Abstract
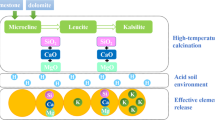
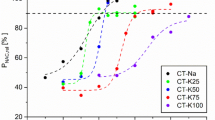
Low-grade phosphate ores are the main phosphorus resources in China, and it’s difficult to upgrade owing to most ores belonging to collophanite with fine-grained dissemination. These inferior ores with high silicon and magnesium contents are favorable raw materials to produce fused magnesium phosphate fertilizer (FMP) for the crop growth. However, high temperature of Ca3(PO4)2–SiO2–MgO system for the smelting of phosphorite resulted in the energy-extensive consumption. In current study, the effect of K2O addition on the smelting of phosphorite was investigated via thermodynamic calculations and experiment validation to reduce the smelting temperature. It has been found that 3–5% K2O can reduce the melting temperature of the Ca3(PO4)2–SiO2–MgO ternary system by about 80 °C. Moreover, K2O addition can enhance the activities of various nutrients such as potassium and magnesium in the FMP products after smelting and water quenching, and the effective conversion of the nutrients is increased by 5% compared with that without K2O addition.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Van Kauwenbergh SJ (2010) World phosphate rock reserves andresources. IFDC, Savannah, pp 16–18
Van Kauwenbergh SJ (2014) Global phosphate rock reserves and resources, the future of phosphate fertilizer. IFDC, Savannah
Ganesapillai M, Simha P, Beknalkar SS, Sekhar DM (2016) Low-grade rock phosphate enriched human urine as novel fertilizer for sustaining and improving agricultural productivity of Cicer arietinum. Sustain Prod Consump, 62–66
Reijnders L (2014) Phosphorus resources, their depletion and conservation, a review. Resour Conserv Recycl, 32–49
PETEA I (2016) Phosphate depletion in china. Fertilizer International, 471:50
Abouzeid AZM (2008) Physical and thermal treatment of phosphate ores-An overview. Int J Miner Process 85(4):59–84
Zafar ZI, Anwar MM, Pritchard DW (1996) Innovations in beneficiation technology for low grade phosphate rocks. Nutr Cycl Agroecosys 46(2):135–151
Izbrodin IA, Ripp GS, Doroshkevich AG (2011) Aluminium phosphate and phosphate-sulphate minerals in kyanite schists of the Ichetuyskoye area, West Transbaikalia, Russia: crystal chemistry and evolution. Miner Petrol 101(1–2):81–96
Hou CH, Miao JY, Gu SY, Wang HB, Wang YY, Xu XC (2019) Innovation of fused calcium magnesium phosphate products to promote industry development. J Plant Nutrition Fertilizer 25(12):2162–2169
Cooper J, Lombardi R, Boardman DI (2011) The future distribution and production of global phosphate rock reserves. Resour Conserv Recycl, 78–86
Cordell D, Drangert J, White S (2009) The story of phosphorus: Global food security and food for thought. Global Environ Chang 19(2):292–305
Luo J, Li G, Rao M, Zhang Y, Peng Z, Zhi Q, Jiang T (2015) Evaluation of sintering behaviors of saprolitic nickeliferous laterite based on quaternary basicity. JOM 67(9):1966–1974
Liu BB, Li RJ, Su SP, Zhang YB (2020) Characterization on the behaviors of Ca and Si constituents during the consolidation of ferruginous manganese ores. In: TMS characterization of minerals, metals, and materials, pp 126–135
Acknowledgements
The authors wish to express their thanks to the National Key Research and Development Program of China (No. 2016YFD0200401, 2018YFC1900200) and the Program for Innovative Research Team (in Science and Technology) in University of Henan Province (No. 19IRTSTHN028) for the financial support.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 The Minerals, Metals & Materials Society
About this paper
Cite this paper
Li, L., Yao, Y., Hou, C., Gu, S., Wang, H. (2021). Preparation of Multifunctional Fused Magnesium Phosphate Fertilizer from Low-Grade Phosphate Ores. In: Li, J., et al. Characterization of Minerals, Metals, and Materials 2021. The Minerals, Metals & Materials Series. Springer, Cham. https://doi.org/10.1007/978-3-030-65493-1_11
Download citation
DOI: https://doi.org/10.1007/978-3-030-65493-1_11
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-65492-4
Online ISBN: 978-3-030-65493-1
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)