding the mechanism of drug resistance in cancer stem cells should lead to establish more effective therapeutic strategies. Actually, different molecular mechanisms are conceivable for cancer stem cells acquiring drug resistance. These mechanisms include not only cytoplasmic signaling pathways but also the intercellular communications in the tumor microenvironment. Recently, a great deal of successful reports challenged to elucidate the mechanisms of drug resistance and to develop novel treatments targeting cancer stem cells.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Afify SM, Seno M (2019) Conversion of stem cells to cancer stem cells: Undercurrent of cancer Initiation. Cancers (Basel) 11(3). https://doi.org/10.3390/cancers11030345
Ajani JA, Wang X, Song S, Suzuki A, Taketa T, Sudo K, Berry D (2014) ALDH-1 expression levels predict response or resistance to preoperative chemoradiation in resectable esophageal cancer patients. Mol Oncol 8(1):142–149. https://doi.org/10.1016/j.molonc.2013.10.007
Almozyan S, Colak D, Mansour F, Alaiya A, Al-Harazi O, Qattan A, Ghebeh H (2017) PD-L1 promotes OCT4 and Nanog expression in breast cancer stem cells by sustaining PI3K/AKT pathway activation. Int J Cancer 141(7):1402–1412. https://doi.org/10.1002/ijc.30834
Bao S, Wu Q, Li Z, Sathornsumetee S, Wang H, McLendon RE, Rich JN (2008) Targeting cancer stem cells through L1CAM suppresses glioma growth. Cancer Res 68(15):6043–6048. https://doi.org/10.1158/0008-5472.Can-08-1079
Barna G, Mihalik R, Timár B, Tömböl J, Csende Z, Sebestyén A, Matolcsy A (2011) ROR1 expression is not a unique marker of CLL. Hematol Oncol 29(1):17–21. https://doi.org/10.1002/hon.948
Barnawi R, Al-Khaldi S, Bakheet T, Fallatah M, Alaiya A, Ghebeh H, Al-Alwan M (2020) Fascin activates β-catenin signaling and promotes breast cancer stem cell function mainly through focal adhesion kinase (fak): relation with disease progression. Front Oncol 10:440. https://doi.org/10.3389/fonc.2020.00440
Barnawi R, Al-Khaldi S, Majed Sleiman G, Sarkar A, Al-Dhfyan A, Al-Mohanna F, Al-Alwan M (2016) Fascin is critical for the maintenance of breast cancer stem cell pool predominantly via the activation of the notch self-renewal pathway. Stem Cells 34(12):2799–2813. https://doi.org/10.1002/stem.2473
Baskar S, Kwong KY, Hofer T, Levy JM, Kennedy MG, Lee E, Rader C (2008) Unique cell surface expression of receptor tyrosine kinase ROR1 in human B-cell chronic lymphocytic leukemia. Clin Cancer Res 14(2):396–404. https://doi.org/10.1158/1078-0432.Ccr-07-1823
Batlle E, Clevers H (2017) Cancer stem cells revisited. Nat Med 23(10):1124–1134. https://doi.org/10.1038/nm.4409
Beachy PA, Hymowitz SG, Lazarus RA, Leahy DJ, Siebold C (2010) Interactions between Hedgehog proteins and their binding partners come into view. Genes Dev 24(18):2001–2012. https://doi.org/10.1101/gad.1951710
Bhat M, Robichaud N, Hulea L, Sonenberg N, Pelletier J, Topisirovic I (2015) Targeting the translation machinery in cancer. Nat Rev Drug Discov 14(4):261–278. https://doi.org/10.1038/nrd4505
Cazet AS, Hui MN, Elsworth BL, Wu SZ, Roden D, Chan CL, Swarbrick A (2018) Targeting stromal remodeling and cancer stem cell plasticity overcomes chemoresistance in triple negative breast cancer. Nat Commun 9(1):2897. https://doi.org/10.1038/s41467-018-05220-6
Chen C, Cao F, Bai L, Liu Y, Xie J, Wang W, Luo Y (2015) IKKβ enforces a LIN28B/TCF7L2 positive feedback loop that promotes cancer cell stemness and metastasis. Cancer Res 75(8):1725–1735. https://doi.org/10.1158/0008-5472.Can-14-2111
Chiou GY, Yang TW, Huang CC, Tang CY, Yen JY, Tsai MC, Jong YJ (2017) Musashi-1 promotes a cancer stem cell lineage and chemoresistance in colorectal cancer cells. Sci Rep 7(1):2172. https://doi.org/10.1038/s41598-017-02057-9
Clement V, Sanchez P, de Tribolet N, Radovanovic I, Ruiz i Altaba, A. (2007) HEDGEHOG-GLI1 signaling regulates human glioma growth, cancer stem cell self-renewal, and tumorigenicity. Curr Biol 17(2):165–172. https://doi.org/10.1016/j.cub.2006.11.033
Clevers H, Nusse R (2012) Wnt/β-catenin signaling and disease. Cell 149(6):1192–1205. https://doi.org/10.1016/j.cell.2012.05.012
Cochrane CR, Szczepny A, Watkins DN, Cain JE (2015) Hedgehog signaling in the maintenance of cancer stem cells. Cancers (Basel) 7(3):1554–1585. https://doi.org/10.3390/cancers7030851
Cojoc M, Mäbert K, Muders MH, Dubrovska A (2015) A role for cancer stem cells in therapy resistance: cellular and molecular mechanisms. Semin Cancer Biol 31:16–27. https://doi.org/10.1016/j.semcancer.2014.06.004
Dagogo-Jack I, Shaw AT (2018) Tumour heterogeneity and resistance to cancer therapies. Nat Rev Clin Oncol 15(2):81–94. https://doi.org/10.1038/nrclinonc.2017.166
Dai D, Wang H, Zhu L, Jin H, Wang X (2018) N6-methyladenosine links RNA metabolism to cancer progression. Cell Death Dis 9(2):124. https://doi.org/10.1038/s41419-017-0129-x
Dai G, Deng S, Guo W, Yu L, Yang J, Zhou S, Gao T (2019) Notch pathway inhibition using DAPT, a γ-secretase inhibitor (GSI), enhances the antitumor effect of cisplatin in resistant osteosarcoma. Mol Carcinog 58(1):3–18. https://doi.org/10.1002/mc.22873
Daneshmanesh AH, Mikaelsson E, Jeddi-Tehrani M, Bayat AA, Ghods R, Ostadkarampour M, Rabbani H (2008) Ror1, a cell surface receptor tyrosine kinase is expressed in chronic lymphocytic leukemia and may serve as a putative target for therapy. Int J Cancer 123(5):1190–1195. https://doi.org/10.1002/ijc.23587
Das S, Mukherjee P, Chatterjee R, Jamal Z, Chatterji U (2019) Enhancing chemosensitivity of breast cancer stem cells by downregulating SOX2 and ABCG2 using Wedelolactone-encapsulated nanoparticles. Mol Cancer Ther 18(3):680–692. https://doi.org/10.1158/1535-7163.Mct-18-0409
Davis BN, Hata A (2009) Regulation of MicroRNA Biogenesis: a miRiad of mechanisms. Cell Commun Signal 7:18. https://doi.org/10.1186/1478-811x-7-18
Desai A, Yan Y, Gerson SL (2019) Concise reviews: cancer stem cell targeted therapies: toward clinical success. Stem Cells Transl Med 8(1):75–81. https://doi.org/10.1002/sctm.18-0123
Diehn M, Cho RW, Lobo NA, Kalisky T, Dorie MJ, Kulp AN, Clarke MF (2009) Association of reactive oxygen species levels and radioresistance in cancer stem cells. Nature 458(7239):780–783. https://doi.org/10.1038/nature07733
Domingo-Domenech J, Vidal SJ, Rodriguez-Bravo V, Castillo-Martin M, Quinn SA, Rodriguez-Barrueco R, Cordon-Cardo C (2012) Suppression of acquired docetaxel resistance in prostate cancer through depletion of notch- and hedgehog-dependent tumor-initiating cells. Cancer Cell 22(3):373–388. https://doi.org/10.1016/j.ccr.2012.07.016
Edris B, Espinosa I, Mühlenberg T, Mikels A, Lee CH, Steigen SE, van de Rijn M (2012) ROR2 is a novel prognostic biomarker and a potential therapeutic target in leiomyosarcoma and gastrointestinal stromal tumour. J Pathol 227(2):223–233. https://doi.org/10.1002/path.3986
Emons G, Spitzner M, Reineke S, Möller J, Auslander N, Kramer F, Grade M (2017) Chemoradiotherapy resistance in colorectal cancer cells is mediated by Wnt/β-catenin signaling. Mol Cancer Res 15(11):1481–1490. https://doi.org/10.1158/1541-7786.Mcr-17-0205
Enomoto M, Hayakawa S, Itsukushima S, Ren DY, Matsuo M, Tamada K, Minami Y (2009) Autonomous regulation of osteosarcoma cell invasiveness by Wnt5a/Ror2 signaling. Oncogene 28(36):3197–3208. https://doi.org/10.1038/onc.2009.175
Fang T, Lv H, Wu F, Wang C, Li T, Lv G, Wang H (2017) Musashi 2 contributes to the stemness and chemoresistance of liver cancer stem cells via LIN28A activation. Cancer Lett 384:50–59. https://doi.org/10.1016/j.canlet.2016.10.007
Galimberti S, Grassi S, Baratè C, Guerrini F, Ciabatti E, Perutelli F, Mattii L (2018) The Polycomb BMI1 Protein Is Co-expressed With CD26+ in Leukemic stem cells of chronic myeloid Leukemia. Front Oncol 8:555. https://doi.org/10.3389/fonc.2018.00555
Gallipoli P, Pellicano F, Morrison H, Laidlaw K, Allan EK, Bhatia R, Holyoake TL (2013) Autocrine TNF-α production supports CML stem and progenitor cell survival and enhances their proliferation. Blood 122(19):3335–3339. https://doi.org/10.1182/blood-2013-02-485607
Galluzzi L, Vitale I, Aaronson SA, Abrams JM, Adam D, Agostinis P, Kroemer G (2018) Molecular mechanisms of cell death: recommendations of the Nomenclature Committee on Cell Death 2018. Cell Death Differ 25(3):486–541. https://doi.org/10.1038/s41418-017-0012-4
Gentile A, Lazzari L, Benvenuti S, Trusolino L, Comoglio PM (2011) Ror1 is a pseudokinase that is crucial for Met-driven tumorigenesis. Cancer Res 71(8):3132–3141. https://doi.org/10.1158/0008-5472.Can-10-2662
Green J, Nusse R, van Amerongen R (2014) The role of Ryk and Ror receptor tyrosine kinases in Wnt signal transduction. Cold Spring Harb Perspect Biol, 6(2). https://doi.org/10.1101/cshperspect.a009175
Gulino A, Di Marcotullio L, Screpanti I (2010) The multiple functions of Numb. Exp Cell Res 316(6):900–906. https://doi.org/10.1016/j.yexcr.2009.11.017
Guo Y, Zhang K, Cheng C, Ji Z, Wang X, Wang M, Gao WQ (2017) Numb(-/low) enriches a Castration-resistant prostate cancer cell subpopulation associated with Enhanced Notch and Hedgehog signaling. Clin Cancer Res 23(21):6744–6756. https://doi.org/10.1158/1078-0432.Ccr-17-0913
Haga RB, Ridley AJ (2016) Rho GTPases: Regulation and roles in cancer cell biology. Small GTPases 7(4):207–221. https://doi.org/10.1080/21541248.2016.1232583
Harvey KF, Pfleger CM, Hariharan IK (2003) The Drosophila Mst ortholog, hippo, restricts growth and cell proliferation and promotes apoptosis. Cell 114(4):457–467. https://doi.org/10.1016/s0092-8674(03)00557-9
Hayden MS, Ghosh S (2008) Shared principles in NF-kappaB signaling. Cell 132(3):344–362. https://doi.org/10.1016/j.cell.2008.01.020
Hemler ME (2003) Tetraspanin proteins mediate cellular penetration, invasion, and fusion events and define a novel type of membrane microdomain. Annu Rev Cell Dev Biol 19:397–422. https://doi.org/10.1146/annurev.cellbio.19.111301.153609
Hemmings BA, Restuccia DF (2012) PI3K-PKB/Akt pathway. Cold Spring Harb Perspect Biol 4(9):a011189. https://doi.org/10.1101/cshperspect.a011189
Holohan C, Van Schaeybroeck S, Longley DB, Johnston PG (2013) Cancer drug resistance: an evolving paradigm. Nat Rev Cancer 13(10):714–726. https://doi.org/10.1038/nrc3599
Hu Y, Yan C, Mu L, Huang K, Li X, Tao D, Qin J (2015) Fibroblast-derived exosomes contribute to chemoresistance through priming cancer stem cells in colorectal cancer. PLoS ONE 10(5):e0125625. https://doi.org/10.1371/journal.pone.0125625
Huijbers EJ, van Beijnum JR, Thijssen VL, Sabrkhany S, Nowak-Sliwinska P, Griffioen AW (2016) Role of the tumor stroma in resistance to anti-angiogenic therapy. Drug Resist Updat 25:26–37. https://doi.org/10.1016/j.drup.2016.02.002
Irvin DK, Jouanneau E, Duvall G, Zhang XX, Zhai Y, Sarayba D, Wheeler CJ (2010) T cells enhance stem-like properties and conditional malignancy in gliomas. PLoS ONE 5(6):e10974. https://doi.org/10.1371/journal.pone.0010974
Kahn M (2018) Wnt signaling in stem cells and cancer stem Cells: a tale of two coactivators. Prog Mol Biol Transl Sci 153:209–244. https://doi.org/10.1016/bs.pmbts.2017.11.007
Karin M (2009) NF-kappaB as a critical link between inflammation and cancer. Cold Spring Harb Perspect Biol 1(5):a000141. https://doi.org/10.1101/cshperspect.a000141
Kasper M, Jaks V, Fiaschi M, Toftgård R (2009) Hedgehog signalling in breast cancer. Carcinogenesis 30(6):903–911. https://doi.org/10.1093/carcin/bgp048
Katoh M (2017) Canonical and non-canonical WNT signaling in cancer stem cells and their niches: Cellular heterogeneity, omics reprogramming, targeted therapy and tumor plasticity (Review). Int J Oncol 51(5):1357–1369. https://doi.org/10.3892/ijo.2017.4129
Khan MA, Srivastava SK, Zubair H, Patel GK, Arora S, Khushman M, Singh AP (2020) Co-targeting of CXCR4 and hedgehog pathways disrupts tumor-stromal crosstalk and improves chemotherapeutic efficacy in pancreatic cancer. J Biol Chem 295(25):8413–8424. https://doi.org/10.1074/jbc.RA119.011748
Kleffel S, Schatton T (2013) Tumor dormancy and cancer stem cells: two sides of the same coin? Adv Exp Med Biol 734:145–179. https://doi.org/10.1007/978-1-4614-1445-2_8
Kobune M, Takimoto R, Murase K, Iyama S, Sato T, Kikuchi S, Kato J (2009) Drug resistance is dramatically restored by hedgehog inhibitors in CD34+ leukemic cells. Cancer Sci 100(5):948–955. https://doi.org/10.1111/j.1349-7006.2009.01111.x
Krause M, Dubrovska A, Linge A, Baumann M (2017) Cancer stem cells: Radioresistance, prediction of radiotherapy outcome and specific targets for combined treatments. Adv Drug Deliv Rev 109:63–73. https://doi.org/10.1016/j.addr.2016.02.002
Lee JE, Park HS, Lee D, Yoo G, Kim T, Jeon H, Chung C (2016) Hippo pathway effector YAP inhibition restores the sensitivity of EGFR-TKI in lung adenocarcinoma having primary or acquired EGFR-TKI resistance. Biochem Biophys Res Commun 474(1):154–160. https://doi.org/10.1016/j.bbrc.2016.04.089
Li F, Xu Y, Liu B, Singh PK, Zhao W, Jin J, Ajani JA (2019a) YAP1-Mediated CDK6 activation confers radiation resistance in esophageal Cancer—rationale for the combination of YAP1 and CDK4/6 inhibitors in esophageal cancer. Clin Cancer Res 25(7):2264–2277. https://doi.org/10.1158/1078-0432.Ccr-18-1029
Li H, Meng F, Jiang L, Ren Y, Qiu Z, Yu P, Peng J (2019b) Comparison of LC-MS/MS-based targeted proteomics and conventional analytical methods for monitoring breast cancer resistance protein expression. Life Sci 231:116548. https://doi.org/10.1016/j.lfs.2019.116548
Li L, Bhatia R (2011) Stem cell quiescence. Clin Cancer Res 17(15):4936–4941. https://doi.org/10.1158/1078-0432.Ccr-10-1499
Li P, Harris D, Liu Z, Liu J, Keating M, Estrov Z (2010) Stat3 activates the receptor tyrosine kinase like orphan receptor-1 gene in chronic lymphocytic leukemia cells. PLoS ONE 5(7):e11859. https://doi.org/10.1371/journal.pone.0011859
Li Z, Wang Y, Zhu Y, Yuan C, Wang D, Zhang W, Cheng J (2015) The Hippo transducer TAZ promotes epithelial to mesenchymal transition and cancer stem cell maintenance in oral cancer. Mol Oncol 9(6):1091–1105. https://doi.org/10.1016/j.molonc.2015.01.007
Lin J, Feng J, Yang H, Yan Z, Li Q, Wei L, Peng J (2017) Scutellaria barbata D. Don inhibits 5-fluorouracil resistance in colorectal cancer by regulating PI3K/AKT pathway. Oncol Rep, 38(4), 2293–2300. https://doi.org/10.3892/or.2017.5892
Lin L, Sabnis AJ, Chan E, Olivas V, Cade L, Pazarentzos E, Bivona TG (2015) The Hippo effector YAP promotes resistance to RAF- and MEK-targeted cancer therapies. Nat Genet 47(3):250–256. https://doi.org/10.1038/ng.3218
Lin Q, Peng S, Yang Y (2018) Inhibition of CD9 expression reduces the metastatic capacity of human hepatocellular carcinoma cell line MHCC97-H. Int J Oncol 53(1):266–274. https://doi.org/10.3892/ijo.2018.4381
Liu M, Sakamaki T, Casimiro MC, Willmarth NE, Quong AA, Ju X, Pestell RG (2010) The canonical NF-kappaB pathway governs mammary tumorigenesis in transgenic mice and tumor stem cell expansion. Cancer Res 70(24):10464–10473. https://doi.org/10.1158/0008-5472.Can-10-0732
Liu R, Chen Y, Liu G, Li C, Song Y, Cao Z, Liu Y (2020) PI3K/AKT pathway as a key link modulates the multidrug resistance of cancers. Cell Death Dis 11(9):797. https://doi.org/10.1038/s41419-020-02998-6
Liu T, Zhang L, Joo D, Sun SC (2017) NF-κB signaling in inflammation. Signal Transduct Target Ther 2, 17023 https://doi.org/10.1038/sigtrans.2017.23
Liu WT, Jing YY, Yu GF, Han ZP, Yu DD, Fan QM, Wei LX (2015) Toll like receptor 4 facilitates invasion and migration as a cancer stem cell marker in hepatocellular carcinoma. Cancer Lett 358(2):136–143. https://doi.org/10.1016/j.canlet.2014.12.019
Lukacs RU, Memarzadeh S, Wu H, Witte ON (2010) Bmi-1 is a crucial regulator of prostate stem cell self-renewal and malignant transformation. Cell Stem Cell 7(6):682–693. https://doi.org/10.1016/j.stem.2010.11.013
Ma S, Pradeep S, Hu W, Zhang D, Coleman R, Sood A (2018) The role of tumor microenvironment in resistance to anti-angiogenic therapy. F1000Res 7, 326. https://doi.org/10.12688/f1000research.11771.1
MacKeigan JP, Murphy LO, Blenis J (2005) Sensitized RNAi screen of human kinases and phosphatases identifies new regulators of apoptosis and chemoresistance. Nat Cell Biol 7(6):591–600. https://doi.org/10.1038/ncb1258
Malladi S, Macalinao DG, Jin X, He L, Basnet H, Zou Y, Massagué J (2016) Metastatic Latency and Immune Evasion through Autocrine Inhibition of WNT. Cell 165(1):45–60. https://doi.org/10.1016/j.cell.2016.02.025
Matsui WH (2016) Cancer Stem Cell Signaling Pathways. Medicine (baltimore) 95(1 Suppl 1):S8-s19. https://doi.org/10.1097/md.0000000000004765
Meng E, Mitra A, Tripathi K, Finan MA, Scalici J, McClellan S, Rocconi RP (2014) ALDH1A1 maintains ovarian cancer stem cell-like properties by altered regulation of cell cycle checkpoint and DNA repair network signaling. PLoS ONE 9(9):e107142. https://doi.org/10.1371/journal.pone.0107142
Milosevic V, Kopecka J, Salaroglio IC, Libener R, Napoli F, Izzo S, Riganti C (2020) Wnt/IL-1β/IL-8 autocrine circuitries control chemoresistance in mesothelioma initiating cells by inducing ABCB5. Int J Cancer 146(1):192–207. https://doi.org/10.1002/ijc.32419
Minassian LM, Cotechini T, Huitema E, Graham CH (2019) Hypoxia-Induced Resistance to Chemotherapy in Cancer. Adv Exp Med Biol 1136:123–139. https://doi.org/10.1007/978-3-030-12734-3_9
Moitra K (2015) Overcoming Multidrug Resistance in Cancer Stem Cells. Biomed Res Int 2015:635745. https://doi.org/10.1155/2015/635745
Morgenroth A, Vogg AT, Ermert K, Zlatopolskiy B, Mottaghy FM (2014) Hedgehog signaling sensitizes glioma stem cells to endogenous nano-irradiation. Oncotarget 5(14):5483–5493. https://doi.org/10.18632/oncotarget.2123
Morin PJ (2003) Drug resistance and the microenvironment: nature and nurture. Drug Resist Updat 6(4):169–172. https://doi.org/10.1016/s1368-7646(03)00059-1
Morioka K, Tanikawa C, Ochi K, Daigo Y, Katagiri T, Kawano H, Matsuda K (2009) Orphan receptor tyrosine kinase ROR2 as a potential therapeutic target for osteosarcoma. Cancer Sci 100(7):1227–1233. https://doi.org/10.1111/j.1349-7006.2009.01165.x
Moroishi T, Hansen CG, Guan KL (2015) The emerging roles of YAP and TAZ in cancer. Nat Rev Cancer 15(2):73–79. https://doi.org/10.1038/nrc3876
Naeli P, Pourhanifeh MH, Karimzadeh MR, Shabaninejad Z, Movahedpour A, Tarrahimofrad H, Hamblin MR (2020) Circular RNAs and gastrointestinal cancers: Epigenetic regulators with a prognostic and therapeutic role. Crit Rev Oncol Hematol 145:102854. https://doi.org/10.1016/j.critrevonc.2019.102854
Nahand JS, Taghizadeh-Boroujeni S, Karimzadeh M, Borran S, Pourhanifeh MH, Moghoofei M, Mirzaei H (2019) microRNAs: New prognostic, diagnostic, and therapeutic biomarkers in cervical cancer. J Cell Physiol 234(10):17064–17099. https://doi.org/10.1002/jcp.28457
Nascimento AS, Peres LL, Fari AVS, Milani R, Silva RA, da Costa Fernandes CJ, Zambuzzi WF (2017) Phosphoproteome profiling reveals critical role of JAK-STAT signaling in maintaining chemoresistance in breast cancer. Oncotarget 8(70):114756–114768. https://doi.org/10.18632/oncotarget.21801
Nassar D, Blanpain C (2016) Cancer Stem Cells: Basic Concepts and Therapeutic Implications. Annu Rev Pathol 11:47–76. https://doi.org/10.1146/annurev-pathol-012615-044438
Nebenfuehr S, Kollmann K, Sexl V (2020) The role of CDK6 in cancer. Int J Cancer 147(11):2988–2995. https://doi.org/10.1002/ijc.33054
Nguyen PH, Giraud J, Chambonnier L, Dubus P, Wittkop L, Belleannée G, Varon C (2017) characterization of biomarkers of tumorigenic and chemoresistant cancer stem cells in human gastric carcinoma. Clin Cancer Res 23(6):1586–1597. https://doi.org/10.1158/1078-0432.Ccr-15-2157
O’Connell MP, Fiori JL, Baugher KM, Indig FE, French AD, Camilli TC, Weeraratna AT (2009) Wnt5A activates the calpain-mediated cleavage of filamin A. J Invest Dermatol 129(7):1782–1789. https://doi.org/10.1038/jid.2008.433
Ojo D, Lin X, Wu Y, Cockburn J, Bane A, Tang D (2018) Polycomb complex protein BMI1 confers resistance to tamoxifen in estrogen receptor positive breast cancer. Cancer Lett 426:4–13. https://doi.org/10.1016/j.canlet.2018.03.048
Oshimori N, Oristian D, Fuchs E (2015) TGF-β promotes heterogeneity and drug resistance in squamous cell carcinoma. Cell 160(5):963–976. https://doi.org/10.1016/j.cell.2015.01.043
Park IK, Qian D, Kiel M, Becker MW, Pihalja M, Weissman IL, Clarke MF (2003) Bmi-1 is required for maintenance of adult self-renewing haematopoietic stem cells. Nature 423(6937):302–305. https://doi.org/10.1038/nature01587
Park KS, Martelotto LG, Peifer M, Sos ML, Karnezis AN, Mahjoub MR, Sage J (2011) A crucial requirement for Hedgehog signaling in small cell lung cancer. Nat Med 17(11):1504–1508. https://doi.org/10.1038/nm.2473
Peacock CD, Wang Q, Gesell GS, Corcoran-Schwartz IM, Jones E, Kim J, Matsui W (2007) Hedgehog signaling maintains a tumor stem cell compartment in multiple myeloma. Proc Natl Acad Sci U S A 104(10):4048–4053. https://doi.org/10.1073/pnas.0611682104
Pestova TV, Kolupaeva VG, Lomakin IB, Pilipenko EV, Shatsky IN, Agol VI, Hellen CU (2001) Molecular mechanisms of translation initiation in eukaryotes. Proc Natl Acad Sci U S A 98(13):7029–7036. https://doi.org/10.1073/pnas.111145798
Phi LTH, Sari IN, Yang YG, Lee SH, Jun N, Kim KS, Kwon HY (2018) Cancer Stem cells (CSCs) in drug resistance and their therapeutic implications in cancer treatment. Stem Cells Int 2018:5416923. https://doi.org/10.1155/2018/5416923
Phillips E, Lang V, Bohlen J, Bethke F, Puccio L, Tichy D, Goidts V (2016) Targeting atypical protein kinase C iota reduces viability in glioblastoma stem-like cells via a notch signaling mechanism. Int J Cancer 139(8):1776–1787. https://doi.org/10.1002/ijc.30234
Podergajs N, Motaln H, Rajčević U, Verbovšek U, Koršič M, Obad N, Turnšek TL (2016) Transmembrane protein CD9 is glioblastoma biomarker, relevant for maintenance of glioblastoma stem cells. Oncotarget 7(1):593–609. https://doi.org/10.18632/oncotarget.5477
Pors K, Moreb JS (2014) Aldehyde dehydrogenases in cancer: an opportunity for biomarker and drug development? Drug Discov Today 19(12):1953–1963. https://doi.org/10.1016/j.drudis.2014.09.009
Porta C, Paglino C, Mosca A (2014) Targeting PI3K/Akt/mTOR Signaling in Cancer. Front Oncol 4:64. https://doi.org/10.3389/fonc.2014.00064
Prasad S, Ravindran J, Aggarwal BB (2010) NF-kappaB and cancer: how intimate is this relationship. Mol Cell Biochem 336(1–2):25–37. https://doi.org/10.1007/s11010-009-0267-2
Raha D, Wilson TR, Peng J, Peterson D, Yue P, Evangelista M, Settleman J (2014) The cancer stem cell marker aldehyde dehydrogenase is required to maintain a drug-tolerant tumor cell subpopulation. Cancer Res 74(13):3579–3590. https://doi.org/10.1158/0008-5472.Can-13-3456
Regev O, Kidan N, Nicola M, Khamisie H, Ruthardt M, Mahajna J (2021) Mesenchymal soluble factors confer imatinib drug resistance in chronic myelogenous leukemia cells. Arch Med Sci 17(1):266–274. https://doi.org/10.5114/aoms.2020.101042
Reinmuth N, Fan F, Liu W, Parikh AA, Stoeltzing O, Jung YD, Ellis LM (2002) Impact of insulin-like growth factor receptor-I function on angiogenesis, growth, and metastasis of colon cancer. Lab Invest 82(10):1377–1389. https://doi.org/10.1097/01.lab.0000032411.41603.c2
Reya T, Morrison SJ, Clarke MF, Weissman IL (2001) Stem cells, cancer, and cancer stem cells. Nature 414(6859):105–111. https://doi.org/10.1038/35102167
Rodova M, Fu J, Watkins DN, Srivastava RK, Shankar S (2012) Sonic hedgehog signaling inhibition provides opportunities for targeted therapy by sulforaphane in regulating pancreatic cancer stem cell self-renewal. PLoS ONE 7(9):e46083. https://doi.org/10.1371/journal.pone.0046083
Rodrigues P, Macaya I, Bazzocco S, Mazzolini R, Andretta E, Dopeso H, Arango D (2014) RHOA inactivation enhances Wnt signalling and promotes colorectal cancer. Nat Commun 5:5458. https://doi.org/10.1038/ncomms6458
Rolls A, Shechter R, London A, Ziv Y, Ronen A, Levy R, Schwartz M (2007) Toll-like receptors modulate adult hippocampal neurogenesis. Nat Cell Biol 9(9):1081–1088. https://doi.org/10.1038/ncb1629
Roskoski R Jr (2017) Vascular endothelial growth factor (VEGF) and VEGF receptor inhibitors in the treatment of renal cell carcinomas. Pharmacol Res 120:116–132. https://doi.org/10.1016/j.phrs.2017.03.010
Santini R, Vinci MC, Pandolfi S, Penachioni JY, Montagnani V, Olivito B, Stecca B (2012) Hedgehog-GLI signaling drives self-renewal and tumorigenicity of human melanoma-initiating cells. Stem Cells 30(9):1808–1818. https://doi.org/10.1002/stem.1160
Sari IN, Phi LTH, Jun N, Wijaya YT, Lee S, Kwon HY (2018) Hedgehog signaling in cancer: a prospective therapeutic target for eradicating cancer stem cells. Cells, 7(11). https://doi.org/10.3390/cells7110208
Sato C, Zhao G, Ilagan MX (2012) An overview of notch signaling in adult tissue renewal and maintenance. Curr Alzheimer Res 9(2):227–240. https://doi.org/10.2174/156720512799361600
Siddique HR, Feldman DE, Chen CL, Punj V, Tokumitsu H, Machida K (2015) NUMB phosphorylation destabilizes p53 and promotes self-renewal of tumor-initiating cells by a NANOG-dependent mechanism in liver cancer. Hepatology 62(5):1466–1479. https://doi.org/10.1002/hep.27987
Sims-Mourtada J, Izzo JG, Apisarnthanarax S, Wu TT, Malhotra U, Luthra R, Chao KS (2006) Hedgehog: an attribute to tumor regrowth after chemoradiotherapy and a target to improve radiation response. Clin Cancer Res 12(21):6565–6572. https://doi.org/10.1158/1078-0432.Ccr-06-0176
Smith M, Zakrzewski J, James S, Sadelain M (2018) Posttransplant chimeric antigen receptor therapy. Blood 131(10):1045–1052. https://doi.org/10.1182/blood-2017-08-752121
Song S, Maru DM, Ajani JA, Chan CH, Honjo S, Lin HK, Mishra L (2013) Loss of TGF-β adaptor β2SP activates notch signaling and SOX9 expression in esophageal adenocarcinoma. Cancer Res 73(7):2159–2169. https://doi.org/10.1158/0008-5472.Can-12-1962
Stein SJ, Baldwin AS (2013) Deletion of the NF-κB subunit p65/RelA in the hematopoietic compartment leads to defects in hematopoietic stem cell function. Blood 121(25):5015–5024. https://doi.org/10.1182/blood-2013-02-486142
Sun M, Yang C, Zheng J, Wang M, Chen M, Le DQS, Bünger CE (2015) Enhanced efficacy of chemotherapy for breast cancer stem cells by simultaneous suppression of multidrug resistance and antiapoptotic cellular defense. Acta Biomater 28:171–182. https://doi.org/10.1016/j.actbio.2015.09.029
Sun SC, Chang JH, Jin J (2013) Regulation of nuclear factor-κB in autoimmunity. Trends Immunol 34(6):282–289. https://doi.org/10.1016/j.it.2013.01.004
Sun Y (2016) Tumor microenvironment and cancer therapy resistance. Cancer Lett 380(1):205–215. https://doi.org/10.1016/j.canlet.2015.07.044
Takeda H, Okada M, Suzuki S, Kuramoto K, Sakaki H, Watarai H, Kitanaka C (2016) Rho-Associated protein kinase (ROCK) inhibitors inhibit survivin expression and sensitize pancreatic cancer stem cells to gemcitabine. Anticancer Res 36(12):6311–6318. https://doi.org/10.21873/anticanres.11227
Varnat F, Duquet A, Malerba M, Zbinden M, Mas C, Gervaz P, Ruiz i Altaba, A. (2009) Human colon cancer epithelial cells harbour active HEDGEHOG-GLI signalling that is essential for tumour growth, recurrence, metastasis and stem cell survival and expansion. EMBO Mol Med 1(6–7):338–351. https://doi.org/10.1002/emmm.200900039
Vermeulen L, De Sousa EMF, van der Heijden M, Cameron K, de Jong JH, Borovski T, Medema JP (2010) Wnt activity defines colon cancer stem cells and is regulated by the microenvironment. Nat Cell Biol 12(5):468–476. https://doi.org/10.1038/ncb2048
Wang T, Fahrmann JF, Lee H, Li YJ, Tripathi SC, Yue C, Yu H (2018) JAK/STAT3-Regulated fatty acid β-Oxidation is critical for breast cancer stem cell Self-renewal and Chemoresistance. Cell Metab 27(1):136-150.e135. https://doi.org/10.1016/j.cmet.2017.11.001
Wei W, Chua MS, Grepper S, So SK (2009) Blockade of Wnt-1 signaling leads to anti-tumor effects in hepatocellular carcinoma cells. Mol Cancer 8:76. https://doi.org/10.1186/1476-4598-8-76
Wright TM, Brannon AR, Gordan JD, Mikels AJ, Mitchell C, Chen S, Rathmell WK (2009) Ror2, a developmentally regulated kinase, promotes tumor growth potential in renal cell carcinoma. Oncogene 28(27):2513–2523. https://doi.org/10.1038/onc.2009.116
Xiao YS, Zeng D, Liang YK, Wu Y, Li MF, Qi YZ, Zhang GJ (2019) Major vault protein is a direct target of Notch1 signaling and contributes to chemoresistance in triple-negative breast cancer cells. Cancer Lett 440–441:156–167. https://doi.org/10.1016/j.canlet.2018.09.031
Yamazaki H, Xu CW, Naito M, Nishida H, Okamoto T, Ghani FI, Morimoto C (2011) Regulation of cancer stem cell properties by CD9 in human B-acute lymphoblastic leukemia. Biochem Biophys Res Commun 409(1):14–21. https://doi.org/10.1016/j.bbrc.2011.04.098
Yan H, Bu P (2018) Non-coding RNAs in cancer stem cells. Cancer Lett 421:121–126. https://doi.org/10.1016/j.canlet.2018.01.027
Yoon C, Park DJ, Schmidt B, Thomas NJ, Lee HJ, Kim TS, Yoon SS (2014) CD44 expression denotes a subpopulation of gastric cancer cells in which Hedgehog signaling promotes chemotherapy resistance. Clin Cancer Res 20(15):3974–3988. https://doi.org/10.1158/1078-0432.Ccr-14-0011
Yoshikawa K, Noguchi K, Nakano Y, Yamamura M, Takaoka K, Hashimoto-Tamaoki T, Kishimoto H (2015) The Hippo pathway transcriptional co-activator, YAP, confers resistance to cisplatin in human oral squamous cell carcinoma. Int J Oncol 46(6):2364–2370. https://doi.org/10.3892/ijo.2015.2948
Yousefnia S, Momenzadeh S, Seyed Forootan F, Ghaedi K, Nasr Esfahani MH (2018) The influence of peroxisome proliferator-activated receptor γ (PPARγ) ligands on cancer cell tumorigenicity. Gene 649:14–22. https://doi.org/10.1016/j.gene.2018.01.018
Yu FX, Zhao B, Guan KL (2015) Hippo pathway in organ size control, tissue homeostasis, and cancer. Cell 163(4):811–828. https://doi.org/10.1016/j.cell.2015.10.044
Zhan T, Huang X, Tian X, Chen X, Ding Y, Luo H, Zhang Y (2018) Downregulation of MicroRNA-455-3p Links to Proliferation and Drug Resistance of Pancreatic Cancer Cells via Targeting TAZ. Mol Ther Nucleic Acids 10:215–226. https://doi.org/10.1016/j.omtn.2017.12.002
Zhang S, Chen L, Cui B, Chuang HY, Yu J, Wang-Rodriguez J, Kipps TJ (2012) ROR1 is expressed in human breast cancer and associated with enhanced tumor-cell growth. PLoS ONE 7(3):e31127. https://doi.org/10.1371/journal.pone.0031127
Zhao C, Chen A, Jamieson CH, Fereshteh M, Abrahamsson A, Blum J, Reya T (2009) Hedgehog signalling is essential for maintenance of cancer stem cells in myeloid leukaemia. Nature 458(7239):776–779. https://doi.org/10.1038/nature07737
Zhao Y, Zhao L, Ischenko I, Bao Q, Schwarz B, Nieß H, Camaj P (2015) Antisense inhibition of microRNA-21 and microRNA-221 in tumor-initiating stem-like cells modulates tumorigenesis, metastasis, and chemotherapy resistance in pancreatic cancer. Target Oncol 10(4):535–548. https://doi.org/10.1007/s11523-015-0360-2
Zhong Y, Shen S, Zhou Y, Mao F, Lin Y, Guan J, Sun Q (2016) NOTCH1 is a poor prognostic factor for breast cancer and is associated with breast cancer stem cells. Onco Targets Ther 9:6865–6871. https://doi.org/10.2147/ott.S109606
Zhou Q, Zhou Y, Liu X, Shen Y (2017) GDC-0449 improves the antitumor activity of nano-doxorubicin in pancreatic cancer in a fibroblast-enriched microenvironment. Sci Rep 7(1):13379. https://doi.org/10.1038/s41598-017-13869-0
Author information
Authors and Affiliations
Corresponding authors
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Editor(s) (if applicable) and The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Zahra, M.H., Nawara, H.M., Hassan, G., Afify, S.M., Seno, A., Seno, M. (2022). Cancer Stem Cells Contribute to Drug Resistance in Multiple Different Ways. In: Tanabe, S. (eds) Cancer Stem Cell Markers and Related Network Pathways. Advances in Experimental Medicine and Biology, vol 1393. Springer, Cham. https://doi.org/10.1007/978-3-031-12974-2_6
Download citation
DOI: https://doi.org/10.1007/978-3-031-12974-2_6
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-12973-5
Online ISBN: 978-3-031-12974-2
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)