Abstract
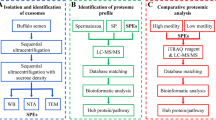
Various studies have sought to determine the optimal abstinence period after which semen samples should be collected, with many contradictory results reported. Several factors influence the semen microenvironment, and thus potentially sperm parameters. In this study, we focused on the secretions of the prostate, seminal vesicles and the epididymis. Semen samples were obtained from healthy normozoospermic males (n = 16) after 4 day and 4 h periods of ejaculatory abstinence (EA), and standard semen analysis was performed using computer-aided sperm analysis, whereas seminal plasma citric acid, neutral alpha-glucosidase and fructose concentrations were measured using assay kits. Proteomic analyses of seminal plasma proteins were performed using LC-MS/MS. There were significant decreases in total sperm count (P < 0.001), sperm concentration (P < 0.05) and semen volume (P < 0.05) after 4 h compared with 4 days ejaculatory abstinence. Furthermore, increases were observed in total sperm motility (P < 0.05), sperm progressive motility (P < 0.01), average path velocity (P < 0.001) and curvilinear velocity (P < 0.05) after a 4 h abstinence period, accompanied by significant reductions in citric acid (P < 0.05), neutral alpha-glucosidase (P < 0.01) and fructose (P < 0.01) concentrations. In addition, due to the decreased number of spermatozoa, these concentrations translated to a significant decrease in fructose (P < 0.05) per spermatozoon. A total of 2889 proteins were identified, with 24 proteins differentially expressed. These proteins have various roles in cellular processes and may influence the improvement of sperm motility after 4 h of EA. All these factors present in the seminal plasma indicate towards an intrinsic mechanism capitalising on alternative sources of energy for increased metabolic function and subsequent sperm motility.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Aebersold R, Mann M (2003) Mass spectrometry-based proteomics. Nature 422(6928):198
Agarwal A, Gupta S, Du Plessis S, Sharma R, Esteves SC, Cirenza C, Eliwa J, Al-Najjar W, Kumaresan D, Haroun N, Philby S, Sabanegh E (2016) Abstinence time and its impact on basic and advanced semen parameters. Urology 94:102–110
Alipour H, Dardmeh F, Van Der Horst G, Manoharan G, Askeland A, Nielsena H (2015) Effect of short abstinence time on sperm motility parameters. Paper presented at the Final Programme, 18th World Congress of in Vitro Ferilization, Isivf
Alipour H, Van Der Horst G, Christiansen O, Dardmeh F, Jørgensen N, Nielsen H, Hnida C (2017) Improved sperm kinematics in semen samples collected after 2 h versus 4–7 days of ejaculation abstinence. Hum Reprod 32(7):1364–1372
Ashworth PJ, Harrison RA, Miller NG, Plummer JM, Watson PF (1994) Survival of ram spermatozoa at high dilution: protective effect of simple constituents of culture media as compared with seminal plasma. Reprod Fertil Dev 6(2):173–180
Association, G. A. o. t. W. M (2014) World medical association declaration of Helsinki: ethical principles for medical research involving human subjects. J Am College Dentist 81(3):14
Ayad BM, Van der Horst G, Du Plessis S (2018a) Revisiting the relationship between the ejaculatory abstinence period and semen characteristics. Int J Fertil Steril 11(4):238
Ayad BM, Van der Horst G, du Plessis S (2018b) Short abstinence: a potential strategy for the improvement of sperm quality. Middle East Fertil Soc J 23(1):37–43
Baas JW, Molan P, Shannon P (1983) Factors in seminal plasma of bulls that affect the viability and motility of spermatozoa. Reproduction 68(2):275–280
Baccetti B (1984) The human spermatozoon. In: Ultrastructure of reproduction. Springer, Berlin, pp 110–126
Bahadur G, Almossawi O, Zaid R, Ilahibuccus A, Al-Habib A, Muneer A, Okolo S (2016) Semen characteristics in consecutive ejaculates with short abstinence in subfertile males. Reprod BioMed Online 32(3):323–328
Balk SP, Ko Y-J, Bubley GJ (2003) Biology of prostate-specific antigen. J Clin Oncol 21(2):383–391
Batruch I, Lecker I, Kagedan D, Smith CR, Mullen BJ, Grober E, Lo KC, Diamandis EP, Jarvi KA (2011) Proteomic analysis of seminal plasma from normal volunteers and post-vasectomy patients identifies over 2000 proteins and candidate biomarkers of the urogenital system. J Proteome Res 10(3):941–953
Bernardini A, Hozbor F, Sanchez E, Fornés M, Alberio R, Cesari A (2011) Conserved ram seminal plasma proteins bind to the sperm membrane and repair cryopreservation damage. Theriogenology 76(3):436–447
Binz P-A, Hochstrasser DF, Appel RD (2003) Mass spectrometry-based proteomics: current status and potential use in clinical chemistry. Clin Chem Lab Med 41(12):1540–1551
Bone W, Jones AR, Morin C, Nieschlag E, Cooper TG (2001) Susceptibility of glycolytic enzyme activity and motility of spermatozoa from rat, mouse, and human to inhibition by proven and putative chlorinated antifertility compounds in vitro. J Androl 22(3):464–470
Borges E Jr, Braga DPDAF, Setti AS (2018) Shorter ejaculatory abstinence interval and maternal endometrium exposure to seminal plasma as tools to improve pregnancy rate in patients undergoing intracytoplasmic sperm injection cycles. JBRA Assist Reprod 22(3):160
Caballero I, Parrilla I, Almiñana C, Del Olmo D, Roca J, Martínez E, Vázquez J (2012) Seminal plasma proteins as modulators of the sperm function and their application in sperm biotechnologies. Reprod Domest Anim 47:12–21
Canale D, Bartelloni M, Negroni A, Meschini P, Izzo P, Bianchi B, Menchini‐Fabris G (1986) Zinc in human semen. Int J Androl 9(6):477–480
Cao W-L, Wang Y-X, Xiang Z-Q, Li Z (2003) Cryopreservation-induced decrease in heat-shock protein 90 in human spermatozoa and its mechanism. Asian J Androl 5(1):43–46
Comar VA, Petersen CG, Mauri AL, Mattila M, Vagnini LD, Renzi A, Petersen B, Nicoletti A, Dieamant F, Oliveira JBAJJAR (2017) Influence of the abstinence period on human sperm quality: analysis of 2,458 semen samples. JBRA Assist Reprod 21(4):306
Cooper T, Yeung C, Nashan D, Jockenhövel F, Nieschlag E (1990) Improvement in the assessment of human epididymal function by the use of inhibitors in the assay of α‐glucosidase in seminal plasma. Int J Androl 13(4):297–305
Dimauro I, Mercatelli N, Caporossi D (2016) Exercise-induced ROS in heat shock proteins response. Free Radic Biol Med 98:46–55
Drabovich AP, Jarvi K, Diamandis EP (2011) Verification of male infertility biomarkers in seminal plasma by multiplex selected reaction monitoring assay. Mol Cell Proteomics 10(12):M110.004127
Duijvesz D, Luider T, Bangma CH, Jenster G (2011) Exosomes as biomarker treasure chests for prostate cancer. Eur Urol 59(5):823–831
Eddy EM, Toshimori K, O’Brien DA (2003) Fibrous sheath of mammalian spermatozoa. Microsc Res Tech 61(1):103–115
Elzanaty S, Richthoff J, Malm J, Giwercman A (2002) The impact of epididymal and accessory sex gland function on sperm motility. Hum Reprod 17(11):2904–2911
Elzanaty S, Malm J, Giwercman A (2005) Duration of sexual abstinence: epididymal and accessory sex gland secretions and their relationship to sperm motility. Hum Reprod 20(1):221–225
Erata GÖ, Toker NK, Durlanık Ö, Kadıoğlu A, Aktan G, Toker GA (2008) The role of heat shock protein 70 (Hsp 70) in male infertility: is it a line of defense against sperm DNA fragmentation? Fertil Steril 90(2):322–327
Evans JP, Kopf G (1998) Molecular mechanisms of sperm–egg interactions and egg activation. Andrologia 30(4–5):297–307
Fourie M, Du Toit D, Bornman M, Van der Merwe M-P, Du Plessis D (1991) α-Glucosidase, sperm ATP concentrations, and epididymal function. Arch Androl 26(3):139–141
Fraser LR, Harrison R, Herod JE (1990) Characterization of a decapacitation factor associated with epididymal mouse spermatozoa. Reproduction 89(1):135–148
Gaudet P, Livstone MS, Lewis SE, Thomas PD (2011) Phylogenetic-based propagation of functional annotations within the Gene Ontology consortium. Brief Bioinform 12(5):449–462
Goodson SG, Qiu Y, Sutton KA, Xie G, Jia W, O’Brien DA (2012) Metabolic substrates exhibit differential effects on functional parameters of mouse sperm capacitation. Biol Reprod 87(3):75, 71-15
Gosálvez J, González-Martínez M, López-Fernández C, Fernández JL, Sánchez-Martín P (2011) Shorter abstinence decreases sperm deoxyribonucleic acid fragmentation in ejaculate. Fertil Steril 96(5):1083–1086
Goss DM, Ayad BM, van der Horst G, Skosana B, du Plessis SS (2019) Improved sperm motility after 4 h of ejaculatory abstinence: role of accessory sex gland secretions. Reprod Fertil Dev 31(5):1009–1016
Graham J (1994) Effect of seminal plasma on the motility of epididymal and ejaculated spermatozoa of the ram and bull during the cryopreservation process. Theriogenology 41(5):1151–1162
Guerin JF, Ali HB, Cottinet D, Rollet J (1990) Seminal alpha‐glucosidase activity as a marker of epididymal pathology in nonazoospermic men consulting for infertility. J Androl 11(3):240–245
Hamada A, Sharma R, du Plessis SS, Willard B, Yadav SP, Sabanegh E, Agarwal A (2013) Two-dimensional differential in-gel electrophoresis-based proteomics of male gametes in relation to oxidative stress. Fertil Steril 99(5):1216-1226.e1212
Heshmat SM, Mullen JB, Jarvi KA, Soosaipillai A, Diamandis EP, Hamilton RJ, Lo KC (2008) Seminal plasma lipocalin-type prostaglandin D synthase: a potential new marker for the diagnosis of obstructive azoospermia. J Urol 179(3):1077–1080
Huang S, Kuo Y, Lee W, Tsou H, Lee Y, Chang H, Wu J, Yang P (1999) Substantial decrease of heat-shock protein 90 precedes the decline of sperm motility during cooling of boar spermatozoa. Theriogenology 51(5):1007–1016
Huang S, Kuo Y, Tsou H, Lee Y, King Y, Huang H, Yang P, Lee W (2000) The decline of porcine sperm motility by geldanamycin, a specific inhibitor of heat-shock protein 90 (HSP90). Theriogenology 53(5):1177–1184
Intasqui P, Camargo M, Del Giudice PT, Spaine DM, Carvalho VM, Cardozo KH, Zylbersztejn DS, Bertolla RP (2013) Sperm nuclear DNA fragmentation rate is associated with differential protein expression and enriched functions in human seminal plasma. BJU Int 112(6):835–843
Intasqui P, Camargo M, Antoniassi MP, Cedenho AP, Carvalho VM, Cardozo KHM, Zylbersztejn DS, Bertolla RP (2016) Association between the seminal plasma proteome and sperm functional traits. Fertil Steril 105(3):617–628
Iwasaki A, Gagnon C (1992) Formation of reactive oxygen species in spermatozoa of infertile patients. Fertil Steril 57(2):409–416
Jansen S, Ekhlasi-Hundrieser M, Töpfer-Petersen E (2001) Sperm adhesion molecules: structure and function. Cells Tissues Organs 168(1–2):82–92
Kavanagh J (1994) Isocitric and citric acid in human prostatic and seminal fluid: implications for prostatic metabolism and secretion. Prostate 24(3):139–142
Kessopoulou E, Tomlinson M, Barratt C, Bolton A, Cooke I (1992) Origin of reactive oxygen species in human semen: spermatozoa or leucocytes? Reproduction 94(2):463–470
Krause W, Bohring C (1999) Why do we determine α‐glucosidase activity in human semen during infertility work‐up?—Krause—1999—Andrologia—Wiley Online Library. Andrologia 31(5):289–294
Lehavi O, Botchan A, Paz G, Yogev L, Kleiman S, Yavetz H, Hauser R (2014) Twenty‐four hours abstinence and the quality of sperm parameters. Andrologia 46(6):692–697
Levitas E, Lunenfeld E, Weiss N, Friger M, Har-Vardi I, Koifman A, Potashnik G (2005) Relationship between the duration of sexual abstinence and semen quality: analysis of 9,489 semen samples. Fertil Steril 83(6):1680–1686
Li D, Wang T, Wang X (2018) Characterization of sperm proteome and reproductive outcomes after reduced male abstinence in IVF treatment. Fertil Steril 110(4):e296
Lu Q, Sun QY, Breitbart H, Chen DY (1999) Expression and phosphorylation of mitogen-activated protein kinases during spermatogenesis and epididymal sperm maturation in mice. Archiv Androl 43(1):55–66
Mann T (1946) Studies on the metabolism of semen: 3. Fructose as a normal constituent of seminal plasma. Site of formation and function of fructose in semen. Biochem J 40(4):481
Mann T (1967) Sperm metabolism. In: Fertilization. Elsevier, Amsterdam, pp 99–116
Mann T, Lutwak-Mann C (1981) Male reproductive function and the composition of semen: general considerations. In: Male reproductive function and semen. Springer, Berlin, pp 1–37
Marshburn PB, Alanis M, Matthews ML, Usadi R, Papadakis MH, Kullstam S, Hurst BS (2010) A short period of ejaculatory abstinence before intrauterine insemination is associated with higher pregnancy rates. Fertil Steril 93(1):286–288
Marshburn PB, Giddings A, Causby S, Matthews ML, Usadi RS, Steuerwald N, Hurst BS (2014) Influence of ejaculatory abstinence on seminal total antioxidant capacity and sperm membrane lipid peroxidation. Fertil Steril 102(3):705–710
Martínez‐Heredia JEJMBJL, Oliva R (2006) Proteomic identification of human sperm proteins. Proteomics 6:15
Mayorga-Torres BJ, Camargo M, Agarwal A, du Plessis SS, Cadavid AP, Cardona Maya WD (2015) Influence of ejaculation frequency on seminal parameters. Reprod Biol Endocrinol 13:47
Medrano A, Fernández‐Novell JM, Ramió L, Alvarez J, Goldberg E, Montserrat Rivera M, Guinovart JJ, Rigau T, Rodríguez‐Gil JE (2006) Utilization of citrate and lactate through a lactate dehydrogenase and ATP‐regulated pathway in boar spermatozoa. Molecul Reprod Develop Incorporat Gamete Res 73(3):369–378
Miki K, Qu W, Goulding EH, Willis WD, Bunch DO, Strader LF, Perreault SD, Eddy EM, O’Brien DA (2004) Glyceraldehyde 3-phosphate dehydrogenase-S, a sperm-specific glycolytic enzyme, is required for sperm motility and male fertility. Proc Natl Acad Sci 101(47):16501–16506
Milardi D, Grande G, Vincenzoni F, Messana I, Pontecorvi A, De Marinis L, Castagnola M, Marana R (2012) Proteomic approach in the identification of fertility pattern in seminal plasma of fertile men. Fertil Steril 97(1):67–73.e61
Mukai C, Okuno M (2019) Glycolysis plays a major role for adenosine triphosphate supplementation in mouse sperm flagellar movement. Biol Reprod 71(2):540–547
Mukherjee SLPCELRVWCHCLBC, Hooper LV (2009) Regulation of C-type lectin antimicrobial activity by a flexible N-terminal prosegment. J Biol Chem 284(8):4881–4888
Mustafi SB, Chakraborty PK, Dey RS, Raha S (2009) Heat stress upregulates chaperone heat shock protein 70 and antioxidant manganese superoxide dismutase through reactive oxygen species (ROS), p38MAPK, and Akt. Cell Stress Chaperones 14(6):579
NagDas SK, Winfrey VP, Olson GE (2002) Identification of ras and its downstream signalling elements and their potential role in hamster sperm motility. Biol Reprod 67(4):1058–1066
Naz RK, Ahmad K, Kaplan P (1992) Expression and function of ras proto-oncogene proteins in human sperm cells. J Cell Sci 102(3):487–494
Oliva R, de Mateo S, Estanyol JM (2009) Sperm cell proteomics. Proteomics 9(4):1004–1017
Patel S, Skandhan K, Mehta Y (1988) Seminal plasma fructose and glucose in normal and pathological conditions. Acta Eur Fertil 19(6):329–332
Pilch B, Mann M (2006) Large-scale and high-confidence proteomic analysis of human seminal plasma. Genome Biol 7(5):R40
Poliakov A, Spilman M, Dokland T, Amling CL, Mobley JA (2009) Structural heterogeneity and protein composition of exosome‐like vesicles (prostasomes) in human semen. Prostate 69(2):159–167
Primakoff P, Myles DG (2002) Penetration, adhesion, and fusion in mammalian sperm-egg interaction. Science 296(5576):2183–2185
Raziel A, Friedler S, Schachter M, Kaufman S, Omanski A, Soffer Y, Ron-El R (2001) Influence of a short or long abstinence period on semen parameters in the ejaculate of patients with nonobstructive azoospermia. Fertil Steril 76(3):485–490
Rees JM, Ford W, Hull M (1990) Effect of caffeine and of pentoxifylline on the motility and metabolism of human spermatozoa. Reproduction 90(1):147–156
Rodriguez‐Gil J (2006) Mammalian sperm energy resources management and survival during conservation in refrigeration. Reprod Domest Anim 41:11–20
Rotem R, Paz GF, Homonnai ZT, Kalina M, Naor Z (1990) Protein kinase C is present in human sperm: possible role in flagellar motility. Proc Natl Acad Sci 87(18):7305–7308
Said L, Galeraud‐Denis I, Carreau S, Saâd A (2009) Relationship between semen quality and seminal plasma components: alpha‐glucosidase, fructose and citrate in infertile men compared with a normospermic population of Tunisian men—Said—2009—Andrologia—Wiley Online Library. Andrologia 41(3):150–156
Scarselli F, Cursio E, Muzzì S, Casciani V, Ruberti A, Gatti S, Greco P, Varricchio MT, Minasi MG, Greco E (2019) How 1 h of abstinence improves sperm quality and increases embryo euploidy rate after PGT-A: a study on 106 sibling biopsied blastocysts. J Assist Reprod Genet 36(8):1591–1597
Schoenfeld CYARDDL, Numeroff M (1979) Prolactin, fructose, and zinc levels found in human seminal plasma. Fertil Steril 32(2):206–208
Shacoski JC (2012) Heat shock proteins as markers for cellular stress in mammalian sperm. University of California, Davis
Sharma R, Agarwal A, Mohanty G, Du Plessis SS, Gopalan B, Willard B, Yadav SP, Sabanegh E (2013a) Proteomic analysis of seminal fluid from men exhibiting oxidative stress. Reprod Biol Endocrinol 11:85
Sharma R, Agarwal A, Mohanty G, Hamada AJ, Gopalan B, Willard B, Yadav S, du Plessis S (2013b) Proteomic analysis of human spermatozoa proteins with oxidative stress. Reprod Biol Endocrinol 11:48
Shen Z, Shi B, Wang T, Jiao J, Shang X, Wu Q, Zhou Y, Cao T, Du Q, Wang X, Li D (2019) Characterization of the sperm proteome and reproductive outcomes with in vitro, fertilization after a reduction in male ejaculatory abstinence period. Mol Cell Proteomics 18(1):S109–S117
Simpson RJ, Lim JWE, Moritz RL, Mathivanan S (2009) Exosomes: proteomic insights and diagnostic potential. Exp Rev Proteom 6(3):267–283
Storey BT (2004) Mammalian sperm metabolism: oxygen and sugar, friend and foe. Int J Developm Biol 52:427–437
Storey BT, Kayne FJ (1975) Energy metabolism of spermatozoa. V. The Embden-Myerhof pathway of glycolysis: activities of...—Abstract—Europe PMC. Fertil Steril 26(12):1257–1265
Travis AJFJARNAVPEGGLKGS, Moss SB (1998) Targeting of a germ cell-specific type 1 hexokinase lacking a porin-binding domain to the mitochondria as well as to the head and fibrous sheath of murine spermatozoa. Mol Biol Cell 9(2):263–276
Turner RM (2003) Tales from the tail: what do we really know about sperm motility? J Androl 24(6):790–803
Turpeenniemi-Hujanen TMPU, Kivirikko KI (1980) Isolation of lysyl hydroxylase, an enzyme of collagen synthesis, from chick embryos as a homogeneous protein. Biochem J 189(2):247–253
Viljoen MHBMSVDMMP, Du Plessis DJ (1990) Alpha‐glucosidase activity and sperm motility. Andrologia 22(3):205–208
Visconti PE (2012) Sperm bioenergetics in a nutshell. Biol Reprod 87(3):72
Wang J, Zhang HR, Shi HJ, Ma D, Zhao HX, Lin B, Li RS (2009) Proteomic analysis of seminal plasma from asthenozoospermia patients reveals proteins that affect oxidative stress responses and semen quality. Asian J Androl 11(4):484–491
Westhoff D, Kamp G (1997) Glyceraldehyde 3-phosphate dehydrogenase is bound to the fibrous sheath of mammalian spermatozoa. J Cell Sci 110(15):1821–1829
WHO (2010) WHO laboratory manual for the examination and processing of human semen, 5th edn. World Health Organization, Geneva
Williams AC, Ford WCL (2001) The role of glucose in supporting motility and capacitation in human spermatozoa. J Androl 22(4):680–695
Yi Y-J, Manandhar G, Sutovsky M, Li R, Jonáková V, Oko R, Sutovsky P (2007) Ubiquitin C-terminal hydrolase-activity is involved in sperm acrosomal function and anti-polyspermy defense during porcine fertilization. Biol Reprod 77(5):780–793
Zhang M, Wang D, Geng Z, Li P, Sun Z, Xu W (2018) Effect of heat shock protein 90 against ROS-induced phospholipid oxidation. Food Chem 240:642–647
Zylbersztejn DSACDGPTSDMBLSGH, Fraietta R (2013) Proteomic analysis of seminal plasma in adolescents with and without varicocele. Fertil Steril 99(1):92–98
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Electronic Supplementary Material
Supplementary Table 1
(DOCX 23 kb)
Supplementary Fig. 1
(DOCX 3736 kb)
Supplementary Fig. 2
(DOCX 3409 kb)
Rights and permissions
Copyright information
© 2021 Springer Nature Switzerland AG
About this paper
Cite this paper
Goss, D.M., Ayad, B., Vlok, M., Hattingh, S.M., van der Horst, G., du Plessis, S.S. (2021). Proteomics and Biomarker Identification in Improved Sperm Motility Parameters After 4 h of Ejaculatory Abstinence. In: Björndahl, L., Flanagan, J., Holmberg, R., Kvist, U. (eds) XIIIth International Symposium on Spermatology. Springer, Cham. https://doi.org/10.1007/978-3-030-66292-9_48
Download citation
DOI: https://doi.org/10.1007/978-3-030-66292-9_48
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-66291-2
Online ISBN: 978-3-030-66292-9
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)