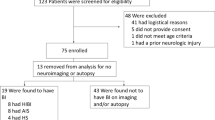
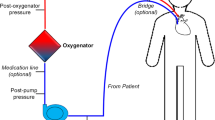
Abstract
Extracorporeal membrane oxygenation (ECMO) is used to provide cardiac and/or pulmonary support in patients refractory to conventional therapies. It is increasingly used in various clinical acute settings and associated with cerebrovascular complications responsible for high morbidity and mortality.
Studies have shown alterations in cerebral blood flow (CBF) during ECMO support are potentially associated with neurological complications. Early detection of cerebral hemodynamics changes could improve patient’s prognosis, and various neuromonitoring tools are available to assess CBF during ECMO support. Transcranial Doppler (TCD) has numerous advantages, as it is a noninvasive procedure, available at bedside, and able to detect cerebral hemodynamic changes in real time and microembolism.
The aim of this chapter is to review literature concerning dynamic monitoring of CBF by TCD during ECMO and to give practical considerations.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Davies A, et al. Extracorporeal membrane oxygenation for 2009 influenza A(H1N1) acute respiratory distress syndrome. JAMA. 2009;302(17):1888–95.
Luyt CE, et al. Long-term outcomes of pandemic 2009 influenza A(H1N1)-associated severe ARDS. Chest. 2012;142(3):583–92.
Combes A, et al. Position paper for the organization of extracorporeal membrane oxygenation programs for acute respiratory failure in adult patients. Am J Respir Crit Care Med. 2014;190(5):488–96.
Abrams D, et al. Position paper for the organization of ECMO programs for cardiac failure in adults. Intensive Care Med. 2018;44(6):717–29.
Marasco SF, et al. Review of ECMO (extra corporeal membrane oxygenation) support in critically ill adult patients. Heart Lung Circ. 2008;17(Suppl 4):S41–7.
Brechot N, et al. Intra-aortic balloon pump protects against hydrostatic pulmonary oedema during peripheral venoarterial-extracorporeal membrane oxygenation. Eur Heart J Acute Cardiovasc Care. 2018;7(1):62–9.
Madershahian N, et al. The acute effect of IABP-induced pulsatility on coronary vascular resistance and graft flow in critical ill patients during ECMO. J Cardiovasc Surg. 2011;52(3):411–8.
Madershahian N, et al. The impact of intraaortic balloon counterpulsation on bypass graft flow in patients with peripheral ECMO. J Card Surg. 2009;24(3):265–8.
Ma P, et al. Combining ECMO with IABP for the treatment of critically ill adult heart failure patients. Heart Lung Circ. 2014;23(4):363–8.
Chung ES, et al. Results of extracorporeal membrane oxygenation (ECMO) support before coronary reperfusion in cardiogenic shock with acute myocardial infarction. Korean J Thorac Cardiovasc Surg. 2011;44(4):273–8.
Combes A, et al. Extracorporeal membrane oxygenation for severe acute respiratory distress syndrome. N Engl J Med. 2018;378(21):1965–75.
Extracorporeal Life Support Organization. Registry report for all ECLS cases.
Mateen FJ, et al. Neurological injury in adults treated with extracorporeal membrane oxygenation. Arch Neurol. 2011;68(12):1543–9.
Mehta A, Ibsen LM. Neurologic complications and neurodevelopmental outcome with extracorporeal life support. World J Crit Care Med. 2013;2(4):40–7.
Risnes I, et al. Cerebral outcome in adult patients treated with extracorporeal membrane oxygenation. Ann Thorac Surg. 2006;81(4):1401–6.
Lewandowski K, et al. High survival rate in 122 ARDS patients managed according to a clinical algorithm including extracorporeal membrane oxygenation. Intensive Care Med. 1997;23(8):819–35.
Luyt CE, et al. Brain injury during venovenous extracorporeal membrane oxygenation. Intensive Care Med. 2016;23:23.
O’Brien NF, Hall MW. Extracorporeal membrane oxygenation and cerebral blood flow velocity in children. Pediatr Crit Care Med. 2013;14(3):e126–34.
Kavi T, et al. Transcranial Doppler changes in patients treated with extracorporeal membrane oxygenation. J Stroke Cerebrovasc Dis. 2016;25(12):2882–5.
Papademetriou MD, et al. Multichannel near infrared spectroscopy indicates regional variations in cerebral autoregulation in infants supported on extracorporeal membrane oxygenation. J Biomed Opt. 2012;17(6):067008.
Lassen NA, Christensen MS. Physiology of cerebral blood flow. Br J Anaesth. 1976;48(8):719–34.
Kuschinsky W, Wahl M. Local chemical and neurogenic regulation of cerebral vascular resistance. Physiol Rev. 1978;58(3):656–89.
Winn HR, et al. Brain adenosine production in rat during sustained alteration in systemic blood pressure. Am J Phys. 1980;239(5):H636–41.
Wei EP, Kontos HA. Increased venous pressure causes myogenic constriction of cerebral arterioles during local hyperoxia. Circ Res. 1984;55(2):249–52.
Hamel E. Perivascular nerves and the regulation of cerebrovascular tone. J Appl Physiol (1985). 2006;100(3):1059–64.
Waeber C, Moskowitz MA. Migraine as an inflammatory disorder. Neurology. 2005;64(10 Suppl 2):S9–15.
McHenry LC, et al. Cerebral autoregulation in man. Stroke. 1974;5(6):695–706.
Faraci FM, Mayhan WG, Heistad DD. Segmental vascular responses to acute hypertension in cerebrum and brain stem. Am J Phys. 1987;252(4 Pt 2):H738–42.
Bouma GJ, et al. Blood pressure and intracranial pressure-volume dynamics in severe head injury: relationship with cerebral blood flow. J Neurosurg. 1992;77(1):15–9.
Czosnyka M, et al. Cerebral autoregulation following head injury. J Neurosurg. 2001;95(5):756–63.
Powers WJ, et al. Autoregulation after ischaemic stroke. J Hypertens. 2009;27(11):2218–22.
Panerai RB, et al. Dynamic cerebral autoregulation following acute ischaemic stroke: comparison of transcranial Doppler and magnetic resonance imaging techniques. J Cereb Blood Flow Metab. 2016;36(12):2194–202.
Oeinck M, et al. Dynamic cerebral autoregulation in acute intracerebral hemorrhage. Stroke. 2013;44(10):2722–8.
Ma H, et al. Temporal course of dynamic cerebral autoregulation in patients with intracerebral hemorrhage. Stroke. 2016;47(3):674–81.
Budohoski KP, et al. Impairment of cerebral autoregulation predicts delayed cerebral ischemia after subarachnoid hemorrhage: a prospective observational study. Stroke. 2012;43(12):3230–7.
Otite F, et al. Impaired cerebral autoregulation is associated with vasospasm and delayed cerebral ischemia in subarachnoid hemorrhage. Stroke. 2014;45(3):677–82.
Short BL. The effect of extracorporeal life support on the brain: a focus on ECMO. Semin Perinatol. 2005;29(1):45–50.
Liem KD, et al. Cerebral oxygenation and hemodynamics during induction of extracorporeal membrane oxygenation as investigated by near infrared spectrophotometry. Pediatrics. 1995;95(4):555–61.
Fenik JC, Rais-Bahrami K. Neonatal cerebral oximetry monitoring during ECMO cannulation. J Perinatol. 2009;29(5):376–81.
Tranmer BI, et al. Pulsatile versus nonpulsatile blood flow in the treatment of acute cerebral ischemia. Neurosurgery. 1986;19(5):724–31.
Joshi B, et al. Predicting the limits of cerebral autoregulation during cardiopulmonary bypass. Anesth Analg. 2012;114(3):503–10.
Hori D, et al. Hypotension after cardiac operations based on autoregulation monitoring leads to brain cellular injury. Ann Thorac Surg. 2015;100(2):487–93.
Short BL, et al. Impairment of cerebral autoregulation during extracorporeal membrane oxygenation in newborn lambs. Pediatr Res. 1993;33(3):289–94.
Tian F, et al. Impairment of cerebral autoregulation in pediatric extracorporeal membrane oxygenation associated with neuroimaging abnormalities. Neurophotonics. 2017;4(4):041410.
Tian F, et al. Wavelet coherence analysis of dynamic cerebral autoregulation in neonatal hypoxic-ischemic encephalopathy. Neuroimage Clin. 2016;11:124–32.
Cakici M, et al. Controlled flow diversion in hybrid venoarterial-venous extracorporeal membrane oxygenation. Interact Cardiovasc Thorac Surg. 2018;26(1):112–8.
Doll N, et al. Five-year results of 219 consecutive patients treated with extracorporeal membrane oxygenation for refractory postoperative cardiogenic shock. Ann Thorac Surg. 2004;77(1):151–7. discussion 157
Yang F, et al. Effects of intra-aortic balloon pump on cerebral blood flow during peripheral venoarterial extracorporeal membrane oxygenation support. J Transl Med. 2014;12:106.
Meng L, Gelb AW. Regulation of cerebral autoregulation by carbon dioxide. Anesthesiology. 2015;122(1):196–205.
Weber TR, Kountzman B. The effects of venous occlusion on cerebral blood flow characteristics during ECMO. J Pediatr Surg. 1996;31(8):1124–7.
Fukuda S, et al. Comparison of venoarterial versus venovenous access in the cerebral circulation of newborns undergoing extracorporeal membrane oxygenation. Pediatr Surg Int. 1999;15(2):78–84.
Taylor GA, et al. Intracranial flow patterns in infants undergoing extracorporeal membrane oxygenation: preliminary observations with Doppler US. Radiology. 1987;165(3):671–4.
van de Bor M, et al. Extracorporeal membrane oxygenation and cerebral blood flow velocity in newborn infants. Crit Care Med. 1990;18(1):10–3.
Schachtrupp A, et al. Influence of intra-aortic balloon pumping on cerebral blood flow pattern in patients after cardiac surgery. Eur J Anaesthesiol. 2005;22(3):165–70.
Gee W, et al. Assessment of intra-aortic balloon pumping by ocular pneumoplethysmography. Am Surg. 1986;52(9):489–91.
Marinoni M, et al. Retrospective analysis of transcranial Doppler patterns in veno-arterial extracorporeal membrane oxygenation patients: feasibility of cerebral circulatory arrest diagnosis. ASAIO J. 2018;64(2):175–82.
Nasr DM, Rabinstein AA. Neurologic complications of extracorporeal membrane oxygenation. J Clin Neurol. 2015;11(4):383–9.
Omar HR, et al. Incidence and predictors of ischemic cerebrovascular stroke among patients on extracorporeal membrane oxygenation support. J Crit Care. 2016;32:48–51.
Meyer AD, et al. Platelet-derived microparticles generated by neonatal extracorporeal membrane oxygenation systems. ASAIO J. 2015;61(1):37–42.
Foster PP, et al. Patent foramen ovale and paradoxical systemic embolism: a bibliographic review. Aviat Space Environ Med. 2003;74(6 Pt 2):B1–64.
Stocchetti N, et al. Hyperventilation in head injury: a review. Chest. 2005;127(5):1812–27.
Brunser AM, et al. The role of TCD in the evaluation of acute stroke. J Neuroimaging. 2016;26(4):420–5.
Muehrcke DD, et al. Complications of extracorporeal life support systems using heparin-bound surfaces. The risk of intracardiac clot formation. J Thorac Cardiovasc Surg. 1995;110(3):843–51.
Jiang J, et al. Microembolic signal monitoring of TOASTclassified cerebral infarction patients. Mol Med Rep. 2013;8(4):1135–42.
Best LM, et al. Transcranial Doppler ultrasound detection of microemboli as a predictor of cerebral events in patients with symptomatic and asymptomatic carotid disease: a systematic review and meta-analysis. Eur J Vasc Endovasc Surg. 2016;52(5):565–80.
Zanatta P, et al. Microembolic signals and strategy to prevent gas embolism during extracorporeal membrane oxygenation. J Cardiothorac Surg. 2010;5:5.
Marinoni M, et al. Cerebral microemboli detected by transcranial doppler in patients treated with extracorporeal membrane oxygenation. Acta Anaesthesiol Scand. 2016;60(7):934–44.
Hornig CR, Dorndorf W, Agnoli AL. Hemorrhagic cerebral infarction–a prospective study. Stroke. 1986;17(2):179–85.
Kasirajan V, et al. Risk factors for intracranial hemorrhage in adults on extracorporeal membrane oxygenation. Eur J Cardiothorac Surg. 1999;15(4):508–14.
Martucci G, Lo Re V, Arcadipane A. Neurological injuries and extracorporeal membrane oxygenation: the challenge of the new ECMO era. Neurol Sci. 2016;19:19.
Hingorani A, et al. Causes of early post carotid endartectomy stroke in a recent series: the increasing importance of hyperperfusion syndrome. Acta Chir Belg. 2002;102(6):435–8.
Giani M, et al. Apnea test during brain death assessment in mechanically ventilated and ECMO patients. Intensive Care Med. 2016;42(1):72–81.
Sloan MA, et al. Assessment: transcranial Doppler ultrasonography: report of the Therapeutics and Technology Assessment Subcommittee of the American Academy of Neurology. Neurology. 2004;62(9):1468–81.
Wahlster S, et al. Brain death declaration: practices and perceptions worldwide. Neurology. 2015;84(18):1870–9.
Pugin D, Woimant F. Stroke care in the ICU: general supportive treatment. Experts’ recommendations. Rev Neurol (Paris). 2012;168(6–7):490–500.
Atkinson JL, Anderson RE, Sundt TM. The effect of carbon dioxide on the diameter of brain capillaries. Brain Res. 1990;517(1–2):333–40.
Kazmi SO, et al. Cerebral pathophysiology in extracorporeal membrane oxygenation: pitfalls in daily clinical management. Crit Care Res Pract. 2018;2018:3237810.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Algorithm
Algorithm

ABCD airway-breathing-circulation-disability, GCS Glasgow coma score, LVEF left ventricular ejection fraction, CBF Cerebral blood flow, MV mechanical ventilation, IABP Intra-aortic blood pump, PI pulsatility index, EDV end-diastolic velocity, FA femoral artery, SCA subclavian artery, AAo aortic, CBV cerebral blood volume, FO foramen ovale, AKI acute renal injury, CPP cerebral perfusion pressure
Rights and permissions
Copyright information
© 2022 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Le Guennec, L., Combes, A. (2022). ECMO Patient in Intensive Care Unit: Usefulness of Neurosonology in Neurologic Monitoring. In: Rodríguez, C.N., et al. Neurosonology in Critical Care . Springer, Cham. https://doi.org/10.1007/978-3-030-81419-9_48
Download citation
DOI: https://doi.org/10.1007/978-3-030-81419-9_48
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-81418-2
Online ISBN: 978-3-030-81419-9
eBook Packages: MedicineMedicine (R0)