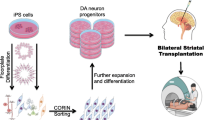
Abstract
Human aging is the most important factor leading to an increased incidence of neurodegenerative diseases worldwide. One common structural hallmark of neurodegenerative disorders is the progressive loss of distinct vulnerable neural cells most likely linked to pathological aggregates of specific proteins. The most prevalent neurodegenerative movement disorder is Parkinson’s disease (PD) characterized by cardinal motor symptoms such as bradykinesia, rigidity, and tremor. The current gold-standard therapy applying Levodopa results in severe adverse effects leading to hyper- and dyskinesia as well as a decreasing therapeutic response. Due to an intraneuronal deposition of alpha-synuclein in dopaminergic midbrain neurons and its consequent severe loss of these cells, PD represents the prototypical disease of the brain for cell-based therapies using a defined neurotransmitter phenotype. In the late 1960s, first pioneer cell-based therapies were initiated leading to transplantation approaches of human fetal ventral mesencephalic tissues in the 1990s for cellular replacement in PD. Despite initial promising outcomes based on first proof-of-concept transplantation efforts in a low number of PD patients, larger randomized double-blind follow-up studies were not able to demonstrate a clinical meaningful benefit for transplanted PD patients. Notably, grafted fetal cells developed inclusions of synuclein several years postgrafting, thereby increasing the pressure to search for alternate cell sources suitable for neural transplantation. After the discovery of human embryonic stem cells in the late 1990s, Takahashi and Yamanaka succeeded to generate somatic cell-derived human-induced pluripotent stem cells (hiPSCs) in 2007. Initial preclinical studies in nonhuman primates demonstrated the high restorative potential of hiPSCs as prospective cell source for transplantation. In 2018, the first in-human studies for hiPSC transplantation in Parkinson’s disease were initiated in Japan. In 2020, a single case was reported in the New England Journal of Medicine describing the successful autologous transplantation of hiPSC-derived dopaminergic neurons in a PD patient. In this review, we will highlight the history as well as the current efforts toward cell-based therapy in PD with a special emphasis on important biological, clinical, and ethical aspects of this regenerative cell-based approach.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
Notes
- 1.
Dugger and Dickson (2017)
- 2.
Pereira, Ferreiro, Cardoso, & de Oliveira (2004), p. 97
- 3.
Rinne (1993), p. 31
- 4.
Heumann et al. (2014), p. 472
- 5.
Sharma (2019), p. 1479
- 6.
Gage (2000), p. 1433
- 7.
Olson and Seiger (1972), p. 175
- 8.
Olson and Seiger (1975), p. 141
- 9.
Seiger and Olson (1975), p. 325
- 10.
Lancaster and Knoblich (2014), p. 2329
- 11.
Takahashi et al. (2007), p. 861
- 12.
Spillantini et al. (1997), p. 839
- 13.
Lesage and Brice (2009), p. R48
- 14.
Kiely et al. (2013), p. 753
- 15.
Kruger et al. (1998), p. 106
- 16.
Pasanen et al. (2014), p. 2180 e1–5
- 17.
Polymeropoulos et al. (1997), p. 2045
- 18.
Zarranz et al. (2004), p. 164
- 19.
Proukakis et al. (2013), p. 1062
- 20.
Jankovic (2008), p. 368
- 21.
Baba et al. (1998), p. 879
- 22.
Lindvall (2016), p.30
- 23.
Benabid (2003), p. 696
- 24.
Backlund et al. (1985), p. 169
- 25.
Kontur, Leranth, Redmond, Roth, & Robbins (1993), p. 172
- 26.
Lindvall et al. (1990), p. 574
- 27.
Kordower et al. (1998), p. 383
- 28.
Kordower et al. (1995), p. 1118
- 29.
Brundin et al. (2000), p. 1380
- 30.
Freed et al. (2001), p. 710
- 31.
Olanow et al. (2003), p. 403
- 32.
Kordower, Chu, Hauser, Freeman, & Olanow (2008)), p. 504
- 33.
Li et al. (2008), p. 501
- 34.
Li et al. (2010), p. 1091
- 35.
Frodl, Nakao, & Brundin (1994), p. 2393
- 36.
Nakao, Frodl, Duan, Widner, & Brundin (1994), p. 12408
- 37.
Wennberg et al. (2001), p. 1797
- 38.
Thomson et al. (1998), p. 1145
- 39.
Evans and Kaufman (1981), p. 154
- 40.
Itskovitz-Eldor et al. (2000), p. 88
- 41.
Schuldiner, Yanuka, Itskovitz-Eldor, Melton, & Benvenisty (2000), p. 11307
- 42.
Hentze et al. (2009), p. 198
- 43.
Prokhorova et al. (2009), p. 47
- 44.
Reubinoff et al. (2001), p. 1134
- 45.
Zhang, Wernig, Duncan, Brustle, & Thomson (2001), p. 1129
- 46.
Englund, Fricker-Gates, Lundberg, Bjorklund, & Wictorin (2002), p. 1
- 47.
Kawasaki et al. (2000), p. 31
- 48.
Yan et al. (2005), p. 781
- 49.
Barker, Drouin-Ouellet, & Parmar (2015), 492
- 50.
Park et al. (2005), p. 1265
- 51.
Roy et al. (2006), p. 1259
- 52.
Friling et al. (2009), p. 7613
- 53.
K. Takahashi and Yamanaka (2006), p. 663
- 54.
Tokuzawa et al. (2003), p. 2699
- 55.
Winner, Marchetto, Winkler, & Gage (2014), p. R27
- 56.
Sanchez-Danes et al. (2012), p. 56
- 57.
Hu, Du, & Zhang (2009), p. 1614
- 58.
Brazdis et al. (2020), p. 1180
- 59.
Simmnacher et al. (2020), p. 113466
- 60.
Sommer et al. (2018), p. 123
- 61.
Prots et al. (2018), p. 7813
- 62.
Rubenstein (2013)
- 63.
Eiraku et al. (2008), p. 519
- 64.
Hu and Zhang (2010), p. 123
- 65.
Renner et al. (2017), p. 1316
- 66.
Lancaster et al. (2013), p. 373
- 67.
Qian et al. (2016), p. 1238
- 68.
Kim, Cha, & Chandrasegaran (1996), p. 1156
- 69.
Bibikova et al. (2001), p. 289
- 70.
Boch et al. (2009), p. 1509
- 71.
Christian et al. (2010), p. 757
- 72.
Bedell et al. (2012), p. 114
- 73.
Ding et al. (2013), p. 238
- 74.
Sun and Zhao (2014), p. 1048
- 75.
Yee (2016), p. 3239
- 76.
Doudna and Charpentier (2014), p. 1258096
- 77.
Barrangou et al. (2007), p. 1709
- 78.
Deltcheva et al. (2011), p. 602
- 79.
Mali et al. (2013), p. 957
- 80.
Cong et al. (2013), p. 819
- 81.
Chambers et al. (2009), p. 275
- 82.
Tabar et al. (2008), p. 379
- 83.
Ono et al. (2007), p. 3213
- 84.
Kriks et al. (2011), p. 547
- 85.
Grealish et al. (2014), p. 653
- 86.
Wernig et al. (2008), p. 5856
- 87.
Doi et al. (2014), p. 337
- 88.
Emborg et al. (2013), p. 646
- 89.
Morizane et al. (2013), p. 283
- 90.
Sundberg et al. (2013), p. 1548
- 91.
Hallett et al. (2015), p. 269
- 92.
Mansour et al. (2018), p. 432
- 93.
Daviaud et al. (2018), p. ENEURO.0219–18.2018
- 94.
Barker et al. (2017), p. 569
- 95.
J. Takahashi (2020), p. 18
- 96.
Schweitzer et al. (2020), p. 1926
- 97.
Ginn et al. (2018), p. e3015
- 98.
Hacein-Bey Abina et al. (2015), p. 1550
- 99.
Porter et al. (2011), p. 725
- 100.
Moradi et al. (2019), p. 341
- 101.
Lindvall (2015), p. 20140370
- 102.
Yoshihara et al. (2017), p. 7
- 103.
Volarevic et al. (2018), p. 36
- 104.
Liang and Zhang (2013), p. 149
- 105.
Carcamo-Orive et al. (2017), p. 518
- 106.
Beers et al. (2015), p. 113119
- 107.
Prescott (2011), p. 1575
- 108.
Song et al. (2020), p. 904
- 109.
Chaudhuri et al. (2006), p. 235
- 110.
Grady (2005), p. 1681
- 111.
Jankovic et al. (2020), p. 1312
References
Baba M, Nakajo S, Tu PH, Tomita T, Nakaya K, Lee VM, Trojanowski JQ, Iwatsubo T (1998) Aggregation of alpha-synuclein in Lewy bodies of sporadic Parkinson's disease and dementia with Lewy bodies. Am J Pathol 152:879–884. https://www.ncbi.nlm.nih.gov/pubmed/9546347
Backlund EO, Granberg PO, Hamberger B, Knutsson E, Martensson A, Sedvall G, Seiger A, Olson L (1985) Transplantation of adrenal medullary tissue to striatum in parkinsonism. First clinical trials J Neurosurg 62:169–173. https://www.ncbi.nlm.nih.gov/pubmed/257855810.3171/jns.1985.62.2.0169
Barker RA, Drouin-Ouellet J, Parmar M (2015) Cell-based therapies for Parkinson disease-past insights and future potential. Nat Rev Neurol 11:492–503. https://www.ncbi.nlm.nih.gov/pubmed/2624003610.1038/nrneurol.2015.123
Barker RA, Parmar M, Studer L, Takahashi J (2017) Human trials of stem cell-derived dopamine neurons for Parkinson’s disease: Dawn of a new era. Cell Stem Cell 21:569–573. https://www.ncbi.nlm.nih.gov/pubmed/2910001010.1016/j.stem.2017.09.014
Barrangou R, Fremaux C, Deveau H, Richards M, Boyaval P, Moineau S, Romero DA, Horvath P (2007) CRISPR provides acquired resistance against viruses in prokaryotes. Science 315:1709–1712. https://www.ncbi.nlm.nih.gov/pubmed/1737980810.1126/science.1138140
Bedell VM, Wang Y, Campbell JM, Poshusta TL, Starker CG, Krug RG 2nd, Tan W, Penheiter SG, Ma AC, Leung AY, Fahrenkrug SC, Carlson DF, Voytas DF, Clark KJ, Essner JJ, Ekker SC (2012) In vivo genome editing using a high-efficiency TALEN system. Nature 491:114–118. https://www.ncbi.nlm.nih.gov/pubmed/2300089910.1038/nature11537
Beers J, Linask KL, Chen JA, Siniscalchi LI, Lin Y, Zheng W, Rao M, Chen G (2015) A cost-effective and efficient reprogramming platform for large-scale production of integration-free human induced pluripotent stem cells in chemically defined culture. Sci Rep 5:11319. https://www.ncbi.nlm.nih.gov/pubmed/2606657910.1038/srep11319
Benabid AL (2003) Deep brain stimulation for Parkinson’s disease. Curr Opin Neurobiol 13:696–706. https://www.ncbi.nlm.nih.gov/pubmed/1466237110.1016/j.conb.2003.11.001
Bibikova M, Carroll D, Segal DJ, Trautman JK, Smith J, Kim YG, Chandrasegaran S (2001) Stimulation of homologous recombination through targeted cleavage by chimeric nucleases. Mol Cell Biol 21:289–297. https://www.ncbi.nlm.nih.gov/pubmed/1111320310.1128/MCB.21.1.289-297.2001
Boch J, Scholze H, Schornack S, Landgraf A, Hahn S, Kay S, Lahaye T, Nickstadt A, Bonas U (2009) Breaking the code of DNA binding specificity of TAL-type III effectors. Science 326:1509–1512. https://www.ncbi.nlm.nih.gov/pubmed/1993310710.1126/science.1178811
Brazdis RM, Alecu JE, Marsch D, Dahms A, Simmnacher K, Lorentz S, Brendler A, Schneider Y, Marxreiter F, Roybon L, Winner B, Xiang W, Prots I (2020) Demonstration of brain region-specific neuronal vulnerability in human iPSC-based model of familial Parkinson’s disease. Hum Mol Genet 29:1180–1191. https://www.ncbi.nlm.nih.gov/pubmed/3216028710.1093/hmg/ddaa039
Brundin P, Pogarell O, Hagell P, Piccini P, Widner H, Schrag A, Kupsch A, Crabb L, Odin P, Gustavii B, Bjorklund A, Brooks DJ, Marsden CD, Oertel WH, Quinn NP, Rehncrona S, Lindvall O (2000) Bilateral caudate and putamen grafts of embryonic mesencephalic tissue treated with lazaroids in Parkinson's disease. Brain 123(Pt 7):1380–1390. https://www.ncbi.nlm.nih.gov/pubmed/1086905010.1093/brain/123.7.1380
Carcamo-Orive I, Hoffman GE, Cundiff P, Beckmann ND, D’souza SL, Knowles JW, Patel A, Papatsenko D, Abbasi F, Reaven GM, Whalen S, Lee P, Shahbazi M, Henrion MYR, Zhu K, Wang S, Roussos P, Schadt EE, Pandey G, Chang R, Quertermous T, Lemischka I (2017) Analysis of transcriptional variability in a large human iPSC library reveals genetic and non-genetic determinants of heterogeneity. Cell Stem Cell 20(518–532):e519. https://www.ncbi.nlm.nih.gov/pubmed/2801779610.1016/j.stem.2016.11.005
Chambers SM, Fasano CA, Papapetrou EP, Tomishima M, Sadelain M, Studer L (2009) Highly efficient neural conversion of human ES and iPS cells by dual inhibition of SMAD signaling. Nat Biotechnol 27:275–280. https://www.ncbi.nlm.nih.gov/pubmed/1925248410.1038/nbt.1529
Chaudhuri KR, Healy DG, Schapira AH, National Institute for Clinical E (2006) Non-motor symptoms of Parkinson's disease: diagnosis and management. Lancet Neurol 5:235–245. https://www.ncbi.nlm.nih.gov/pubmed/1648837910.1016/S1474-4422(06)70373-8
Christian M, Cermak T, Doyle EL, Schmidt C, Zhang F, Hummel A, Bogdanove AJ, Voytas DF (2010) Targeting DNA double-strand breaks with TAL effector nucleases. Genetics 186:757–761. https://www.ncbi.nlm.nih.gov/pubmed/2066064310.1534/genetics.110.120717
Cong L, Ran FA, Cox D, Lin S, Barretto R, Habib N, Hsu PD, Wu X, Jiang W, Marraffini LA, Zhang F (2013) Multiplex genome engineering using CRISPR/Cas systems. Science 339:819–823. https://www.ncbi.nlm.nih.gov/pubmed/2328771810.1126/science.1231143
Daviaud N, Friedel RH, Zou H (2018) Vascularization and Engraftment of Transplanted Human Cerebral Organoids in Mouse Cortex. eNeuro:5. https://www.ncbi.nlm.nih.gov/pubmed/3046033110.1523/ENEURO.0219-18.2018
Deltcheva E, Chylinski K, Sharma CM, Gonzales K, Chao Y, Pirzada ZA, Eckert MR, Vogel J, Charpentier E (2011) CRISPR RNA maturation by trans-encoded small RNA and host factor RNase III. Nature 471:602–607. https://www.ncbi.nlm.nih.gov/pubmed/2145517410.1038/nature09886
Ding Q, Lee YK, Schaefer EA, Peters DT, Veres A, Kim K, Kuperwasser N, Motola DL, Meissner TB, Hendriks WT, Trevisan M, Gupta RM, Moisan A, Banks E, Friesen M, Schinzel RT, Xia F, Tang A, Xia Y, Figueroa E, Wann A, Ahfeldt T, Daheron L, Zhang F, Rubin LL, Peng LF, Chung RT, Musunuru K, Cowan CA (2013) A TALEN genome-editing system for generating human stem cell-based disease models. Cell Stem Cell 12:238–251. https://www.ncbi.nlm.nih.gov/pubmed/2324648210.1016/j.stem.2012.11.011
Doi D, Samata B, Katsukawa M, Kikuchi T, Morizane A, Ono Y, Sekiguchi K, Nakagawa M, Parmar M, Takahashi J (2014) Isolation of human induced pluripotent stem cell-derived dopaminergic progenitors by cell sorting for successful transplantation. Stem Cell Reports 2:337–350. https://www.ncbi.nlm.nih.gov/pubmed/2467275610.1016/j.stemcr.2014.01.013
Doudna JA, Charpentier E (2014) Genome editing. The new frontier of genome engineering with CRISPR-Cas9. Science 346:1258096. https://www.ncbi.nlm.nih.gov/pubmed/2543077410.1126/science.1258096
Dugger BN, Dickson DW (2017) Pathology of neurodegenerative diseases. Cold Spring Harb Perspect Biol 9. https://www.ncbi.nlm.nih.gov/pubmed/2806256310.1101/cshperspect.a028035
Eiraku M, Watanabe K, Matsuo-Takasaki M, Kawada M, Yonemura S, Matsumura M, Wataya T, Nishiyama A, Muguruma K, Sasai Y (2008) Self-organized formation of polarized cortical tissues from ESCs and its active manipulation by extrinsic signals. Cell Stem Cell 3:519–532. https://www.ncbi.nlm.nih.gov/pubmed/1898396710.1016/j.stem.2008.09.002
Emborg ME, Liu Y, Xi J, Zhang X, Yin Y, Lu J, Joers V, Swanson C, Holden JE, Zhang SC (2013) Induced pluripotent stem cell-derived neural cells survive and mature in the nonhuman primate brain. Cell Rep 3:646–650. https://www.ncbi.nlm.nih.gov/pubmed/2349944710.1016/j.celrep.2013.02.016
Englund U, Fricker-Gates RA, Lundberg C, Bjorklund A, Wictorin K (2002) Transplantation of human neural progenitor cells into the neonatal rat brain: extensive migration and differentiation with long-distance axonal projections. Exp Neurol 173:1–21. https://www.ncbi.nlm.nih.gov/pubmed/1177193510.1006/exnr.2001.7750
Evans MJ, Kaufman MH (1981) Establishment in culture of pluripotential cells from mouse embryos. Nature 292:154–156. https://www.ncbi.nlm.nih.gov/pubmed/724268110.1038/292154a0
Freed CR, Greene PE, Breeze RE, Tsai WY, Dumouchel W, Kao R, Dillon S, Winfield H, Culver S, Trojanowski JQ, Eidelberg D, Fahn S (2001) Transplantation of embryonic dopamine neurons for severe Parkinson’s disease. N Engl J Med 344:710–719. https://www.ncbi.nlm.nih.gov/pubmed/1123677410.1056/NEJM200103083441002
Friling S, Andersson E, Thompson LH, Jonsson ME, Hebsgaard JB, Nanou E, Alekseenko Z, Marklund U, Kjellander S, Volakakis N, Hovatta O, El Manira A, Bjorklund A, Perlmann T, Ericson J (2009) Efficient production of mesencephalic dopamine neurons by Lmx1a expression in embryonic stem cells. Proc Natl Acad Sci U S A 106:7613–7618. https://www.ncbi.nlm.nih.gov/pubmed/1938378910.1073/pnas.0902396106
Frodl EM, Nakao N, Brundin P (1994) Lazaroids improve the survival of cultured rat embryonic mesencephalic neurones. Neuroreport 5:2393–2396. https://www.ncbi.nlm.nih.gov/pubmed/788106610.1097/00001756-199411000-00045
Gage FH (2000) Mammalian neural stem cells. Science 287:1433–1438. https://www.ncbi.nlm.nih.gov/pubmed/1068878310.1126/science.287.5457.1433
Ginn SL, Amaya AK, Alexander IE, Edelstein M, Abedi MR (2018) Gene therapy clinical trials worldwide to 2017: an update. J Gene Med 20:e3015. https://www.ncbi.nlm.nih.gov/pubmed/2957537410.1002/jgm.3015
Grady C (2005) Payment of clinical research subjects. J Clin Invest 115:1681–1687. https://www.ncbi.nlm.nih.gov/pubmed/1600724410.1172/JCI25694
Grealish S, Diguet E, Kirkeby A, Mattsson B, Heuer A, Bramoulle Y, Van Camp N, Perrier AL, Hantraye P, Bjorklund A, Parmar M (2014) Human ESC-derived dopamine neurons show similar preclinical efficacy and potency to fetal neurons when grafted in a rat model of Parkinson's disease. Cell Stem Cell 15:653–665. https://www.ncbi.nlm.nih.gov/pubmed/2551746910.1016/j.stem.2014.09.017
Hacein-Bey Abina S, Gaspar HB, Blondeau J, Caccavelli L, Charrier S, Buckland K, Picard C, Six E, Himoudi N, Gilmour K, Mcnicol AM, Hara H, Xu-Bayford J, Rivat C, Touzot F, Mavilio F, Lim A, Treluyer JM, Heritier S, Lefrere F, Magalon J, Pengue-Koyi I, Honnet G, Blanche S, Sherman EA, Male F, Berry C, Malani N, Bushman FD, Fischer A, Thrasher AJ, Galy A, Cavazzana M (2015) Outcomes following gene therapy in patients with severe Wiskott-Aldrich syndrome. JAMA 313:1550–1563. https://www.ncbi.nlm.nih.gov/pubmed/2589805310.1001/jama.2015.3253
Hallett PJ, Deleidi M, Astradsson A, Smith GA, Cooper O, Osborn TM, Sundberg M, Moore MA, Perez-Torres E, Brownell AL, Schumacher JM, Spealman RD, Isacson O (2015) Successful function of autologous iPSC-derived dopamine neurons following transplantation in a non-human primate model of Parkinson's disease. Cell Stem Cell 16:269–274. https://www.ncbi.nlm.nih.gov/pubmed/2573224510.1016/j.stem.2015.01.018
Hentze H, Soong PL, Wang ST, Phillips BW, Putti TC, Dunn NR (2009) Teratoma formation by human embryonic stem cells: evaluation of essential parameters for future safety studies. Stem Cell Res 2:198–210. https://www.ncbi.nlm.nih.gov/pubmed/1939359310.1016/j.scr.2009.02.002
Heumann R, Moratalla R, Herrero MT, Chakrabarty K, Drucker-Colin R, Garcia-Montes JR, Simola N, Morelli M (2014) Dyskinesia in Parkinson's disease: mechanisms and current non-pharmacological interventions. J Neurochem 130:472–489. https://www.ncbi.nlm.nih.gov/pubmed/2477303110.1111/jnc.12751
Hu BY, Zhang SC (2010) Directed differentiation of neural-stem cells and subtype-specific neurons from hESCs. Methods Mol Biol 636:123–137. https://www.ncbi.nlm.nih.gov/pubmed/2033652010.1007/978-1-60761-691-7_8
Itskovitz-Eldor J, Schuldiner M, Karsenti D, Eden A, Yanuka O, Amit M, Soreq H, Benvenisty N (2000) Differentiation of human embryonic stem cells into embryoid bodies compromising the three embryonic germ layers. Mol Med 6:88–95. https://www.ncbi.nlm.nih.gov/pubmed/10859025
Jankovic J (2008) Parkinson's disease: clinical features and diagnosis. J Neurol Neurosurg Psychiatry 79:368–376. https://www.ncbi.nlm.nih.gov/pubmed/1834439210.1136/jnnp.2007.131045
Jankovic J, Okun MS, Kordower JH (2020) Stem cells: scientific and ethical quandaries of a personalized approach to Parkinson's disease. Mov Disord 35:1312–1314. https://www.ncbi.nlm.nih.gov/pubmed/3252966210.1002/mds.28187
Kawasaki H, Mizuseki K, Nishikawa S, Kaneko S, Kuwana Y, Nakanishi S, Nishikawa SI, Sasai Y (2000) Induction of midbrain dopaminergic neurons from ES cells by stromal cell-derived inducing activity. Neuron 28:31–40. https://www.ncbi.nlm.nih.gov/pubmed/1108698110.1016/s0896-6273(00)00083-0
Kiely AP, Asi YT, Kara E, Limousin P, Ling H, Lewis P, Proukakis C, Quinn N, Lees AJ, Hardy J, Revesz T, Houlden H, Holton JL (2013) Alpha-Synucleinopathy associated with G51D SNCA mutation: a link between Parkinson's disease and multiple system atrophy? Acta Neuropathol 125:753–769. https://www.ncbi.nlm.nih.gov/pubmed/2340437210.1007/s00401-013-1096-7
Kim YG, Cha J, Chandrasegaran S (1996) Hybrid restriction enzymes: zinc finger fusions to Fok I cleavage domain. Proc Natl Acad Sci U S A 93:1156–1160. https://www.ncbi.nlm.nih.gov/pubmed/857773210.1073/pnas.93.3.1156
Kontur PJ, Leranth C, Redmond DE Jr, Roth RH, Robbins RJ (1993) Tyrosine hydroxylase immunoreactivity and monoamine and metabolite levels in cryopreserved human fetal ventral mesencephalon. Exp Neurol 121:172–180. https://www.ncbi.nlm.nih.gov/pubmed/768796010.1006/exnr.1993.1084
Kordower JH, Chu Y, Hauser RA, Freeman TB, Olanow CW (2008) Lewy body-like pathology in long-term embryonic nigral transplants in Parkinson's disease. Nat Med 14:504–506. https://www.ncbi.nlm.nih.gov/pubmed/1839196210.1038/nm1747
Kordower JH, Freeman TB, Chen EY, Mufson EJ, Sanberg PR, Hauser RA, Snow B, Olanow CW (1998) Fetal nigral grafts survive and mediate clinical benefit in a patient with Parkinson's disease. Mov Disord 13:383–393. https://www.ncbi.nlm.nih.gov/pubmed/961372610.1002/mds.870130303
Kordower JH, Freeman TB, Snow BJ, Vingerhoets FJ, Mufson EJ, Sanberg PR, Hauser RA, Smith DA, Nauert GM, Perl DP et al (1995) Neuropathological evidence of graft survival and striatal reinnervation after the transplantation of fetal mesencephalic tissue in a patient with Parkinso’s disease. N Engl J Med 332:1118–1124. https://www.ncbi.nlm.nih.gov/pubmed/770028410.1056/NEJM199504273321702
Kriks S, Shim JW, Piao J, Ganat YM, Wakeman DR, Xie Z, Carrillo-Reid L, Auyeung G, Antonacci C, Buch A, Yang L, Beal MF, Surmeier DJ, Kordower JH, Tabar V, Studer L (2011) Dopamine neurons derived from human ES cells efficiently engraft in animal models of Parkinson's disease. Nature 480:547–551. https://www.ncbi.nlm.nih.gov/pubmed/2205698910.1038/nature10648
Kruger R, Kuhn W, Muller T, Woitalla D, Graeber M, Kosel S, Przuntek H, Epplen JT, Schols L, Riess O (1998) Ala30Pro mutation in the gene encoding alpha-synuclein in Parkinson’s disease. Nat Genet 18:106–108. https://www.ncbi.nlm.nih.gov/pubmed/946273510.1038/ng0298-106
Lancaster MA, Knoblich JA (2014) Generation of cerebral organoids from human pluripotent stem cells. Nat Protoc 9:2329–2340. https://www.ncbi.nlm.nih.gov/pubmed/2518863410.1038/nprot.2014.158
Lancaster MA, Renner M, Martin CA, Wenzel D, Bicknell LS, Hurles ME, Homfray T, Penninger JM, Jackson AP, Knoblich JA (2013) Cerebral organoids model human brain development and microcephaly. Nature 501:373–379. https://www.ncbi.nlm.nih.gov/pubmed/2399568510.1038/nature12517
Lesage S, Brice A (2009) Parkinson’s disease: from monogenic forms to genetic susceptibility factors. Hum Mol Genet 18:R48–R59. https://www.ncbi.nlm.nih.gov/pubmed/1929740110.1093/hmg/ddp012
Li JY, Englund E, Holton JL, Soulet D, Hagell P, Lees AJ, Lashley T, Quinn NP, Rehncrona S, Bjorklund A, Widner H, Revesz T, Lindvall O, Brundin P (2008) Lewy bodies in grafted neurons in subjects with Parkinson's disease suggest host-to-graft disease propagation. Nat Med 14:501–503. https://www.ncbi.nlm.nih.gov/pubmed/1839196310.1038/nm1746
Li JY, Englund E, Widner H, Rehncrona S, Bjorklund A, Lindvall O, Brundin P (2010) Characterization of Lewy body pathology in 12- and 16-year-old intrastriatal mesencephalic grafts surviving in a patient with Parkinson's disease. Mov Disord 25:1091–1096. https://www.ncbi.nlm.nih.gov/pubmed/2019864510.1002/mds.23012
Liang G, Zhang Y (2013) Genetic and epigenetic variations in iPSCs: potential causes and implications for application. Cell Stem Cell 13:149–159. https://www.ncbi.nlm.nih.gov/pubmed/2391008210.1016/j.stem.2013.07.001
Lindvall O (2016) Clinical translation of stem cell transplantation in Parkinson’s disease. J Intern Med 279:30–40. https://www.ncbi.nlm.nih.gov/pubmed/2633295910.1111/joim.12415
Lindvall O (2015) Treatment of Parkinson's disease using cell transplantation. Philos Trans R Soc Lond Ser B Biol Sci 370:20140370. https://www.ncbi.nlm.nih.gov/pubmed/2641668110.1098/rstb.2014.0370
Lindvall O, Brundin P, Widner H, Rehncrona S, Gustavii B, Frackowiak R, Leenders KL, Sawle G, Rothwell JC, Marsden CD et al (1990) Grafts of fetal dopamine neurons survive and improve motor function in Parkinson's disease. Science 247:574–577. https://www.ncbi.nlm.nih.gov/pubmed/210552910.1126/science.2105529
Mali P, Esvelt KM, Church GM (2013) Cas9 as a versatile tool for engineering biology. Nat Methods 10:957–963. https://www.ncbi.nlm.nih.gov/pubmed/2407699010.1038/nmeth.2649
Mansour AA, Goncalves JT, Bloyd CW, Li H, Fernandes S, Quang D, Johnston S, Parylak SL, Jin X, Gage FH (2018) An in vivo model of functional and vascularized human brain organoids. Nat Biotechnol 36:432–441. https://www.ncbi.nlm.nih.gov/pubmed/2965894410.1038/nbt.4127
Moradi S, Mahdizadeh H, Saric T, Kim J, Harati J, Shahsavarani H, Greber B, Moore JBT (2019) Research and therapy with induced pluripotent stem cells (iPSCs): social, legal, and ethical considerations. Stem Cell Res Ther 10:341. https://www.ncbi.nlm.nih.gov/pubmed/3175303410.1186/s13287-019-1455-y
Morizane A, Doi D, Kikuchi T, Okita K, Hotta A, Kawasaki T, Hayashi T, Onoe H, Shiina T, Yamanaka S, Takahashi J (2013) Direct comparison of autologous and allogeneic transplantation of iPSC-derived neural cells in the brain of a non-human primate. Stem Cell Reports 1:283–292. https://www.ncbi.nlm.nih.gov/pubmed/2431966410.1016/j.stemcr.2013.08.007
Nakao N, Frodl EM, Duan WM, Widner H, Brundin P (1994) Lazaroids improve the survival of grafted rat embryonic dopamine neurons. Proc Natl Acad Sci U S A 91:12408–12412. https://www.ncbi.nlm.nih.gov/pubmed/780905010.1073/pnas.91.26.12408
Olanow CW, Goetz CG, Kordower JH, Stoessl AJ, Sossi V, Brin MF, Shannon KM, Nauert GM, Perl DP, Godbold J, Freeman TB (2003) A double-blind controlled trial of bilateral fetal nigral transplantation in Parkinson's disease. Ann Neurol 54:403–414. https://www.ncbi.nlm.nih.gov/pubmed/1295327610.1002/ana.10720
Olson L, Seiger A (1972) Brain tissue transplanted to the anterior chamber of the eye. 1. Fluorescence histochemistry of immature catecholamine and 5-hydroxytryptamine neurons reinnervating the rat iris. Z Zellforsch Mikrosk Anat 135:175–194. https://www.ncbi.nlm.nih.gov/pubmed/464252710.1007/BF00315125
Olson L, Seiger A (1975) Brain tissue transplanted to the anterior chanber of the eye: 2. Fluorescence histochemistry of immature catecholamine- and 5-hydroxytryptamine neurons innervating the rat vas deferens. Cell Tissue Res 158:141–150. https://www.ncbi.nlm.nih.gov/pubmed/113185910.1007/BF00219957
Ono Y, Nakatani T, Sakamoto Y, Mizuhara E, Minaki Y, Kumai M, Hamaguchi A, Nishimura M, Inoue Y, Hayashi H, Takahashi J, Imai T (2007) Differences in neurogenic potential in floor plate cells along an anteroposterior location: midbrain dopaminergic neurons originate from mesencephalic floor plate cells. Development 134:3213–3225. https://www.ncbi.nlm.nih.gov/pubmed/1767078910.1242/dev.02879
Park CH, Minn YK, Lee JY, Choi DH, Chang MY, Shim JW, Ko JY, Koh HC, Kang MJ, Kang JS, Rhie DJ, Lee YS, Son H, Moon SY, Kim KS, Lee SH (2005) In vitro and in vivo analyses of human embryonic stem cell-derived dopamine neurons. J Neurochem 92:1265–1276. https://www.ncbi.nlm.nih.gov/pubmed/1571567510.1111/j.1471-4159.2004.03006.x
Pasanen P, Myllykangas L, Siitonen M, Raunio A, Kaakkola S, Lyytinen J, Tienari PJ, Poyhonen M, Paetau A (2014) Novel alpha-synuclein mutation A53E associated with atypical multiple system atrophy and Parkinson's disease-type pathology. Neurobiol Aging 35(2180):e2181–e2185. https://www.ncbi.nlm.nih.gov/pubmed/2474636210.1016/j.neurobiolaging.2014.03.024
Pereira C, Ferreiro E, Cardoso SM, De Oliveira CR (2004) Cell degeneration induced by amyloid-beta peptides: implications for Alzheimer's disease. J Mol Neurosci 23:97–104. https://www.ncbi.nlm.nih.gov/pubmed/1512669510.1385/JMN:23:1-2:097
Polymeropoulos MH, Lavedan C, Leroy E, Ide SE, Dehejia A, Dutra A, Pike B, Root H, Rubenstein J, Boyer R, Stenroos ES, Chandrasekharappa S, Athanassiadou A, Papapetropoulos T, Johnson WG, Lazzarini AM, Duvoisin RC, Di Iorio G, Golbe LI, Nussbaum RL (1997) Mutation in the alpha-synuclein gene identified in families with Parkinson's disease. Science 276:2045–2047. https://www.ncbi.nlm.nih.gov/pubmed/919726810.1126/science.276.5321.2045
Porter DL, Levine BL, Kalos M, Bagg A, June CH (2011) Chimeric antigen receptor-modified T cells in chronic lymphoid leukemia. N Engl J Med 365:725–733. https://www.ncbi.nlm.nih.gov/pubmed/2183094010.1056/NEJMoa1103849
Prescott C (2011) The business of exploiting induced pluripotent stem cells. Philos Trans R Soc Lond Ser B Biol Sci 366:2323–2328. https://www.ncbi.nlm.nih.gov/pubmed/2172713810.1098/rstb.2011.0047
Prokhorova TA, Harkness LM, Frandsen U, Ditzel N, Schroder HD, Burns JS, Kassem M (2009) Teratoma formation by human embryonic stem cells is site dependent and enhanced by the presence of Matrigel. Stem Cells Dev 18:47–54. https://www.ncbi.nlm.nih.gov/pubmed/1839367310.1089/scd.2007.0266
Prots I, Grosch J, Brazdis RM, Simmnacher K, Veber V, Havlicek S, Hannappel C, Krach F, Krumbiegel M, Schutz O, Reis A, Wrasidlo W, Galasko DR, Groemer TW, Masliah E, Schlotzer-Schrehardt U, Xiang W, Winkler J, Winner B (2018) Alpha-Synuclein oligomers induce early axonal dysfunction in human iPSC-based models of synucleinopathies. Proc Natl Acad Sci U S A 115:7813–7818. https://www.ncbi.nlm.nih.gov/pubmed/2999159610.1073/pnas.1713129115
Proukakis C, Dudzik CG, Brier T, Mackay DS, Cooper JM, Millhauser GL, Houlden H, Schapira AH (2013) A novel alpha-synuclein missense mutation in Parkinson disease. Neurology 80:1062–1064. https://www.ncbi.nlm.nih.gov/pubmed/2342732610.1212/WNL.0b013e31828727ba
Qian X, Nguyen HN, Song MM, Hadiono C, Ogden SC, Hammack C, Yao B, Hamersky GR, Jacob F, Zhong C, Yoon KJ, Jeang W, Lin L, Li Y, Thakor J, Berg DA, Zhang C, Kang E, Chickering M, Nauen D, Ho CY, Wen Z, Christian KM, Shi PY, Maher BJ, Wu H, Jin P, Tang H, Song H, Ming GL (2016) Brain-region-specific organoids using mini-bioreactors for modeling ZIKV exposure. Cell 165:1238–1254. https://www.ncbi.nlm.nih.gov/pubmed/2711842510.1016/j.cell.2016.04.032
Renner M, Lancaster MA, Bian S, Choi H, Ku T, Peer A, Chung K, Knoblich JA (2017) Self-organized developmental patterning and differentiation in cerebral organoids. EMBO J 36:1316–1329. https://www.ncbi.nlm.nih.gov/pubmed/2828358210.15252/embj.201694700
Reubinoff BE, Itsykson P, Turetsky T, Pera MF, Reinhartz E, Itzik A, Ben-Hur T (2001) Neural progenitors from human embryonic stem cells. Nat Biotechnol 19:1134–1140. https://www.ncbi.nlm.nih.gov/pubmed/1173178210.1038/nbt1201-1134
Rinne JO (1993) Nigral degeneration in Parkinson's disease. Mov Disord 8(Suppl 1):S31–S35. https://www.ncbi.nlm.nih.gov/pubmed/830230510.1002/mds.870080507
Roy NS, Cleren C, Singh SK, Yang L, Beal MF, Goldman SA (2006) Functional engraftment of human ES cell-derived dopaminergic neurons enriched by coculture with telomerase-immortalized midbrain astrocytes. Nat Med 12:1259–1268. https://www.ncbi.nlm.nih.gov/pubmed/1705770910.1038/nm1495
Rubenstein J, Rakic P (2013) Patterning and cell type specification in the developing CNS and PNS: comprehensive developmental neuroscience. Academic Press 1
Sanchez-Danes A, Consiglio A, Richaud Y, Rodriguez-Piza I, Dehay B, Edel M, Bove J, Memo M, Vila M, Raya A, Izpisua Belmonte JC (2012) Efficient generation of A9 midbrain dopaminergic neurons by lentiviral delivery of LMX1A in human embryonic stem cells and induced pluripotent stem cells. Hum Gene Ther 23:56–69. https://www.ncbi.nlm.nih.gov/pubmed/2187792010.1089/hum.2011.054
Schuldiner M, Yanuka O, Itskovitz-Eldor J, Melton DA, Benvenisty N (2000) Effects of eight growth factors on the differentiation of cells derived from human embryonic stem cells. Proc Natl Acad Sci U S A 97:11307–11312. https://www.ncbi.nlm.nih.gov/pubmed/1102733210.1073/pnas.97.21.11307
Schweitzer JS, Song B, Herrington TM, Park TY, Lee N, Ko S, Jeon J, Cha Y, Kim K, Li Q, Henchcliffe C, Kaplitt M, Neff C, Rapalino O, Seo H, Lee IH, Kim J, Kim T, Petsko GA, Ritz J, Cohen BM, Kong SW, Leblanc P, Carter BS, Kim KS (2020) Personalized iPSC-derived dopamine progenitor cells for Parkinson’s disease. N Engl J Med 382:1926–1932. https://www.ncbi.nlm.nih.gov/pubmed/3240216210.1056/NEJMoa1915872
Seiger A, Olson L (1975) Brain tissue transplanted to the anterior chamber of the eye: 3. Substitution of lacking central noradrenaline input by host iris sympathetic fibers in the isolated cerebral cortex developed in oculo. Cell Tissue Res 159:325–338. https://www.ncbi.nlm.nih.gov/pubmed/109711810.1007/BF00221780
Sharma K (2019) Cholinesterase inhibitors as Alzheimer's therapeutics (review). Mol Med Rep 20:1479–1487. https://www.ncbi.nlm.nih.gov/pubmed/3125747110.3892/mmr.2019.10374
Simmnacher K, Krach F, Schneider Y, Alecu JE, Mautner L, Klein P, Roybon L, Prots I, Xiang W, Winner B (2020) Unique signatures of stress-induced senescent human astrocytes. Exp Neurol 334:113466. https://www.ncbi.nlm.nih.gov/pubmed/3294957210.1016/j.expneurol.2020.113466
Sommer A, Marxreiter F, Krach F, Fadler T, Grosch J, Maroni M, Graef D, Eberhardt E, Riemenschneider MJ, Yeo GW, Kohl Z, Xiang W, Gage FH, Winkler J, Prots I, Winner B (2018) Th17 lymphocytes induce neuronal cell death in a human iPSC-based model of Parkinson's disease. Cell Stem Cell 23(123–131):e126. https://www.ncbi.nlm.nih.gov/pubmed/2997998610.1016/j.stem.2018.06.015
Song B, Cha Y, Ko S, Jeon J, Lee N, Seo H, Park KJ, Lee IH, Lopes C, Feitosa M, Luna MJ, Jung JH, Kim J, Hwang D, Cohen BM, Teicher MH, Leblanc P, Carter BS, Kordower JH, Bolshakov VY, Kong SW, Schweitzer JS, Kim KS (2020) Human autologous iPSC-derived dopaminergic progenitors restore motor function in Parkinson's disease models. J Clin Invest 130:904–920. https://www.ncbi.nlm.nih.gov/pubmed/3171489610.1172/JCI130767
Spillantini MG, Schmidt ML, Lee VM, Trojanowski JQ, Jakes R, Goedert M (1997) Alpha-synuclein in Lewy bodies. Nature 388:839–840. https://www.ncbi.nlm.nih.gov/pubmed/927804410.1038/42166
Sun N, Zhao H (2014) Seamless correction of the sickle cell disease mutation of the HBB gene in human induced pluripotent stem cells using TALENs. Biotechnol Bioeng 111:1048–1053. https://www.ncbi.nlm.nih.gov/pubmed/2392885610.1002/bit.25018
Sundberg M, Bogetofte H, Lawson T, Jansson J, Smith G, Astradsson A, Moore M, Osborn T, Cooper O, Spealman R, Hallett P, Isacson O (2013) Improved cell therapy protocols for Parkinson's disease based on differentiation efficiency and safety of hESC-, hiPSC-, and non-human primate iPSC-derived dopaminergic neurons. Stem Cells 31:1548–1562. https://www.ncbi.nlm.nih.gov/pubmed/2366660610.1002/stem.1415
Tabar V, Tomishima M, Panagiotakos G, Wakayama S, Menon J, Chan B, Mizutani E, Al-Shamy G, Ohta H, Wakayama T, Studer L (2008) Therapeutic cloning in individual parkinsonian mice. Nat Med 14:379–381. https://www.ncbi.nlm.nih.gov/pubmed/1837640910.1038/nm1732
Takahashi J (2020) iPS cell-based therapy for Parkinson's disease: a Kyoto trial. Regen Ther 13:18–22. https://www.ncbi.nlm.nih.gov/pubmed/3349031910.1016/j.reth.2020.06.002
Takahashi K, Okita K, Nakagawa M, Yamanaka S (2007) Induction of pluripotent stem cells from fibroblast cultures. Nat Protoc 2:3081–3089. https://www.ncbi.nlm.nih.gov/pubmed/1807970710.1038/nprot.2007.418
Takahashi K, Yamanaka S (2006) Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 126:663–676. https://www.ncbi.nlm.nih.gov/pubmed/1690417410.1016/j.cell.2006.07.024
Thomson JA, Itskovitz-Eldor J, Shapiro SS, Waknitz MA, Swiergiel JJ, Marshall VS, Jones JM (1998) Embryonic stem cell lines derived from human blastocysts. Science 282:1145–1147. https://www.ncbi.nlm.nih.gov/pubmed/980455610.1126/science.282.5391.1145
Tokuzawa Y, Kaiho E, Maruyama M, Takahashi K, Mitsui K, Maeda M, Niwa H, Yamanaka S (2003) Fbx15 is a novel target of Oct3/4 but is dispensable for embryonic stem cell self-renewal and mouse development. Mol Cell Biol 23:2699–2708. https://www.ncbi.nlm.nih.gov/pubmed/1266557210.1128/mcb.23.8.2699-2708.2003
Volarevic V, Markovic BS, Gazdic M, Volarevic A, Jovicic N, Arsenijevic N, Armstrong L, Djonov V, Lako M, Stojkovic M (2018) Ethical and safety issues of stem cell-based therapy. Int J Med Sci 15:36–45. https://www.ncbi.nlm.nih.gov/pubmed/2933308610.7150/ijms.21666
Wennberg L, Czech KA, Larsson LC, Mirza B, Bennet W, Song Z, Widner H (2001) Effects of immunosuppressive treatment on host responses against intracerebral porcine neural tissue xenografts in rats. Transplantation 71:1797–1806. https://www.ncbi.nlm.nih.gov/pubmed/1145526110.1097/00007890-200106270-00016
Wernig M, Zhao JP, Pruszak J, Hedlund E, Fu D, Soldner F, Broccoli V, Constantine-Paton M, Isacson O, Jaenisch R (2008) Neurons derived from reprogrammed fibroblasts functionally integrate into the fetal brain and improve symptoms of rats with Parkinson's disease. Proc Natl Acad Sci U S A 105:5856–5861. https://www.ncbi.nlm.nih.gov/pubmed/1839119610.1073/pnas.0801677105
Winner B, Marchetto MC, Winkler J, Gage FH (2014) Human-induced pluripotent stem cells pave the road for a better understanding of motor neuron disease. Hum Mol Genet 23:R27–R34. https://www.ncbi.nlm.nih.gov/pubmed/2482170410.1093/hmg/ddu205
Yan Y, Yang D, Zarnowska ED, Du Z, Werbel B, Valliere C, Pearce RA, Thomson JA, Zhang SC (2005) Directed differentiation of dopaminergic neuronal subtypes from human embryonic stem cells. Stem Cells 23:781–790. https://www.ncbi.nlm.nih.gov/pubmed/1591747410.1634/stemcells.2004-0365
Yee JK (2016) Off-target effects of engineered nucleases. FEBS J 283:3239–3248. https://www.ncbi.nlm.nih.gov/pubmed/2720870110.1111/febs.13760
Yoshihara M, Hayashizaki Y, Murakawa Y (2017) Genomic instability of iPSCs: challenges towards their clinical applications. Stem Cell Rev Rep 13:7–16. https://www.ncbi.nlm.nih.gov/pubmed/2759270110.1007/s12015-016-9680-6
Zarranz JJ, Alegre J, Gomez-Esteban JC, Lezcano E, Ros R, Ampuero I, Vidal L, Hoenicka J, Rodriguez O, Atares B, Llorens V, Gomez Tortosa E, Del Ser T, Munoz DG, De Yebenes JG (2004) The new mutation, E46K, of alpha-synuclein causes Parkinson and Lewy body dementia. Ann Neurol 55:164–173. https://www.ncbi.nlm.nih.gov/pubmed/1475571910.1002/ana.10795
Zhang SC, Wernig M, Duncan ID, Brustle O, Thomson JA (2001) In vitro differentiation of transplantable neural precursors from human embryonic stem cells. Nat Biotechnol 19:1129–1133. https://www.ncbi.nlm.nih.gov/pubmed/1173178110.1038/nbt1201-1129
Acknowledgments
Je.W. and Y.S. are graduate students of the research training group 2162 “Neurodevelopment and Vulnerability of the Central Nervous System” of the Deutsche Forschungsgemeinschaft (DFG 270949263/GRK2162). S.T. was a postdoctoral researcher and consequent principle investigator of the research training group 2162 “Neurodevelopment and Vulnerability of the Central Nervous System” of the Deutsche Forschungsgemeinschaft (DFG 270949263/GRK2162). J.W. is a member of the research training group 2162 of the Deutsche Forschungsgemeinschaft (DFG 270949263/GRK2162) and speaker of the Bavarian Research Network for Induced Pluripotent Stem Cells (ForIPS), as well as consequent member of the Bavarian Research Network of Interaction of human brain cells (ForInter). Je.W., Y.S., and S.T are associated with the Bavarian Research Networks for Induced Pluripotent Stem Cells (ForIPS) and for the Interaction of human brain cells (ForInter). The work was supported by the Forschungsstiftung Medizin, University Hospital Erlangen. All figures were created with BioRender.com.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Schneider, Y., Wihan, J., Turan, S., Winkler, J. (2022). Cell-Based Therapy and Genome Editing in Parkinson’s Disease: Quo Vadis?. In: Dederer, HG., Hamburger, D. (eds) Brain Organoids in Research and Therapy. Advances in Neuroethics. Springer, Cham. https://doi.org/10.1007/978-3-030-97641-5_3
Download citation
DOI: https://doi.org/10.1007/978-3-030-97641-5_3
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-97640-8
Online ISBN: 978-3-030-97641-5
eBook Packages: MedicineMedicine (R0)