Abstract
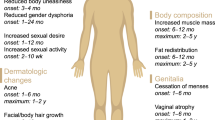
Gender affirming hormonal treatment (GAHT) in transgender men consists of testosterone treatment in different formulations. The main goal of testosterone treatment is to achieve cisgender male serum testosterone levels in order to induce virilization. The desired effects include increased facial and body hair, deepening of the voice, cessation of menses, fat redistribution and increased lean mass and strength, as well as improvement of psychological well-being. However, testosterone treatment may induce potential undesired effects and risks, such as acne, androgenetic alopecia, increase in systolic blood pressure, haematocrit and changes in lipid profile. GAHT in transgender men is considered safe on the short term and middle term, although several aspects, such as long-term cardiovascular and oncological safety, need to be adequately assessed in the future through long-term prospective studies.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
World Health Organisation. International classification of diseases 11th revision. Geneva: World Health Organisation; 2018. Available at: https://icd.who.int/. Accessed 27 Aug 2018.
American Psychiatric Association. Diagnostic and statistical manual of mental disorders (DSM-5). Arlington: American Psychiatric Publishers; 2013.
Arcelus J, De Vries ALC, Fisher AD, Nieder TO, Özer M, Motmans J. European Society for Sexual Medicine Position Statement “Assessment and hormonal management in adolescent and adult trans people, with attention for sexual function and satisfaction”. J Sex Med. 2020;pii:S1743-6095(20)30045-X. https://doi.org/10.1016/j.jsxm.2020.01.012. [Epub ahead of print].
Coleman E, Bockting W, Botzer M, et al. Standards of care for the health of transsexual, transgender, and gender-nonconforming people, version 7. Int J Transgenderism. 2012;13:165–232.
Heylens G, Verroken C, De Cock S, et al. Effects of different steps in gender reassignment therapy on psychopathology: a prospective study of persons with a gender identity disorder. J Sex Med. 2014;11(1):119–26.
Fisher AD, Castellini G, Bandini E, et al. Cross-sex hormonal treatment and body uneasiness in individuals with gender dysphoria. J Sex Med. 2014;11(3):709–19.
Fisher AD, Castellini G, Ristori J, et al. Cross-sex hormone treatment and psychobiological changes in transsexual persons: two-year follow-up data. J Clin Endocrinol Metab. 2016;101(11):4260–9.
Gorin-Lazard A, Baumstarck K, Boyer L, et al. Is hormonal therapy associated with better quality of life in transsexuals? A cross-sectional study. J Sex Med. 2012;9(2):531–41.
Hembree WC, Cohen-Kettenis PT, Gooren L, et al. Endocrine treatment of gender-dysphoric/gender-incongruent persons: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2017;102(11):3869–903.
Cocchetti C, Ristori J, Romani A, et al. Hormonal treatment strategies tailored to non-binary transgender individuals. J Clin Med. 2020;9(6):1609.
Defreyne J, Vantomme B, Van Caenegem E, et al. Prospective evaluation of hematocrit in gender-affirming hormone treatment: results from European Network for the Investigation of Gender Incongruence. Andrology. 2018;6:446–54.
Pelusi C, Costantino A, Martelli V, et al. Effects of three different testosterone formulations in female-to-male transsexual persons. J Sex Med. 2014;11:3002–11.
Evans S, Neave N, Wakelin D, et al. The relationship between testosterone and vocal frequencies in human males. Physiol Behav. 2008;93(4–5):783–8.
Bultynck C, Pas C, Defreyne J, et al. Self-perception of voice in transgender persons during cross-sex hormone therapy. Laryngoscope. 2017;127(12):2796–804.
Abitbol J, Abitbol P, Abitbol B. Sex hormones and the female voice. J Voice. 1999;13(3):424–46.
Cosyns M, Borsel J, Wierckx K, et al. Voice in female-to-male transsexual persons after long-term androgen therapy. Laryngoscope. 2014;124(6):1409–14.
Watt SO, Tskhay KO, Rule NO. Masculine voices predict well-being in female-tomale transgender individuals. Arch Sex Behav. 2017;47:963–72.
Giltay EJ, Gooren LJG. Effects of sex steroid deprivation/administration on hair growth and skin sebum production in transsexual males and females. J Clin Endocrinol Metab. 2000;85(8):2913–21.
Wierckx K, Van de Peer F, Verhaeghe E, et al. Short-and long-term clinical skin effects of testosterone treatment in trans men. J Sex Med. 2014;11(1):222–9.
Mueller A, Kiesewetter F, Binder H, et al. Long-term administration of testosterone undecanoate every 3 months for testosterone supplementation in female-to-male transsexuals. J Clin Endocrinol Metab. 2007;92(9):3470–5.
Nakamura A, Watanabe M, Sugimoto M, Sako T, Mahmood S, Kaku H, Nasu Y, Ishii K, Nagai A, Kumon H. Dose-response analysis of testosterone replacement therapy in patients with female to male gender identity disorder. Endocr J. 2013;60(3):275–81. Epub 2012 Oct 27.
Wierckx K, Van Caenegem E, Schreiner T, et al. Cross-sex hormone therapy in trans persons is safe and effective at short-time follow-up: results from the European network for the investigation of gender incongruence. J Sex Med. 2014;11:1999–2011.
Allan CA, Forbes EA, Strauss BJG, et al. Testosterone therapy increases sexual desire in ageing men with low-normal testosterone levels and symptoms of androgen deficiency. Int J Impot Res. 2008;20(4):396–401.
Costantino A, Cerpolini S, Alvisi S, et al. A prospective study on sexual function and mood in female-to-male transsexuals during testosterone administration and after sex reassignment surgery. J Sex Marital Ther. 2013;39(4):321–35.
Somboonporn W, Davis S, Seif MW, et al. Testosterone for peri- and postmenopausal women. Cochrane Database Syst Rev. 2005;(4):CD004509.
Wierckx K, Elaut E, Van Caenegem E, et al. Sexual desire in female-to-male transsexual persons: exploration of the role of testosterone administration. Eur J Endocrinol. 2011;165(2):331–7.
Ristori J, Cocchetti C, Castellini G, Pierdominici M, Cipriani A, Testi D, Gavazzi G, Mazzoli F, Mosconi M, Meriggiola MC, Cassioli E, Vignozzi L, Ricca V, Maggi M, Fisher AD. Hormonal treatment effect on sexual distress in transgender persons: 2-year follow-up data. J Sex Med. 2020;17(1):142–51.
Berra M, Armillotta F, D’Emidio L, et al. Testosterone decreases adiponectin levels in female to male transsexuals. Asian J Androl. 2006;8:725–9.
Colizzi M, Costa R, Scaramuzzi F, et al. Concomitant psychiatric problems and hormonal treatment induced metabolic syndrome in gender dysphoria individuals: a 2 year follow-up study. J Psychosom Res. 2015;78:399–406.
Mueller A, Haeberle L, Zollver H, et al. Effects of intramuscular testosterone undecanoate on body composition and bone mineral density in female-to-male transsexuals. J Sex Med. 2010;7:3190–8.
Quiros C, Patrascioiu I, Mora M, et al. Effect of cross-sex hormone treatment on cardiovascular risk factors in transsexual individuals. Experience in a specialized unit in Catalonia. Endocrinol Nutr. 2015;62:210–6.
Van Caenegem E, Wierckx K, Taes Y, et al. Body composition, bone turnover, and bone mass in trans men during testosterone treatment: 1-year follow-up data from a prospective case-controlled study (ENIGI). Eur J Endocrinol. 2015;172:163–71.
Deplewski D, Rosenfiled R. Role of hormones in pilosebaceous unit development. Endocr Rev. 2000;21:363–92.
Ebling FJ, Skinner J. The measurements of sebum production in rats treated with T and oestradiol. Br J Dermatol. 1967;79:386–93.
Baldassarre M, Giannone FA, Foschini MP, et al. Effects of long-term high dose testosterone administration on vaginal epithelium structure and estrogen receptor-α and -β expression of young women. Int J Impot Res. 2013;25:172–7.
Schlatterer K, Von Werder K, Stalla GK. Multistep treatment concept of transsexual patients. Exp Clin Endocrinol Diabetes. 1996;104(06):413–9.
Grynberg M, Fanchin R, Dubost G, et al. Histology of genital tract and breast tissue after long-term testosterone administration in a female-to-male transsexual population. Reprod Biomed Online. 2010;20:553–8.
Perrone AM, Cerpolini S, Maria Salfi NC, et al. Effect of long-term testosterone administration on the endometrium of female-to-male (FtM) transsexuals. J Sex Med. 2009;6:3193–200.
Finkle WD, Greenland S, Ridgeway GK, et al. Increased risk of non-fatal myocardial infarction following testosterone therapy prescription in men. PLoS One. 2014;9:e85805.
Elamin MB, Garcia MZ, Murad MH, Erwin PJ, Montori VM. Effect of sex steroid use on cardiovascular risk in transsexual individuals: a systematic review and meta-analyses. Clin Endocrinol (Oxf). 2010;72:1–10.
Olson-Kennedy J, Okonta V, Clark LF, et al. Physiologic response to gender affirming hormones among transgender youth. J Adolesc Health. 2017;62(4):397–401.
Fernandez JD, Tannock LR. Metabolic effects of hormone therapy in transgender patients. Endocr Pract. 2015;22(4):383–8.
Vita R, Settineri S, Liotta M, et al. Changes in hormonal and metabolic parameters in transgender subjects on cross-sex hormone therapy: a cohort study. Maturitas. 2018;107:92–6.
Cupisti S, Giltay EJ, Gooren LJ, et al. The impact of testosterone administration to female-to-male transsexuals on insulin resistance and lipid parameters compared with women with polycystic ovary syndrome. Fertil Steril. 2010;94(7):2647–53.
Chandra P, Basra SS, Chen TC, et al. Alterations in lipids and adipocyte hormones in female-to-male transsexuals. Int J Endocrinol. 2010;2010 [pii:945053].
Deutsch MB, Bhakri V, Kubicek K. Effects of cross-sex hormone treatment on transgender women and men. Obstet Gynecol. 2015;125(3):605–10.
Wultsch A, Kaufmann U, Ott J, et al. Profound changes in sex hormone levels during cross-sex hormone therapy of transsexuals do not alter serum cholesterol acceptor capacity. J Sex Med. 2015;12(6):1436–9.
Elbers JMH, Giltay EJ, Teerlink T, et al. Effects of sex steroids on components of the insulin resistance syndrome in transsexual subjects. Clin Endocrinol (Oxf). 2003;58(5):562–71.
Jacobeit JW, Gooren LJ, Schulte HM. Endocrinology: long-acting intramuscular testosterone undecanoate for treatment of female-to-male transgender individuals. J Sex Med. 2007;4(5):1479–84.
Jacobeit JW, Gooren LJ, Schulte HM. Safety aspects of 36 months of administration of long-acting intramuscular testosterone undecanoate for treatment of female-to-male transgender individuals. Eur J Endocrinol. 2009;161(5):795–8.
Maraka S, Singh Ospina N, Rodriguez-Gutierrez R, et al. Sex steroids and cardiovascular outcomes in transgender individuals: a systematic review and metaanalysis. J Clin Endocrinol Metab. 2017;102(11):3914–23.
Giltay EJ, Toorians AWFT, Sarabjitsingh AR, et al. Established risk factors for coronary heart disease are unrelated to androgen-induced baldness in female-tomale transsexuals. J Endocrinol. 2004;180(1):107–12.
Polderman KH, Gooren LJ, Asscheman H, et al. Induction of insulin resistance by androgens and estrogens. J Clin Endocrinol Metab. 1994;79(1):265–71.
Auer MK, Ebert T, Pietzner M, Defreyne J, Fuss J, Stalla GK, T’Sjoen G. Effects of sex hormone treatment on the metabolic syndrome in transgender individuals: focus on metabolic cytokines. J Clin Endocrinol Metab. 2018;103(2):790–802.
Toorians AW, Thomassen MC, Zweegman S, et al. Venous thrombosis and changes of hemostatic variables during cross-sex hormone treatment in transsexual people. J Clin Endocrinol Metab. 2003;88:5723–9.
Wierckx K, Mueller S, Weyers S, et al. Long-term evaluation of cross-sex hormone treatment in transsexual persons. J Sex Med. 2012;9:2641–51.
Ott J, Kaufmann U, Bentz EK, Huber JC, Tempfer CB. Incidence of thrombophilia and venous thrombosis in transsexuals under cross-sex hormone therapy. Fertil Steril. 2010;93:1267–72.
Wierckx K, Elaut E, Declercq E, et al. Prevalence of cardiovascular disease and cancer during cross-sex hormone therapy in a large cohort of trans persons: a case-control study. Eur J Endocrinol. 2013;169:471–8.
Bird D, Vowles K, Anthony PP. Spontaneous rupture of a liver cell adenoma after long term methyltestosterone: report of a case successfully treated by emergency right hepatic lobectomy. Br J Surg. 1979;66(3):212–3.
Westaby D, Paradinas FJ, Ogle SJ, et al. Liver damage from long-term methyltestosterone. Lancet. 1977;310(8032):261–3.
Sorelle JA, Jiao R, Gao E, Veazey J, Frame I, Quinn AM, Day P, Pagels P, Gimpel N, Patel K. Impact of hormone therapy on laboratory values in transgender patients. Clin Chem. 2019;65(1):170–9.
Singh-Ospina N, Maraka S, Rodriguez-Gutierrez R, et al. Effect of sex steroids on the bone health of transgender individuals: a systematic review and metaanalysis. J Clin Endocrinol Metab. 2017;102(11):3904–13.
Van Caenegem E, Wierckx K, Taes Y, et al. Bone mass, bone geometry, and body composition in female-to-male transsexual persons after long-term cross-sex hormonal therapy. J Clin Endocrinol Metab. 2012;97(7):2503–11.
Miyajima T, Kim YT, Oda H. A study of changes in bone metabolism in cases of gender identity disorder. J Bone Miner Metab. 2012;30(4):468–73.
Goh HHV, Ratnam SS. Effects of hormone deficiency, androgen therapy and calcium supplementation on bone mineral density in female transsexuals. Maturitas. 1997;26(1):45–52.
Van Kesteren P, Lips P, Gooren LJG, et al. Long-term follow-up of bone mineral density and bone metabolism in transsexuals treated with cross-sex hormones. Clin Endocrinol (Oxf). 1998;48(3):347–54.
Slagter MH, Gooren LJ, Scorilas A, Petraki CD, Diamandis EP. Effects of long-term androgen administration on breast tissue of female-to-male transsexuals. J Histochem Cytochem. 2006;54:905–10.
Stone JP, Hartley RL, Temple-Oberle C. Breast cancer in transgender patients: a systematic review. Part 2: female to male. Eur J Surg Oncol. 2018;44:1463–8.
Nikolic DV, Djordjevic ML, Granic M, et al. Importance of revealing a rare case of breast cancer in a female to male transsexual after bilateral mastectomy. World J Surg Oncol. 2012;10(1):280.
Burcombe RJ, Makris A, Pittam M, et al. Breast cancer after bilateral subcutaneous mastectomy in a female-to-male trans-sexual. Breast. 2003;12(4):290–3.
Shao T, Grossbard ML, Klein P. Breast cancer in female-to-male transsexuals: two cases with a review of physiology and management. Clin Breast Cancer. 2011;11(6):417–9.
Gooren L, Bowers M, Lips P, et al. Five new cases of breast cancer in transsexual persons. Andrologia. 2015;47(10):1202–5.
Van Renterghem SMJ, Van Dorpe J, Monstrey SJ, et al. Routine histopathological examination after female-to-male gender-confirming mastectomy. Br J Surg. 2018;105(7):885–92.
Urban RR, Teng NNH, Kapp DS. Gynecologic malignancies in female-to-male transgender patients: the need of original gender surveillance. Am J Obstet Gynecol. 2011;204(5):e9–12.
Dria’k D, Samudovsky M. Could a man be affected with carcinoma of cervix?- The first case of cervical carcinoma in trans-sexual person (FtM)-case report. Acta Medica (Hradec Kralove). 2005;48(1):53.
Taylor ET, Bryson MK. Cancer’s margins: trans* and gender nonconforming people’s access to knowledge, experiences of cancer health, and decision-making. LGBT Health. 2016;3(1):79–89.
Brown B, Poteat T, Marg L, Galea JT. Human papillomavirus-related cancer surveillance, prevention, and screening among transgender men and women: neglected populations at high risk. LGBT Health. 2017;4(5):315–9.
Futterweit W. Endocrine therapy of transsexualism and potential complications of long-term treatment. Arch Sex Behav. 1998;27(2):209–26.
Dizon DS, Tejada-Berges T, Koelliker S, et al. Ovarian cancer associated with testosterone supplementation in a female-to-male transsexual patient. Gynecol Obstet Invest. 2006;62(4):226–8.
Hage JJ, Dekker J, Karim RB, et al. Ovarian cancer in female-to-male transsexuals: report of two cases. Gynecol Oncol. 2000;76(3):413–5.
Clements-Nolle K, Marx R, Katz M. Attempted suicide among transgender persons: the influence of gender-based discrimination and victimization. J Homosex. 2006;51(3):53–69.
Bockting WO, Miner MH, Swinburne Romine RE, et al. Stigma, mental health, and resilience in an online sample of the US transgender population. Am J Public Health. 2013;103(5):943–51.
Budge SL, Adelson JL, Howard KAS. Anxiety and depression in transgender individuals: the roles of transition status, loss, social support, and coping. J Consult Clin Psychol. 2013;81(3):545.
Witcomb GL, Bouman WP, Claes L, et al. Levels of depression in transgender people and its predictors: results of a large matched control study with transgender people accessing clinical services. J Affect Disord. 2018;235:308–15.
Defreyne J, Kreukels B, T’Sjoen G, Staphorsius A, Den Heijer M, Heylens G, Elaut E. No correlation between serum testosterone levels and state-level anger intensity in transgender people: results from the European Network for the Investigation of Gender Incongruence. Horm Behav. 2019;110:29–39.
Asscheman H, Giltay EJ, Megens JA, de Ronde WP, van Trotsenburg MA, Gooren LJ. A long-term follow-up study of mortality in transsexuals receiving treatment with cross-sex hormones. Eur J Endocrinol. 2011;164:635–42.
Dhejne C, Lichtenstein P, Boman M, Johansson AL, Langstrom N, Landen M. Long-term follow-up of transsexual persons undergoing sex reassignment surgery: cohort study in Sweden. PLoS One. 2011;6:e16885.
Murad MH, Elamin MB, Garcia MZ, et al. Hormonal therapy and sex reassignment: a systematic review and meta-analysis of quality of life and psychosocial outcomes. Clin Endocrinol (Oxf). 2010;72(2):214–31.
Defreyne J, Motmans J, T’Sjoen G. Healthcare costs and quality of life outcomes following gender affirming surgery in trans men: a review. Expert Rev Pharmacoecon Outcomes Res. 2017;17(6):543–56.
Kuhn A, Bodmer C, Stadlmayr W, et al. Quality of life 15 years after sex reassignment surgery for transsexualism. Fertil Steril. 2009;92(5):1685–1689.e3.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Cocchetti, C., Fisher, A.D. (2021). Testosterone in Transgender Population. In: Mulhall, J.P., Maggi, M., Trost, L. (eds) Controversies in Testosterone Deficiency . Springer, Cham. https://doi.org/10.1007/978-3-030-77111-9_9
Download citation
DOI: https://doi.org/10.1007/978-3-030-77111-9_9
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-77110-2
Online ISBN: 978-3-030-77111-9
eBook Packages: MedicineMedicine (R0)