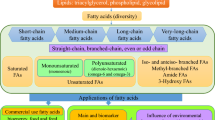
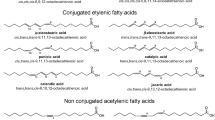
Abstract
Fatty acids are essential components of all living cells and play vital functions in plant and animal organisms. Literature data demonstrate a diverse set of fatty acids in ferns: about 90 structures of fatty acids have been reported for ferns so far. From their ancestors, ferns inherited long-chain polyunsaturated fatty acids, like arachidonic and eicosapentaenoic acids, which are rare or completely absent in the evolutionarily later lineages of gymnosperms and angiosperms. These fatty acids are valuable nutrients for humans making ferns their potential sources. The review summarizes different aspects of fatty acids in ferns (class Polypodiopsida): structural diversity, distribution in complex lipids, developmental and organ specificity, and ecological and environmental factors affecting fatty acid composition and content. The biosynthesis of fatty acids in ferns is poorly investigated, however, and can be deduced from the data established for other plant groups. Finally, the nutritional value of ferns as dietary sources of the long-chain polyunsaturated fatty acids and some other practical applications of fern fatty acids are considered.
Similar content being viewed by others
Abbreviations
- ACP:
-
Acyl-carrier protein
- ARA:
-
Arachidonic acid (20:4n-6)
- DGDG:
-
Digalactosyldiacylglycerol
- DGTS:
-
Diacylglyceryltrimethylhomoserine
- DHA:
-
Docosahexaenoic acid (22:6n-3)
- DW:
-
Dry weight
- EPA:
-
Eicosapentaenoic acid (20:5n-3)
- FAD:
-
Fatty acid desaturase
- LC-PUFA:
-
Long-chain polyunsaturated fatty acid
- MGDG:
-
Monogalactosyldiacylglycerol
- PC:
-
Phosphatidylcholine
- PG:
-
Phosphatidylglycerol
- PE:
-
Phosphatidylethanolamine
- PI:
-
Phosphatidylinositol
- PS:
-
Phosphatidylserine
- PUFA:
-
Polyunsaturated fatty acid
- SQDG:
-
Sulfoquinovosyldiacylglycerol
- TG:
-
Triacylglycerol
- WW:
-
Wet weight
References
Christie WW (2022) Fatty acids and oxylipins. The LipidWeb. https://www.lipidmaps.org/resources/lipidweb/index.php?page=lipids/fa-eic.html. Accessed 16 June 2022
Leray C (2022) Fatty acids. Cyberlipid. http://cyberlipid.gerli.com/description/simple-lipids/fatty-acids/#3. Accessed 16 June 2022
Khotimchenko SV, Vaskovsky VE, Titlyanova TV (2002) Fatty acids of marine algae from the Pacific coast of North California. Bot Mar 45:17–22
van Ginneken VJT, Helsper JPFG, de Visser W, van Keulen H, Brandenburg WA (2011) Polyunsaturated fatty acids in various macroalgal species from north Atlantic and tropical seas. Lipids Health Dis 10:104. https://doi.org/10.1186/1476-511X-10-104
Martins DA, Custodio L, Barreira L, Pereira H, Ben-Hamadou R, Varela J, Abu-Salah KM (2013) Alternative sources of n-3 long-chain polyunsaturated fatty acids in marine microalgae. Mar Drugs 11:2259–2281. https://doi.org/10.3390/md11072259
Biancarosa I, Belghit I, Bruckner CG, Liland NS, Waagbo R, Amlund H, Heesch S, Lock EJ (2018) Chemical characterization of 21 species of marine macroalgae common in Norwegian waters: benefits of and limitations to their potential use in food and feed. J Sci Food Agric 98:2035–2042. https://doi.org/10.1002/jsfa.8798
Dembitsky VM (1993) Lipids of bryophytes. Prog Lipid Res 32:281–356
Lu Y, Eiriksson FF, Thorsteinsdottir M, Simonsen HT (2019) Valuable fatty acids in bryophytes–production, biosynthesis, analysis and applications. Plan Theory 8:524. https://doi.org/10.3390/plants8110524
Wolff RL, Christie WW, Pedrono F, Marpeau AM (1999) Arachidonic, eicosapentaenoic, and biosynthetically related fatty acids in the seed lipids from a primitive gymnosperm, Agathis robusta. Lipids 34:1083–1097. https://doi.org/10.1007/s11745-999-0460-y
Wolff RL, Christie WW (2002) Structures, practical sources (gymnosperm seeds), gas-liquid chromatographic data (equivalent chain lengths), and mass spectrometric characteristics of all-cis Δ5-olefinic acids. Eur J Lipid Sci Technol 104:234–244
Tsevegsuren N, Aitzetmuller K (1997) Unusual Δ5cis-fatty acids in seed oils of Cimicifuga species. J High Res Chromat 20:237–241. https://doi.org/10.1002/jhrc.1240200411
von Aderkas P (1984) Economic history of ostrich fern, Matteuccia struthiopteris, the edible fiddlehead. Econ Bot 38:14–23
Crowe A (1997) A field guide to the native edible plants of New Zealand. Godwit Publishing, Auckland
Khrapko OV (2007) Far Eastern ferns: opportunities for application. Bulletin of the Botanical Garden Institute 1:81–87. http://botsad.ru/media/oldfiles/journal/number1/08.pdf. Accessed 16 May 2022 (In Russian)
Liu Y, Wujisguleng W, Long C (2012) Food uses of ferns in China: a review. Acta Soc Bot Pol 81:263–270. https://doi.org/10.5586/asbp.2012.046
Maroyi A (2014) Not just minor wild edible forest products: consumption of pteridophytes in sub-Saharan Africa. J Ethnobiol Ethnomed 10:78. https://doi.org/10.1186/1746-4269-10-78
Sujarwo W, Lugrayasa IN, Caneva G (2014) Ethnobotanical study of edible ferns used in Bali Indonesia. Asia Pac J Sustain Agric Food Energy 2:1–4
Koide D, Kadoya T (2019) Resource amount and cultural legacy affect spatially unbalanced human use of Japan’s non-timber forest products. Ecol Indic 97:204–210. https://doi.org/10.1016/j.ecolind.2018.10.017
Dyall SC (2015) Long-chain omega-3 fatty acids and the brain: a review of the independent and shared effects of EPA, DPA and DHA. Front Aging Neurosci 7:52. https://doi.org/10.3389/fnagi.2015.00052
Bazinet RP, Metherel AH, Chen CT, Shaikh SR, Nadjar A, Joffre C, Laye S (2020) Brain eicosapentaenoic acid metabolism as a lead for novel therapeutics in major depression. Brain Behav Immun 85:21–28. https://doi.org/10.1016/j.bbi.2019.07.001
Harayama T, Shimizu T (2020) Roles of polyunsaturated fatty acids, from mediators to membranes. J Lipid Res 61:1150–1160. https://doi.org/10.1194/jlr.R120000800
Katz J (1898) Das fette Oel des Rhizoms von Aspidium filix mas. Arch Pharm 236:655–662. https://doi.org/10.1002/ardp.18982360903
Farup P (1904) Ueber die Zusammensetzung des fetten Oeles von Aspidium spinulosum. Arch Pharm 242:17–24. https://doi.org/10.1002/ardp.19042420103
Maizite J (1942) Über das Farnfett. Arch Pharm 280:132–156. https://doi.org/10.1002/ardp.19422800403
Gellerman JL, Schlenk H (1964) The occurrence of arachidonic and related acids in plants. Experientia 20:426–427. https://doi.org/10.1007/BF02152126
Schlenk H, Gellerman JL (1965) Arachidonic, 5,11,14,17-eicosatetraenoic and related acids in plants - identification of unsaturated fatty acids. J Am Oil Chem Soc 42:504–511. https://doi.org/10.1007/BF02540092
Radunz A (1967) Über die Lipide der Pteridophyten – I. Die Isolierung und Identifizierung der Polyensäuren. Phytochemistry 6:399–406
Jamieson GR, Reid EH (1975) The fatty acid composition of fern lipids. Phytochemistry 14:2229–2232. https://doi.org/10.1016/S0031-9422(00)91104-X
Lytle TF, Lytle JS, Caruso A (1976) Hydrocarbons and fatty acids of ferns. Phytochemistry 15:965–970. https://doi.org/10.1016/S0031-9422(00)84381-2
Guervin CM, Laur M-H (1969) Lipochimie vegetale. – Les acides gras des frondes sporiferes du Scolopendrium vulgare Sm. C R Acad Sci Paris 269:583–585
Robinson PM, Smith DL, Safford R, Nichols BW (1973) Lipid metabolism in the fern Polypodium vulgare. Phytochemistry 12:1377–1381. https://doi.org/10.1016/0031-9422(73)80569-2
Haigh WG, Safford R, James AT (1969) Fatty acid composition and biosynthesis in ferns. Biochim Biophys Acta 176:647–650
Gemmrich AR (1977) Fatty acid composition of fern spore lipids. Phytochemistry 16:1044–1046. https://doi.org/10.1016/S0031-9422(00)86721-7
Rozentsvet OA, Bogdanova ES (2009) Influence of ions of lead on growth, the contents and structure of lipids of the membranes of the Matteuccia sthruthiopteris (L.) Todari. Izvestia of Samara Scientific Center of the Russian Academy of Sciences 11:205–211. (In Russian)
Rozentsvet OA, Bogdanova ES, Makurina ON (2010) Influence of cadmium ions on qualitative and quantitative composition of lipids and pigments in the forming leaves of Matteuccia sthruthiopteris. Vestnik Samarskogo Universiteta Estestvenno-Nauchnaya Seriya 6:232–239. (In Russian)
Rozentsvet OA, Bogdanova ES, Murzaeva SV (2011) Composition of lipids and fatty acids in the forming leaves of the fern Matteuccia struthiopteris in the presence of cadmium. Transactions of KarRC RAS 3(97–104) http://transactions.krc.karelia.ru/publ.php?plang=e&id=8769. Accessed 15 May 2022 (In Russian)
Rozentsvet OA, Bogdanova ES, Golovko TK, Zakhozhiy IG (2013) Composition of lipids of the leaves of some representatives of Polypodiophyta in the Polar Urals. Khimija Rastitel’nogo Syr’ja [Chemistry of Plant Raw Material] 4:127–134. http://journal.asu.ru/cw/article/view/jcprm.1304127/147. Accessed 21 June 2022 (In Russian)
Dudareva LV, Nokhsorov VV, Rudikovskaya EG, Petrov KA (2015) Fatty-acid profiles of aerial parts of three horsetail species growing in central and northern Yakutia. Chem Nat Comp 51:220–223. https://doi.org/10.1007/s10600-015-1247-2
Voronkov AS, Ivanova TV (2021) Fatty acids composition of the epiphytic ferns, Platycerium bifurcatum and Asplenium nidus, and the terrestrial fern, Asplenium trichomanes. Am Fern J 111:117–128. https://doi.org/10.1640/0002-8444-111.2.117
Voronkov A, Ivanova T (2022) Significance of lipid fatty acid composition for resistance to winter conditions in Asplenium scolopendrium. Biology 11:507. https://doi.org/10.3390/biology11040507
Bushway AA, Wilson AM, McGann DF, Bushway RJ (1982) The nutrient composition of fresh fiddlehead greens. J Food Sci 47:666–667. https://doi.org/10.1111/j.1365-2621.1982.tb10147.x
DeLong JM, Hodges DM, Prange RK, Forney CF, Toivenon PMA, Bishop MC, Elliot ML, Jordan MA (2011) The unique fatty acid and antioxidant composition of ostrich fern (Matteuccia struthiopteris) fiddleheads. Can J Plant Sci 91:919–930. https://doi.org/10.4141/cjps2010-042
Dvorakova M, Pumprova K, Antoninova Z, Rezek J, Haisel D, Ekrt L, Vanek T, Langhansova L (2021) Nutritional and antioxidant potential of fiddleheads from European ferns. Foods 10:460. https://doi.org/10.3390/foods10020460
Nekrasov EV, Svetashev VI (2021) Edible Far Eastern ferns as a dietary source of long-chain polyunsaturated fatty acids. Foods 10:1220. https://doi.org/10.3390/foods10061220
Salehzadeh A, Naeemi AS, Arasteh A (2014) Biodiesel production from Azolla filiculoides (water fern). Trop J Pharm Res 13:957–960. https://doi.org/10.4314/tjpr.v13i6.19
Brouwer P, van der Werf A, Schluepmann H, Reichart GJ, Nierop KGJ (2016) Lipid yield and composition of Azolla filiculoides and the implications for biodiesel production. Bioenergy Res 9:369–377. https://doi.org/10.1007/s12155-015-9665-3
Miranda AF, Liu Z, Rochfort S, Mouradov A (2018) Lipid production in aquatic plant Azolla at vegetative and reproductive stages and in response to abiotic stress. Plant Physiol Biochem 124:117–125. https://doi.org/10.1016/j.plaphy.2018.01.012
Paoletti C, Bocci F, Lercker G, Capella P, Materassi R (1987) Lipid composition of Azolla caroliniana biomass and its seasonal variation. Phytochemistry 26:1045–1047. https://doi.org/10.1016/S0031-9422(00)82347-X
Nekrasov EV, Tallon SJ, Vyssotski MV, Catchpole OJ (2021) Extraction of lipids from New Zealand fern fronds using near-critical dimethyl ether and dimethyl ether–water–ethanol mixtures. J Supercrit Fluids 170:105137. https://doi.org/10.1016/j.supfu.2020.105137
Pteridophyte Phylogeny Group I (2016) A community-derived classification for extant lycophytes and ferns. J Syst Evol 54:563–603. https://doi.org/10.1111/jse.12229
Rezanka T (1998) Branched and very long-chain dicarboxylic acids from Equisetum species. Phytochemistry 47:1539–1543. https://doi.org/10.1016/S0031-9422(97)00774-7
Akihisa T, Kawashima T, Takahashi S, Sahashi N, Okamoto T, Niiya I, Tamura T (1992) Sterols and fatty acids of a whisk fern Psilotum nudum. J Am Oil Chem Soc 69:1232–1235. https://doi.org/10.1007/BF02637687
Nekrasov EV, Svetashev VI, Khrapko OV, Vyssotski MV (2019) Variability of fatty acid profiles in ferns: relation to fern taxonomy and seasonal development. Phytochemistry 162:47–55. https://doi.org/10.1016/j.phytochem.2019.02.015
Rabert C, Inostroza K, Bravo S, Sepulveda N, Bravo LA (2020) Exploratory study of fatty acid profile in two filmy ferns with contrasting desiccation tolerance reveal the production of very long chain polyunsaturated omega-3 fatty acids. Plants 9:1431. https://doi.org/10.3390/plants9111431
Gemmrich AR (1977) Mobilization of reserve lipids in germinating spores of the fern Anemia phyllitidis L. Plant Sci Let 9:301–307. https://doi.org/10.1016/0304-4211(77)90100-6
Gemmrich AR (1982) Effect of red light and gibberellic acid on lipid metabolism in germinating spores of the fern Anemia phyllitidis. Physiol Plant 54:58–62. https://doi.org/10.1111/j.1399-3054.1982.tb00577.x
Nierop KGJ, Brouwer P, Dekker R, Schluepmann H, Reichart GJ (2018) ω20-Hydroxy and ω9,ω10-dihydroxy biomarker lipids in ferns from the Salviniaceae family. Org Geochem 125:229–242. https://doi.org/10.1016/j.orggeochem.2018.09.014
Shiomi N, Kitoh S (2001) Culture of Azolla in a pond, nutrient composition, and use as fish feed. Soil Sci Plant Nutr 47:27–34. https://doi.org/10.1080/00380768.2001.10408365
Speelman EN, Reichart G-J, de Leeuw JW, Rijpstra WIC, Damste JSS (2009) Biomarker lipids of the freshwater fern Azolla and its fossil counterpart from the Eocene Arctic Ocean. Org Geochem 40:628–637. https://doi.org/10.1016/j.orggeochem.2009.02.001
Rozentsvet OA, Rezanka T, Bosenko ES, Uzhametskaya EA, Dembitskii VM (2005) Fatty acids, phospholipids, and the betaine lipid DGTS from the aquatic fern Salvinia natans. Chem Nat Compd 41:487–490. https://doi.org/10.1007/s10600-005-0189-5
Franich RA, Goodin SJ, Hansen E (1985) Wax esters of the New Zealand silver fern, Cyathea dealbata. Phytochemistry 24:1093–1095. https://doi.org/10.1016/S0031-9422(00)83191-X
Sato N, Furuya M (1984) The composition of lipids and fatty adds determined at various stages of haploid and diploid generations in the fern Adiantum capillus-veneris. Physiol Plant 62:139–147. https://doi.org/10.1111/j.1399-3054.1984.tb00361.x
Etenko OA, Ivankina NF, Anikina EA (2005) Fatty acid composition of bracken fern fronds from the southern and northern zones of Amur Oblast. In: Nekrasov EV (ed) Botanical investigation in Priamurie and borderline territory, proceedings of the region conference, AB BGI FEB RAS, Blagoveshchensk. (In Russian)
Rozentsvet OA, Bogdanova ES, Tabalenkova GN, Golovko TK, Zakhozhiy IG (2011) Ecological and biochemical properties of some typical representatives of cryptogramic plants of Southern Timan. Povolzhskiy J Ecol 10:489–498. (In Russian)
Gellerman JL, Anderson WH, Schlenk H (1972) Highly unsaturated lipids of Mnium, Polytrichum, Marchantia, and Matteuccia. Bryologist 75:550–557
Nekrasov EV, Shelikhan LA, Svetashev VI (2019) Fatty acid composition of gametophytes of Matteuccia struthiopteris (L.) Tod. (Onocleaceae, Polypodiophyta). Botanica Pacifica 8:63–66. https://doi.org/10.17581/bp.2019.08104
John SP, Hasenstein KH (2018) Biochemical responses of the desiccation-tolerant resurrection fern Pleopeltis polypodioides to dehydration and rehydration. J Plant Physiol 228:12–18. https://doi.org/10.1016/j.jplph.2018.05.006
John SP, Hasenstein KH (2020) Desiccation mitigates heat stress in the resurrection fern Pleopeltis polypodioides. Front Plant Sci 11:597731. https://doi.org/10.3389/fpls.2020.597731
WFO (2022) World Flora Online. http://www.worldfloraonline.org. Accessed 16 June 2022
Mongrand S, Bessoule J-J, Cabantous F, Cassagne C (1998) The C16:3/C18:3 fatty acid balance in photosynthetic tissues from 468 plant species. Phytochemistry 49:1049–1064. https://doi.org/10.1016/S0031-9422(98)00243-X
Haufler CH, Pryer KM, Schuettpelz E, Sessa EB, Farrar DR, Moran R, Schneller JJ, Watkins JE, Windham MD (2016) Sex and the single gametophyte: revising the homosporous vascular plant life cycle in light of contemporary research. Bioscience 66:928–937. https://doi.org/10.1093/biosci/biw108
Brownsey PJ, Smith-Dodsworth JC (2000) New Zealand ferns and allied plants. David Bateman, Auckland
Gunstone FD, Harwood JL (2007) Occurrence and characterisation of oils and fats. In: Gunstone FD, Harwood JL, Dijkstra AJ (eds) The lipid handbook with CD-ROM, 3rd edn. CRC Press Taylor & Francis Group, Boca Raton
Auling G, Heinz E, Tulloch AP (1971) Combination and positional distribution of fatty acids in plant galactolipids. Hoppe Seylers Z Physiol Chem 352:905–912
Nichols BW (1965) The lipids of a moss (Hypnum cupressiforme) and of the leaves of green holly (Ilex aquifolium). Phytochemistry 4:769–779. https://doi.org/10.1016/S0031-9422(00)86248-2
Tulloch AP, Heinz E, Fischer W (1973) Combination and positional distribution of fatty acids in plant sulfolipids. Hoppe Seylers Z Physiol Chem 354:879–889
Nekrasov EV, Svetashev VI (2019) Distribution of sciadonic and juniperonic acids in lipid classes in shoots of the horsetail Equisetum arvense during the vegetative season. IX Congress of society physiologists of plants of Russia, Kazan, 18–24 Sept 2019. https://congresskazan2019.ofr.su/viewthesis?id=605. Accessed 18 June 2022 (In Russian)
Boudière L, Michaud M, Petroutsos D, Rébeillé F, Falconet D, Bastien O, Roy S, Finazzi G, Rolland N, Jouhet J, Block MA, Maréchal E (2014) Glycerolipids in photosynthesis: composition, synthesis and trafficking. Biochim Biophys Acta 1837:470–480. https://doi.org/10.1016/j.bbabio.2013.09.007
Hernández ML, Cejudo FJ (2021) Chloroplast lipids metabolism and function. A redox perspective. Front Plant Sci 12. https://doi.org/10.3389/fpls.2021.712022
Ohlrogge JB, Jaworski JG (1997) Regulation of fatty acid synthesis. Ann Rev Plant Physiol Plant Mol Biol 48:109–136. https://doi.org/10.1146/annurev.arplant.48.1.109
Brown AP, Slabas AR, Rafferty JB (2009) Fatty acid biosynthesis in plants – metabolic pathways, structure and organization. In: Wada H, Murata N (eds) Lipids in photosynthesis: essential and regulatory functions. Springer, Dordrecht. https://doi.org/10.1007/978-90-481-2863-1_2
Troncoso-Ponce MA, Nikovics K, Marchive C, Lepiniec L, Baud S (2016) New insights on the organization and regulation of the fatty acid biosynthetic network in the model higher plant Arabidopsis thaliana. Biochimie 120:3–8. https://doi.org/10.1016/j.biochi.2015.05.013
He M, Qin C-X, Wang X, Ding N-Z (2020) Plant unsaturated fatty acids: biosynthesis and regulation. Front Plant Sci 11:390. https://doi.org/10.3389/fpls.2020.00390
Batsale M, Bahammou D, Fouillen L, Mongrand S, Joubès J, Domergue F (2021) Biosynthesis and functions of very-long-chain fatty acids in the responses of plants to abiotic and biotic stresses. Cell 10:1284. https://doi.org/10.3390/cells10061284
Kazaz S, Miray R, Lepiniec L, Baud S (2022) Plant monounsaturated fatty acids: diversity, biosynthesis, functions and uses. Prog Lipid Res 85:101138. https://doi.org/10.1016/j.plipres.2021.101138
Zhukov A, Popov V (2022) Synthesis of C20–38 fatty acids in plant tissues. Int J Mol Sci 23:4731. https://doi.org/10.3390/ijms23094731
Brouwer P, Brautigam A, Kulahoglu C, Tazelaar AOE, Kurz S, Nierop KGJ, van der Werf A, Weber APM, Schluepmann H (2014) Azolla domestication towards a biobased economy? New Phytol 202:1069–1082. https://doi.org/10.1111/nph.12708
Kaewsuwan S, Cahoon EB, Perroud P-F, Wiwat C, Panvisavas N, Quatrano RS, Cove DJ, Bunyapraphatsara N (2006) Identification and functional characterization of the moss Physcomitrella patens Δ5-desaturase gene involved in arachidonic and eicosapentaenoic acids biosynthesis. J Biol Chem 281:21988–21997. https://doi.org/10.1074/jbc.M603022200
Kajikawa M, Matsui K, Ochiai M, Tanaka Y, Kita Y, Ishimoto M, Kohzu Y, Shoji S-i, Yamato KT, Ohyama K, Fukuzawa H, Kohchi T (2008) Production of arachidonic and eicosapentaenoic acids in plants using bryophyte fatty acid Δ6-desaturase, Δ6-elongase, and Δ5-desaturase genes. Biosci Biotechnol Biochem 72:435–444. https://doi.org/10.1271/bbb.70549
Meesapyodsuk D, Qiu X (2012) The front-end desaturase: structure, function, evolution and biotechnological use. Lipids 47:227–237. https://doi.org/10.1007/s11745-011-3617-2
Khozin-Goldberg I, Leu S, Boussiba S (2016) Microalgae as a source for VLC-PUFA production. In: Nakamura Y, Li-Beisson Y (eds) Lipids in plant and algae development, subcellular biochemistry 86. Springer, Cham. https://doi.org/10.1007/978-3-319-25979-6_19
Lee JM, Lee H, Kang SB, Park WJ (2016) Fatty acid desaturases, polyunsaturated fatty acid regulation, and biotechnological advances. Nutrients 8:23. https://doi.org/10.3390/nu8010023
Sayanova O, Haslam R, Caleron MV, Napier JA (2007) Cloning and characterization of unusual fatty acid desaturases from Anemone leveillei: identification of an acyl-coenzyme A C20 Δ5-desaturase responsible for the synthesis of sciadonic acid. Plant Physiol 144:455–467. https://doi.org/10.1104/pp.107.098202
Wu J, Huang J, Hong Y, Zhang H, Ding M, Lou H, Hu Y, Yu W, Song L (2018) De novo transcriptome sequencing of Torreya grandis reveals gene regulation in sciadonic acid biosynthesis pathway. Ind Crop Prod 120:47–60. https://doi.org/10.1016/j.indcrop.2018.04.041
DeLong JM, Prange RK (2008) Fiddlehead fronds: nutrient rich delicacy. Chron Hortic 48:12–15
Gerasimenko N, Korol A, Pikhanova S, Gochachko S (2003) Marketing evaluation of non-wood products of Far Eastern south forests. Practical Marketing 3:17–25. https://www.cfin.ru/press/practical/2003-04/04.shtml Accessed 20 June 2022 (In Russian)
Chamberlain JL, Emery MR, Patel-Weynand T (2018) Assessment of nontimber forest products in the United States under changing conditions. USDA Forest Service, Southern Research Station. https://doi.org/10.2737/SRS-GTR-232
Government of Amur Oblast (2021). https://economy.amurobl.ru/posts/print.php?id=100152. Accessed 08 June 2022 (In Russian)
Haeggstrom JZ, Funk CD (2011) Lipoxygenase and leukotriene pathways: biochemistry, biology, and roles in disease. Chem Rev 111:5866–5898. https://doi.org/10.1021/cr200246d
Serhan CN, Petasis NA (2011) Resolvins and protectins in inflammation resolution. Chem Rev 111:5922–5943. https://doi.org/10.1021/cr100396c
Burdge GC (2018) Polyunsaturated fatty acid biosynthesis and metabolism in adult mammals. In: Burdge GC (ed) Polyunsaturated fatty acid metabolism. AOCS Press. https://doi.org/10.1016/B978-0-12-811230-4.00002-8
Kawashima H (2019) Intake of arachidonic acid-containing lipids in adult humans: dietary surveys and clinical trials. Lipids Health Dis 18:101. https://doi.org/10.1186/s12944-019-1039-y
Deckelbaum RJ, Calder PC (2020) Editorial: is it time to separate EPA from DHA when using omega-3 fatty acids to protect heart and brain? Curr Opin Clin Nutr Met Care 23:65–67. https://doi.org/10.1097/MCO.0000000000000632
Thompson M, Hein N, Hanson C, Smith LM, Anderson-Berry A, Richter CK, Stessy Bisselou K, Kusi Appiah A, Kris-Etherton P, Skulas-Ray AC, Nordgren TM (2019) Omega-3 fatty acid intake by age, gender, and pregnancy status in the United States: national health and nutrition examination survey 2003–2014. Nutrients 11:177. https://doi.org/10.3390/nu11010177
Bushway AA, Serreze DV, McGann DF, True RH, Work TM, Bushway RJ (1985) Effect of processing method and storage time on the nutrient composition of fiddlehead greens. J Food Sci 50:1491–1492. https://doi.org/10.1111/j.1365-2621.1985.tb10508.x
DeLong JM, Hodges DM, Prange RK, Forney CF, Fan L, Bishop MC, Elliot ML, Jordan MA, Doucette C (2013) The influence of cold water storage on fatty acids, antioxidant content and activity, and microbial load in ostrich fern (Matteuccia struthiopteris) fiddleheads. Can J Plant Sci 93:683–697. https://doi.org/10.4141/CJPS2012-165
Shalisko IV, Dmitrichenko MI, Pelenko VV, Shakhov SV (2016) Сhanges in consumer properties of bracken using different storage methods. Proceed Voronezh State Univ Eng Technol 3:151–158. https://doi.org/10.20914/2310-1202-2016-3-151-158. (In Russian)
Znidarcic D, Vidrih R (2009) Content of essential fatty acids in cabbage (Brassica oleracea L.). Acta Agric Slov 93:225–230. https://www.dlib.si/details/URN:NBN:SI:doc-RT6HT80K. Accessed 19 June 2022 (In Slovenian)
Boulom S, Robertson J, Hamid N, Ma Q, Lu J (2014) Seasonal changes in lipid, fatty acid, α-tocopherol and phytosterol contents of seaweed, Undaria pinnatifida, in the Marlborough Sounds, New Zealand. Food Chem 161:261–269. https://doi.org/10.1016/j.foodchem.2014.04.007
Gladyshev MI, Sushchik NN, Gubanenko GA, Demirchieva SM, Kalachova GS (2007) Effect of boiling and frying on the content of essential polyunsaturated fatty acids in muscle tissue of four fish species. Food Chem 101:1694–1700. https://doi.org/10.1016/j.foodchem.2006.04.029
Simopoulos AP, Norman HA, Gillaspy JE, Duke JA (1992) Common purslane: a source of omega-3 fatty acids and antioxidants. J Am Coll Nutr 11:374–382. https://doi.org/10.1080/07315724.1992.10718240
Ghafoorunissa PJ (1993) Vegetables as sources of α-linolenic acid in Indian diets. Food Chem 47:121–124. https://doi.org/10.1016/0308-8146(93)90231-4
Pereira C, Li D, Sinclair AJ (2001) The α-linolenic acid content of green vegetables commonly available in Australia. Int J Vitam Nutr Res 71:223–228. https://doi.org/10.1024/0300-9831.71.4.223
Neff MR, Bhavsar SP, Braekevelt E, Arts MT (2014) Effects of different cooking methods on fatty acid profiles in four fresh water fishes from the Laurentian Great Lakes region. Food Chem 164:544–550. https://doi.org/10.1016/j.foodchem.2014.04.104
Haak L, Sioen I, Raes K, Van Camp J, De Smet S (2007) Effect of pan-frying in different culinary fats on the fatty acid profile of pork. Food Chem 102:857–864. https://doi.org/10.1016/j.foodchem.2006.06.054
Dugan ME, Vahmani P, Turner TD, Mapiye C, Juarez M, Prieto N, Beaulieu AD, Zijlstra RT, Patience JF, Aalhus JL (2015) Pork as a source of omega-3 (n-3) fatty acids. J Clin Med 4:1999–2011. https://doi.org/10.3390/jcm4121956
Droulez V, William PG, Levy G, Stobaus T, Sinclair A (2006) Composition of Australian red meat 2002. 2. Fatty acid profile. Food Australia 58:335–341. https://ro.uow.edu.au/hbspapers/1/. Accessed 19 June 2022
Kouba M, Benatmane F, Blochet JE, Mourot J (2008) Effect of a linseed diet on lipid oxidation, fatty acid composition of muscle, perirenal fat, and raw and cooked rabbit meat. Meat Sci 80:829–834. https://doi.org/10.1016/j.meatsci.2008.03.029
de Souza VJ, Alvarenga TIRC, Andrew NR, McPhee M, Kolakshyapati M, Hopkins DL, Ruhnke I (2021) Technological quality, amino acid and fatty acid profile of broiler meat enhanced by dietary inclusion of black soldier fly larvae. Foods 10:297. https://doi.org/10.3390/foods10020297
Larsen D, Quek SY, Eyres L (2010) Effect of cooking method on the fatty acid profile of New Zealand King Salmon (Oncorhynchus tshawytscha). Food Chem 119:785–790. https://doi.org/10.1016/j.foodchem.2009.07.037
Acknowledgments
This work was financed by the Ministry of Science and Higher Education of the Russian Federation (Project No. 122040800086-1).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 Springer Nature Switzerland AG
About this entry
Cite this entry
Nekrasov, E.V. (2022). Fern Fatty Acids: From Diversity to Dietary Value. In: Murthy, H.N. (eds) Bioactive Compounds in Bryophytes and Pteridophytes. Reference Series in Phytochemistry. Springer, Cham. https://doi.org/10.1007/978-3-030-97415-2_27-1
Download citation
DOI: https://doi.org/10.1007/978-3-030-97415-2_27-1
Received:
Accepted:
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-97415-2
Online ISBN: 978-3-030-97415-2
eBook Packages: Springer Reference Biomedicine and Life SciencesReference Module Biomedical and Life Sciences